The Structural Level Of A Protein Least Affected

Hey there! Grab your mug, settle in. We're gonna chat about something super cool, but maybe a tiny bit nerdy. Proteins, right? These little workhorses of our cells. They do everything. Seriously, from building you up to helping you digest that yummy breakfast. But have you ever thought about how they’re built? Like, what makes them, them?
It's not just a jumbled mess of amino acids, oh no. They have structure. And not just one layer of structure, but like, layers of structure. It's kind of like building with LEGOs, but way, way more complicated. And also, alive. Which is, you know, the main difference.
We usually talk about four levels of protein structure. Think of them like the blueprint, the framing, the walls, and the whole darn house. You've got your primary, secondary, tertiary, and quaternary. Each one building on the last, creating this amazing, intricate three-dimensional shape. It's pretty wild when you stop and think about it.
But today, my friend, we're not going to dive into all four. That would be like trying to drink a whole coffee shop's worth of espresso in one go. We're going to focus on the one that’s, dare I say, the toughest nut to crack. The one that’s, like, the bedrock of the whole operation. Ready?
The Unshakeable Foundation: Primary Structure
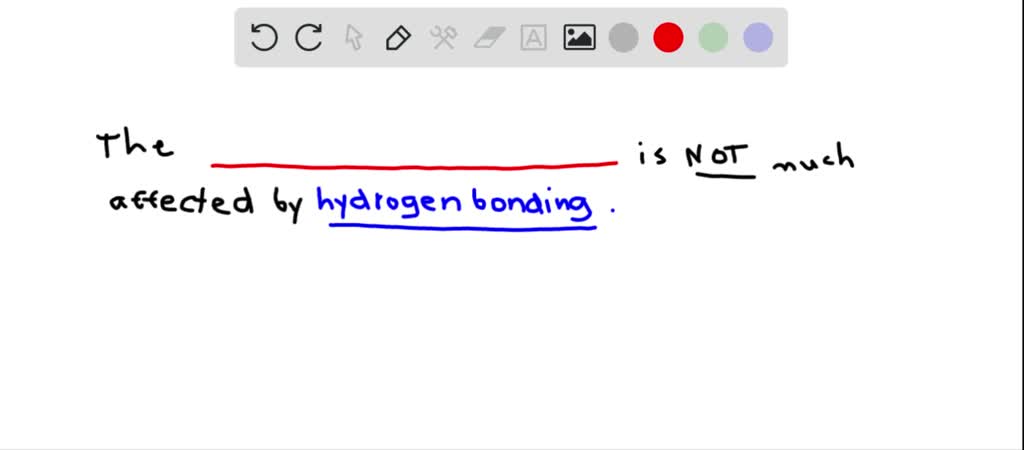
So, let’s start at the very beginning. The absolute, undeniable start. This is your primary structure. What is it, you ask? It’s literally just the sequence of amino acids. Yep, that’s it. Like a string of beads, each bead being a different amino acid. There are 20 common types, by the way, so it's a pretty long alphabet to play with. Pretty neat, huh?
Imagine you’re writing a sentence. The primary structure is like the exact order of the letters. A-T-T, for example, is different from T-A-T, right? Same letters, totally different meaning. It’s the same with proteins. Change even one amino acid in that long chain, and bam! You could have a whole new protein, or worse, a protein that doesn’t work at all. Talk about a typo with serious consequences!
This sequence is determined by your genes. Your DNA tells the cell exactly which amino acid to plop down next. It's like a super-precise recipe. No improvisation allowed here, folks. It’s all about following instructions to the letter. And when I say letter, I mean amino acid. You get it.
So, why is this the least affected, you might wonder? It’s not like it’s invincible, of course. But compared to the fancier structures, it’s pretty darn stable. Think about it. A string of beads. What can really mess with a string of beads? Well, you could cut the string, I guess. Or maybe melt the beads, if they were made of, like, wax. But in a biological context? It's much harder to fundamentally alter that order than it is to mess with how the string folds.
The Strength of the Sequence
Let's get a little deeper, shall we? We're talking about the peptide bonds. These are the chemical links that hold the amino acids together in that chain. They’re pretty strong bonds, folks. Like, really strong. It takes a fair bit of energy and specific enzymes to break them. You don't just accidentally snap a peptide bond while, say, you’re doing the dishes. It’s not that fragile.
Think of it this way: imagine you have a long, beaded necklace. The primary structure is the actual order of the beads on the string. To change the primary structure, you'd have to physically remove a bead and insert a different one. Or maybe you’d have to break the string and re-tie it in a different order. That's a pretty deliberate act, right? It’s not something that happens by accident because you got a little too warm or a little too jostled.
Other levels of protein structure? Oh, they're way more delicate. We'll get to that, but just know that the primary structure is like the foundation of a house. You can redecorate the living room, you can even add an extension, but changing the actual concrete foundation? That’s a whole different ballgame. And usually, a much bigger hassle.
This is why scientists are so fascinated by primary structure. Because if you know the sequence, you know the potential. You know what the protein could become. It's the fundamental blueprint. And if that blueprint is messed up? Well, everything else that follows is going to be a bit wonky, isn't it?
Sometimes, you hear about genetic mutations. These are basically changes in the DNA sequence. And if that DNA sequence codes for a protein, then the primary structure of that protein gets altered. It's like a typo in the instruction manual. Sometimes, it's a minor typo, and the protein still works okay. Other times, it’s a major one, and the protein is completely useless, or even harmful. It’s a stark reminder of how crucial that initial sequence is.
But here's the thing that makes it least affected. While heat, pH changes, or even mechanical stress can totally mess up the higher-level structures (we're talking about folding here, people!), they don't typically break those strong peptide bonds that define the primary sequence. You’d need some serious chemical intervention, or a lot of biological machinery, to actually change the order of amino acids.
So, when you hear about protein denaturation, which is basically proteins losing their shape and therefore their function, what's usually happening? It's the secondary, tertiary, and quaternary structures getting all scrambled. The primary structure? It's usually still hanging in there, just… unable to do its job because its fancy origami has un-origami-ed itself.
Why It's the "Least Affected" Kid on the Block
Let's be real. No structure is truly unaffected. But in the grand scheme of things, the primary structure is the most resilient. Why? Because it's defined by covalent bonds. These are the strongest type of chemical bonds. Think of them as superglue holding the amino acids together. You can’t just pry them apart with a mild suggestion.
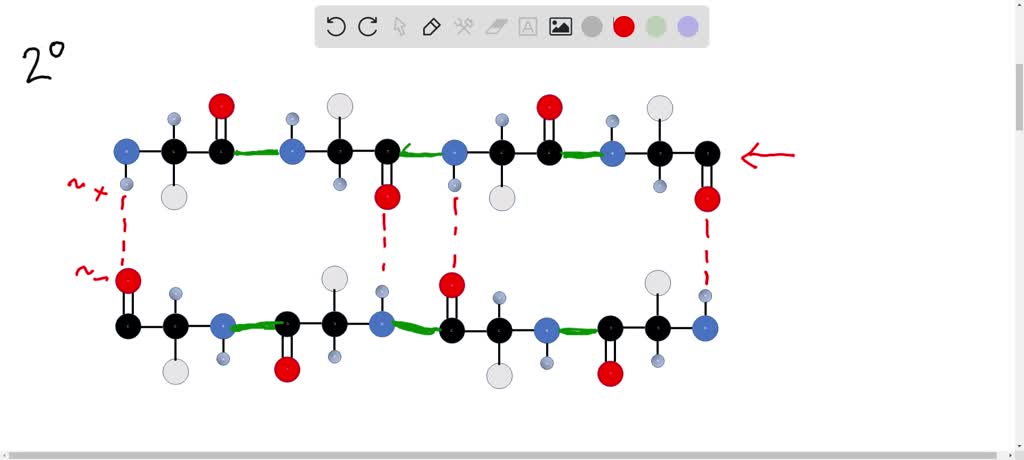
The higher levels of structure, like the secondary (alpha-helices and beta-sheets) and tertiary (the overall 3D fold), are held together by weaker forces. We’re talking hydrogen bonds, ionic interactions, hydrophobic interactions, van der Waals forces. These are like sticky notes and paperclips compared to the superglue of covalent bonds.
So, when a protein gets stressed – maybe by a fever, or a change in stomach acid – these weaker interactions can break. The protein unfolds, like a beautifully folded shirt getting tossed in the wash. It loses its shape, and therefore, its function. It’s a sad day for that protein.
But the sequence of amino acids? The primary structure? Those peptide bonds are still there, holding the chain together. It’s like the shirt is still a shirt, even if it’s all wrinkled and misshapen. The fundamental components are still linked in their original order.

It's like having a really well-built fence. The fence posts (primary structure) are sunk deep and secured with concrete. The wooden planks (secondary and tertiary structure) are attached to those posts. You can bend or break some of the planks if you lean on them too hard. Maybe a storm comes and knocks a few planks loose. But those fence posts? They're gonna take a lot more to move.
This resilience is actually super important. Think about it. If your primary structure was easily messed up, then every time your body temperature went up a degree, you'd be in trouble. Your proteins would be constantly reordering themselves, which sounds… messy. And probably painful. Luckily, evolution is pretty smart.
Plus, the primary structure is the precursor to all the other structures. It's the starting point. If the starting point itself was constantly changing willy-nilly, then there would be no way for the more complex, functional shapes to form reliably. It would be like trying to build a skyscraper when the very first brick you lay keeps changing its composition.
So, while it might sound a little… plain? Just a sequence of letters? This plainness is actually its superpower. It’s the unwavering, the dependable, the absolutely essential starting point. It’s the rock that everything else is built upon. And in the chaotic world of cellular biology, that kind of stability is, well, priceless.
A Little More on Why It's So Tough
Let's ponder this for a sec. Imagine you have a very long sentence written on a piece of paper. The primary structure is that exact sentence. Now, let's say you spill a little water on the paper. The ink might smudge a bit, making some of the letters harder to read. That’s like the higher structures getting a bit messed up. But the actual words? The sequence of letters? They’re still there. You can still decipher the sentence, even if it's a little water-damaged.

Now, what if you wanted to change the actual sentence? You'd have to go in there with an eraser and a pen and deliberately change a letter. Or maybe you'd have to tear out a word and paste in a new one. That’s a much more involved process, right? It's not an accident. It requires intentional effort.
In the biological world, changing the primary structure of a protein typically requires a change in the underlying DNA sequence. This is a mutation. And while mutations happen, they're not usually the result of a protein just chilling out and getting a bit too hot. They're more fundamental genetic events.
So, when we talk about a protein being "affected," it often means its folding is affected. Its 3D shape gets disrupted. But the sequence of amino acids, the primary structure, that’s the core identity. It's like the protein's name tag. You can smudge the name tag, but the name itself is still there, etched in.
It's kind of like the difference between a scar and a broken bone. A broken bone (higher structures) needs time and care to heal and regain its shape. A scar (primary structure, in a way) is a permanent mark, a testament to something that happened, but it doesn't necessarily prevent the underlying tissue from functioning.
The beauty of the primary structure is its predictive power. If you know the sequence, you can often predict how a protein should fold, and therefore what its function should be. It's like knowing the ingredients list on a cake box. You know what's going into it, even before it's baked and frosted.
So, next time you think about proteins, remember the humble primary structure. It might not be the flashiest part, it’s not the part that does all the cool, active folding. But it’s the anchor, the foundation, the unyielding core. It’s the part that’s least likely to be bothered by a little… protein-y drama. Pretty cool, right? Now, go on and finish that coffee!
