The Steady State Assumption As Applied To Enzyme Kinetics Implies
Okay, let's talk about something that sounds super fancy and intimidating: the steady state assumption. Don't worry, it's not as complicated as it sounds. Think of it like this: you're at a really popular coffee shop. The line is long, but somehow, the baristas are keeping up. People are coming in, ordering, getting their drinks, and leaving. And in the middle of all this chaos, there’s this mysterious sweet spot where the number of people in line, the number of people getting drinks, and the number of people leaving is all pretty darn consistent. That, my friends, is basically the steady state assumption, but for tiny things called enzymes.
Now, what does this have to do with enzymes? Well, enzymes are basically nature's little helpers. They're proteins that speed up chemical reactions in our bodies. Think of them as the super-efficient chefs in our internal kitchen. They take ingredients (called substrates) and turn them into something new (called products). And they do it really fast.
So, when scientists are trying to understand how these enzymes work, they often pretend that things are happening in this magical steady state. It’s like they’re looking at that coffee shop and saying, "Alright, let's ignore the initial rush when everyone is first ordering and the final lull when everyone's gone. Let's focus on that busy middle part where things are just… flowing."
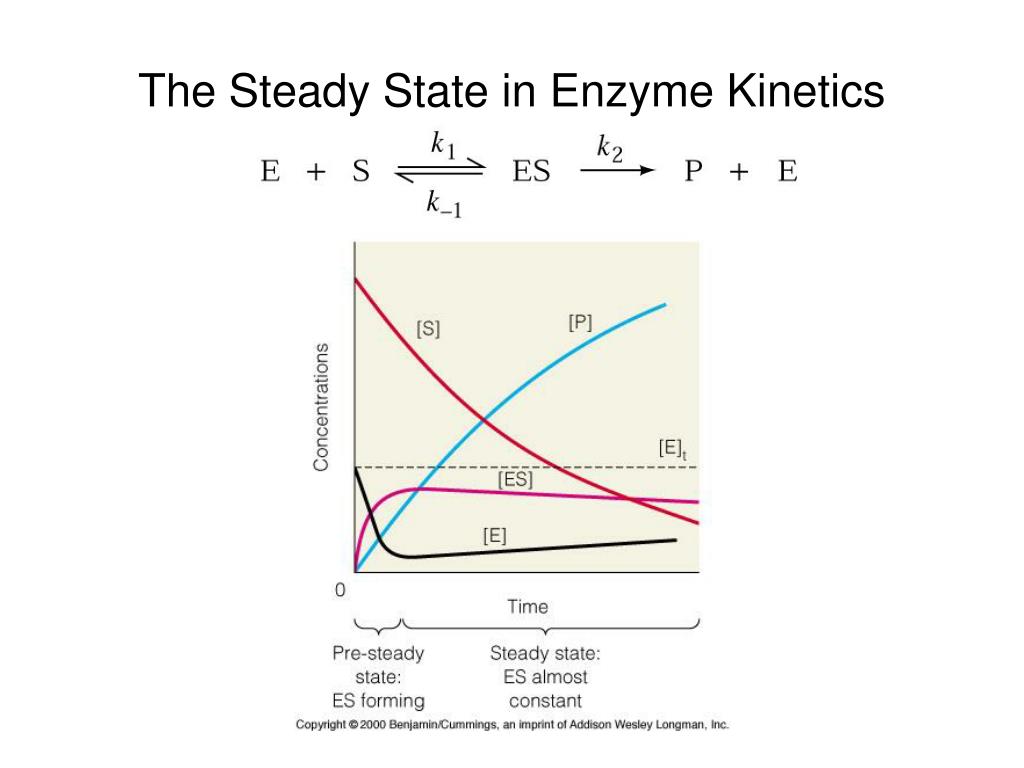
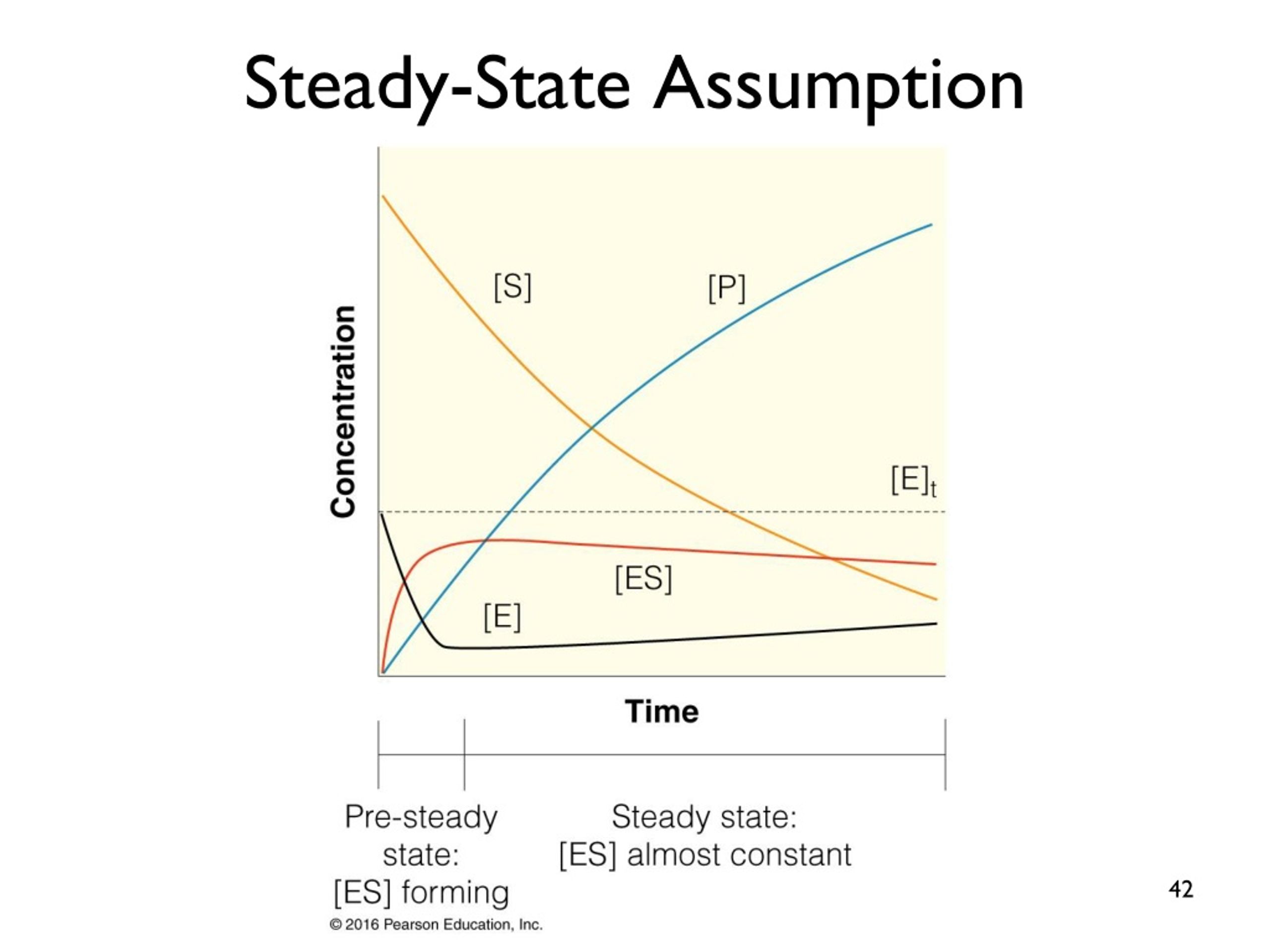
What does this steady state assumption imply? Well, it implies that the amount of that in-between stuff, the enzyme-substrate complex, stays pretty much the same over time. You know that moment at the coffee shop where the barista is grabbing your milk, pouring it, and handing you your cup? That’s the enzyme-substrate complex in action. It's the temporary union of the enzyme and the substrate, getting ready to do its thing. The steady state assumption says that the rate at which these complexes are forming is equal to the rate at which they are breaking down.
It's like saying, "Look, the number of these fleeting little partnerships between the enzyme and the substrate isn't really piling up or disappearing. It's just… there, at a consistent level." This is a huge simplification, but it's a super useful one. It’s the scientific equivalent of saying, "Let's not worry about the exact moment the barista fumbles the lid or when someone gets a free muffin. Let's just focus on the smooth, consistent flow of coffee production."

This handy-dandy assumption allows scientists to come up with some really neat equations. The most famous one is called the Michaelis-Menten equation. Sounds fancy, right? But all it really does is help us understand how the speed of the reaction (how fast the enzyme is working) changes as we add more and more substrate. It's like figuring out how many more coffees the baristas can make if you add ten more people to the line versus adding fifty. You can see the point where adding more people doesn't make things that much faster because the baristas are already working at their max speed. That's the maximum velocity, or Vmax, for the enzyme!
So, the steady state assumption, in a nutshell, implies that we can ignore the messy, dynamic beginnings and endings of an enzyme reaction. We can pretend there’s a stable, predictable period where the formation and breakdown of the enzyme-substrate complex are balanced. This allows us to treat the enzyme system like a well-oiled machine, humming along at a consistent pace. It’s a little bit of scientific wishful thinking, if you ask me, but it works wonders!
It implies that instead of getting bogged down in the intricate details of every single fleeting moment, we can focus on the overall, observable behavior of the enzyme. It's like judging the performance of a band by listening to their entire song, not by analyzing the micro-second pauses between each note. We get the big picture, the smooth rhythm, the overall groove.
And honestly, who doesn't love a good simplification? Life is complicated enough. If we can pretend that our enzymes are just chillin' in a steady state, making things happen smoothly and consistently, then maybe our own lives can feel a little less like a chaotic rush hour commute and a bit more like that perfectly brewed cup of coffee from a well-functioning shop.

So, the next time you hear about the steady state assumption, just picture that busy, but surprisingly organized, coffee shop. It's a little bit of magic, a lot of clever math, and a whole lot of making sense of the tiny, incredibly important work that enzymes do. And if you ask me, that's pretty darn cool.
It implies that we can get away with not tracking every single molecule of the enzyme-substrate complex. Phew!
It's like having a cheat code for understanding complex biological processes. We get to focus on the important stuff: how fast the enzyme works and how it responds to changes in substrate concentration. The steady state assumption is the unsung hero of many a biochemistry lab, quietly allowing for the unraveling of enzyme mysteries without us having to perform calculus on the exact lifespan of every single transient enzyme-substrate complex. And for that, I am eternally grateful.
