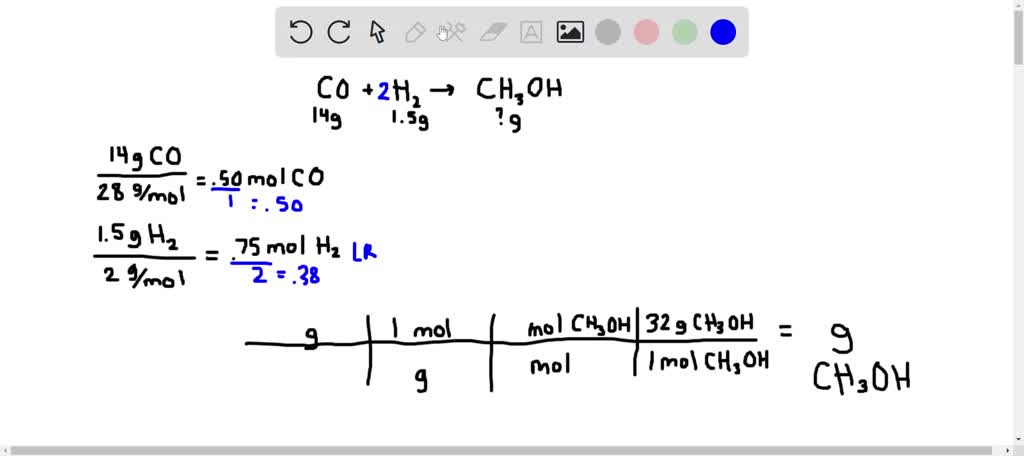
The Synthesis Of Ch3oh From Co And H2 Is Represented
Hey there, super-smart humans! Ever find yourself staring at a perfectly good bottle of hand sanitizer and wondering, "Where did this magic liquid come from?" Or maybe you've seen that little "CH3OH" on a chemical label and thought, "Ooh, fancy science words!" Well, buckle up, because we're about to dive into a little chemical shindig that's way more exciting than it sounds: the creation of methanol from carbon monoxide and hydrogen!
Yeah, I know, "synthesis of CH3OH from CO and H2" sounds like something your chemistry teacher would whisper in a sleepy voice. But trust me, this is where the real fun begins! Think of it as a cosmic recipe, where two unlikely ingredients are combined to make something incredibly useful. It's like taking two boring old socks and turning them into a superhero cape!
The Dynamic Duo: Carbon Monoxide and Hydrogen
So, let's meet our stars. First up, we have carbon monoxide (CO). Now, this guy has a bit of a bad rap. It's a gas, and if you're not careful, it can be a bit… well, dangerous. But in the grand scheme of things, it's just a humble molecule with one carbon atom and one oxygen atom chilling together. Don't let its reputation fool you; it's got a lot of potential!
And then there's hydrogen (H2). This is the universe's most abundant element, the fuel for stars! It's a light, airy gas, and when it teams up with other things, it can create all sorts of amazing substances. Imagine hydrogen as the energetic friend who's always ready for an adventure.
The Chemical Kitchen: Where the Magic Happens
Now, how do these two get together to make something as awesome as methanol (CH3OH)? It's not exactly a casual "hey, let's grab a coffee" situation. This is a more involved process, happening in special chemical kitchens, often called reactors. These reactors are like super-powered ovens, but instead of cookies, they're baking molecules!
To get CO and H2 to bond and form methanol, they need a little nudge. Think of it like trying to get two shy people to start dancing. You need some upbeat music and maybe a friendly introduction. In our case, the "upbeat music" is provided by heat and pressure. We're talking seriously high temperatures and squishing these gases together like they're at a rock concert!

But the real secret ingredient, the choreographer of this molecular dance, is a catalyst. Catalysts are like tiny chemical matchmakers. They help the reaction happen faster and more efficiently without actually being used up themselves. It's like having a really good wingman who makes introductions and gets the party started.
The most common catalyst used in this methanol synthesis is a mixture of copper, zinc oxide, and aluminum oxide. Sounds complicated, right? But really, it's just a team of specialized helpers that guide the carbon monoxide and hydrogen to link up in just the right way.
The Grand Transformation: CO + H2 → CH3OH
So, picture this: we've got our carbon monoxide, our hydrogen, our fiery reactor, and our trusty catalyst. They're all in there, feeling the heat, under pressure, and the catalyst is giving them the ultimate pep talk. And then, poof! A chemical transformation happens.

The carbon monoxide molecule (CO) gets a little makeover. It's like it's saying, "Okay, hydrogen, you and I need to get closer." And the hydrogen molecules (H2) are like, "You got it, CO! Let's make some magic!" Together, with the help of the catalyst and those intense conditions, they rearrange themselves into methanol (CH3OH).
Methanol, my friends, is a super versatile little molecule. It's a clear, colorless liquid, and it's got a ton of uses. You probably have some methanol in your life right now, even if you don't realize it! It's used in everything from making plastics and paints to being a solvent in laboratories. And, as we mentioned earlier, it's a key ingredient in some disinfectants and hand sanitizers. So, next time you're sanitizing your hands, give a little nod to this incredible chemical process!
Why is This So Cool (Besides the Hand Sanitizer)?
Okay, so it makes hand sanitizer. That's pretty neat. But there's more! This whole process of turning carbon monoxide and hydrogen into methanol is a big deal for a few reasons:
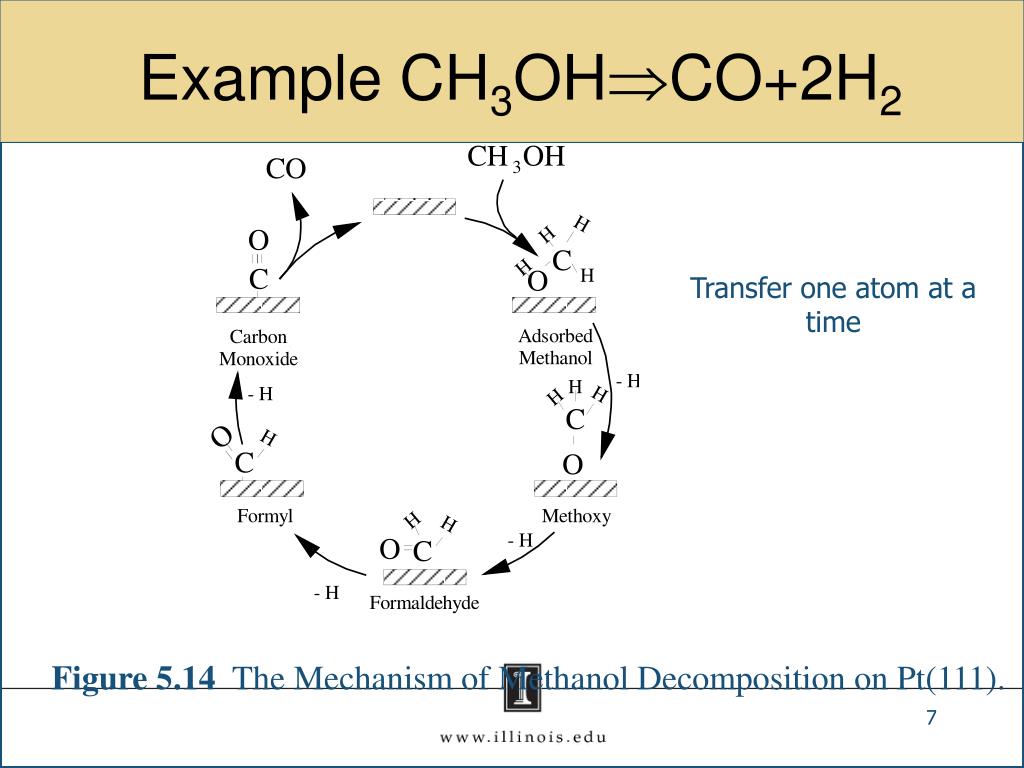
It's about making things from "waste": Sometimes, carbon monoxide can be a byproduct of other industrial processes. Instead of just letting it go, we can capture it and turn it into something valuable like methanol. It's like finding treasure in your recycling bin!
It's a building block for the future: Methanol is a fundamental chemical. It can be further transformed into all sorts of other chemicals and fuels. Think of it as a LEGO brick that can be used to build a whole city of cool stuff. Some people are even looking at methanol as a potential clean fuel for vehicles, which is super exciting for our planet!
It highlights the power of chemistry: This reaction is a beautiful example of how atoms and molecules can be manipulated and rearranged to create entirely new substances with amazing properties. It's a testament to the ingenuity and power of scientific exploration. It’s like the universe decided to play a giant game of molecular Tetris, and they’ve mastered the moves!

Making Life More Fun with Methanol
Seriously, think about it! The fact that we can take humble gases, put them under the right conditions with a little help, and create a liquid that powers industries and keeps us healthy? That's pretty darn fun! It’s about turning problems into possibilities. It's about understanding the hidden forces that shape our world and using that knowledge to make our lives better, and maybe even a little bit cleaner.
So, the next time you see that "CH3OH" or use a product that contains it, remember the incredible journey it took to get there. Remember the carbon monoxide, the hydrogen, the heat, the pressure, and the ever-so-important catalyst. It’s a tiny piece of the vast, amazing puzzle of chemistry that makes our modern world tick. And it’s a reminder that even the most seemingly simple substances have a fascinating story to tell.
Isn't that inspiring? The idea that we can take fundamental elements and, through clever processes, create things that improve our lives is truly remarkable. It’s a call to curiosity, a nudge to explore the world around us with a sense of wonder. So, go on, be a chemical detective! Dive deeper into the world of synthesis and discover the countless other molecular marvels that are shaping our future. The universe is full of amazing reactions just waiting for you to uncover them!
