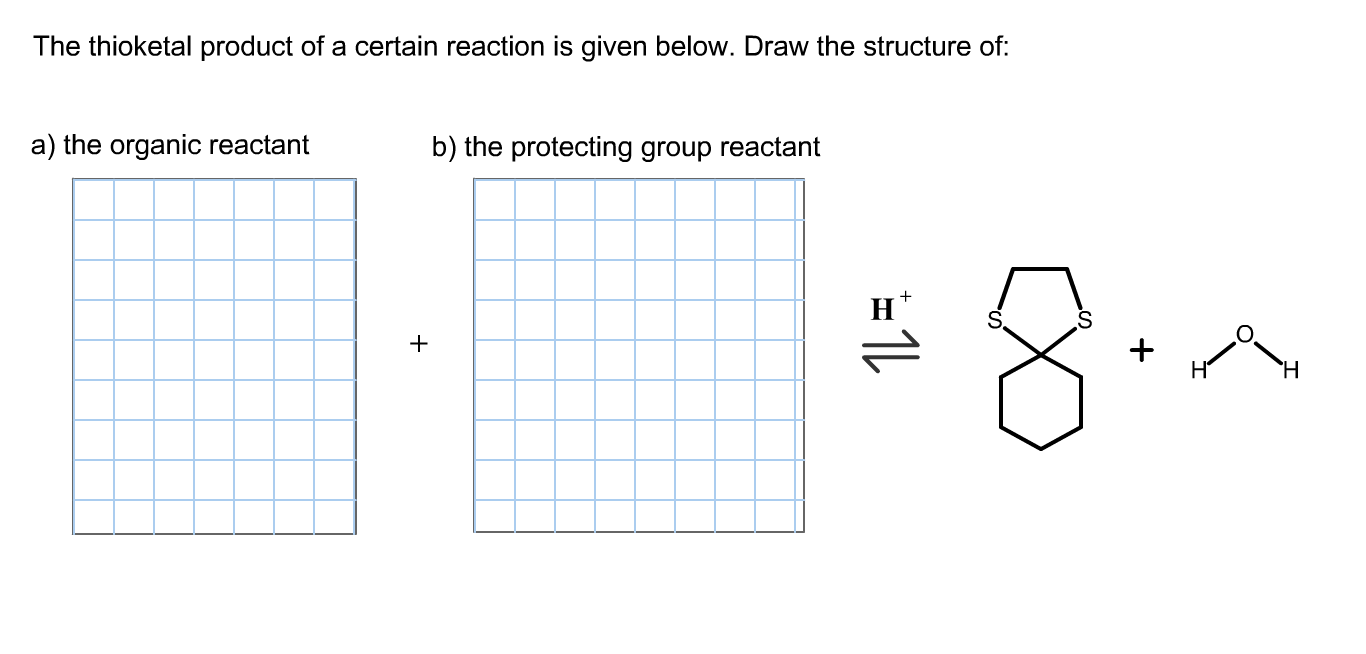
The Thioketal Product Of A Certain Reaction Is Given.
Get ready for a little bit of chemical magic! We're about to dive into the fascinating world of a special molecule that’s the star of a certain chemical reaction. Now, you might be thinking, "Chemistry? Fun?" Absolutely! Think of it like unlocking secret codes or discovering hidden talents in everyday things. This particular product, a thioketal, is like a versatile superhero in the world of organic chemistry, popping up in all sorts of exciting applications. It’s not just a lab curiosity; it’s a real workhorse that helps scientists and engineers create amazing things.
What's the Big Deal with Thioketals?
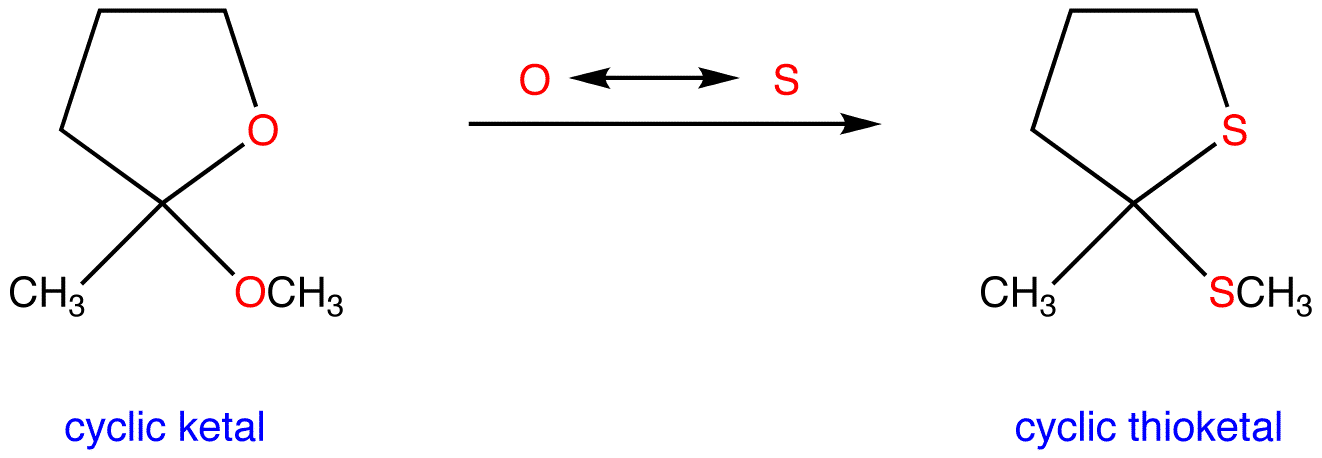
So, what exactly is this thioketal and why should you care? Imagine a regular ketal, which is formed from a ketone (a type of organic molecule) and two alcohol groups. A thioketal is its sulfur-loving cousin! Instead of those oxygen atoms from alcohols, we're swapping them out for sulfur atoms. This simple switch makes a world of difference. Sulfur, being a bit more reactive and having different bonding properties than oxygen, imbues the thioketal with unique characteristics that make it incredibly useful.
The beauty of thioketals lies in their ability to act as protecting groups. In the complex process of synthesizing other molecules, chemists often need to temporarily "hide" or "protect" certain parts of a molecule so they don't react unintentionally while other parts are being modified. Think of it like putting a temporary cap on a chemical functional group. Thioketals are particularly good at this because they are relatively stable under a variety of reaction conditions but can be selectively removed when their job is done, revealing the original group. This precision is key to building complex organic structures efficiently and without unwanted side reactions.
The Many Talents of a Thioketal
But wait, there’s more! Beyond their role as trusty protectors, thioketals are also valuable building blocks themselves. Their sulfur atoms can participate in a range of interesting chemical transformations. For instance, they can be precursors to other sulfur-containing compounds, which are important in many areas, including pharmaceuticals and materials science. The flexibility in how thioketals can be manipulated makes them a chemist’s best friend when designing new synthetic routes.
One of the most significant benefits of using thioketals is the selectivity they offer. In intricate chemical syntheses, where multiple reactive sites might exist on a molecule, being able to control which site reacts and when is paramount. Thioketals allow for this fine-tuning. Imagine building a house; you wouldn't just start hammering nails everywhere. You'd work on the foundation, then the walls, then the roof, in a specific order. Similarly, thioketals help chemists build molecules step-by-step, ensuring that each reaction happens exactly where and when it’s intended.
Furthermore, the formation and cleavage of thioketals are often quite robust processes. This means they can withstand many different chemical environments without breaking down prematurely. This reliability is crucial for long, multi-step syntheses where the protecting group might need to endure several different reaction conditions before it's finally removed. The stability of the thioketal ensures that the protected functional group remains safe until the appropriate stage of the synthesis.
Think about the pharmaceutical industry, for example. Developing new medicines often involves creating very complex organic molecules with precise structures. Even a small mistake can render a drug ineffective or, worse, harmful. Thioketals play a vital role in ensuring the accurate construction of these life-saving compounds. By temporarily masking a reactive carbonyl group (the C=O found in ketones and aldehydes), chemists can perform other necessary reactions on different parts of the molecule without interfering with the carbonyl. Once those other steps are complete, the thioketal can be cleaved, regenerating the carbonyl group in its pristine state.

Beyond pharmaceuticals, thioketals find applications in the synthesis of natural products, agrochemicals, and advanced materials. Their ability to introduce or mask functional groups with high precision makes them indispensable tools for chemists pushing the boundaries of molecular design and synthesis. So, the next time you hear about a thioketal, remember it’s not just a string of chemical jargon, but a testament to the cleverness and precision that makes modern chemistry so powerful and, dare we say, fun!
