The Skeletal Formula Represents What Type Of Alcohol
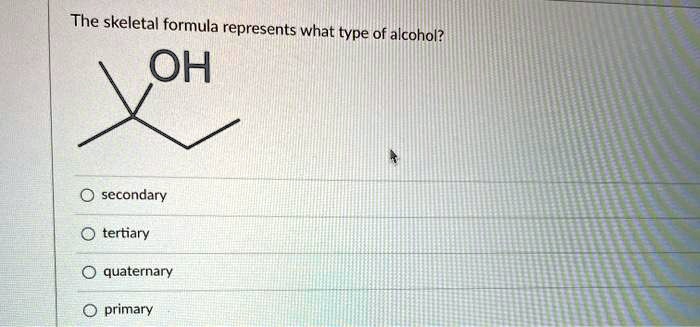
Ever find yourself staring at those weird stick-figure drawings that chemists whip out? You know, the ones with lines and letters all over the place? Sometimes, they look like a secret code for something super complicated, right? Well, today, we're going to decode one of those mysteries: what kind of alcohol does a specific skeletal formula represent?
It sounds a bit daunting, doesn't it? Like you need a PhD in chemistry to even begin to understand it. But honestly, it's way more like solving a fun puzzle than tackling a brutal exam. Think of it like this: those skeletal formulas are just a shorthand, a super efficient way for chemists to draw out the tiny building blocks that make up molecules. They’re like the LEGO bricks of the chemical world, but instead of plastic, we're talking atoms, and instead of snapping them together, they're bonded in specific ways.
So, when we’re talking about an alcohol specifically, what are we even looking for in these drawings? In the grand scheme of things, alcohols are a pretty common and, dare I say, friendly bunch of molecules. You’ve probably encountered them a million times without even realizing it! Think about the stuff that cleans your windows (isopropyl alcohol, aka rubbing alcohol) or the spirit in your favorite drink (ethanol). Yep, those are alcohols!
Chemically speaking, what makes an alcohol an alcohol is a very specific little group of atoms. It's like a secret handshake that these molecules do. This special group is called a hydroxyl group. And how do we represent that in our skeletal formula drawings? It's usually a letter 'O' connected to a letter 'H'. So, you're basically looking for a '-OH' somewhere in that line-drawing party.
Now, here's where it gets interesting. The skeletal formula doesn't just show us if there's an alcohol present. It also tells us where it is and, by extension, a whole lot about the molecule's structure. Remember those lines? Each line represents a chemical bond, and where lines meet, that's usually where an atom hangs out. Typically, the endpoints of lines and the junctions where lines meet represent carbon atoms. This is the super clever shorthand part. They just assume there’s a carbon there and don't bother drawing the letter 'C' unless it’s attached to something else weird.
So, to figure out what type of alcohol we're dealing with, we need to do a little detective work. First, we hunt for that tell-tale '-OH' group. Once we find it, we look at the carbon atom that the '-OH' group is directly attached to. This is the key player, the host of the hydroxyl party!
The type of alcohol is determined by how many other carbon atoms are connected to that particular carbon atom. It's like figuring out someone's social circle. Is this carbon atom the life of the party, connected to tons of other carbons? Or is it a bit more introverted, only hanging out with one or two others?
Primary, Secondary, and Tertiary: The Alcohol Social Ladder
This is where we get our classification: primary, secondary, and tertiary alcohols. It's like a little social ladder for our alcohol molecules!
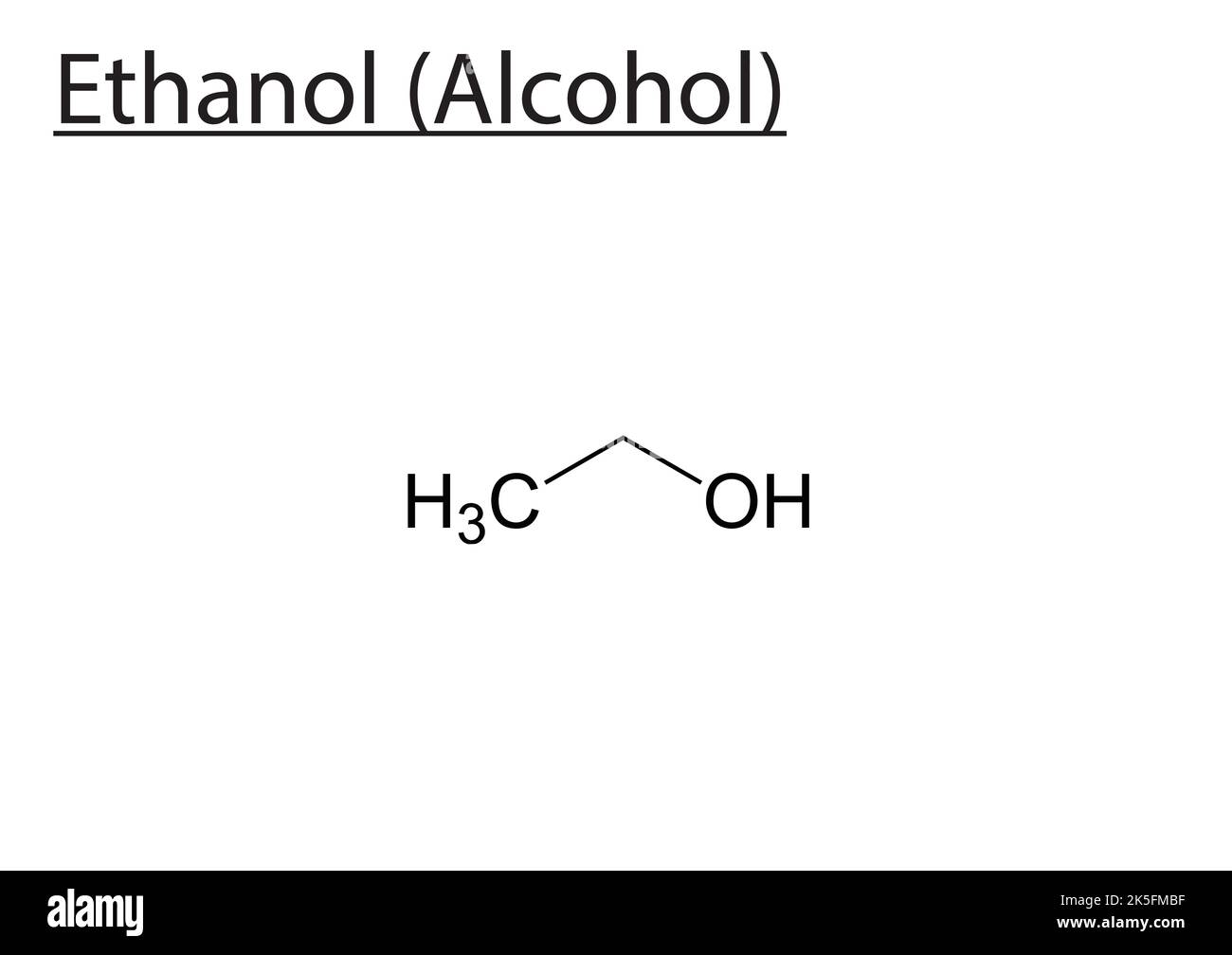
Let's start with the simplest. If the carbon atom that's bonded to the '-OH' group is only attached to one other carbon atom, we call that a primary alcohol. Imagine this carbon is at a party, and it’s only really chatting with one other person (another carbon). The '-OH' group is also there, hanging out with this primary carbon. Think of ethanol (the alcohol in drinks) – its '-OH' is attached to a carbon that's only bonded to one other carbon. So, ethanol is a primary alcohol. Easy peasy, right?
Next up, we have secondary alcohols. In this case, the carbon atom holding the '-OH' group is attached to two other carbon atoms. This carbon is a bit more social, it’s got a couple of carbon buddies to hang out with. Think of rubbing alcohol, or isopropyl alcohol. If you were to draw its skeletal formula, you'd see the '-OH' attached to a carbon that has two other carbons branching off it. It’s like our carbon friend is in a small group conversation, with two carbons on either side.
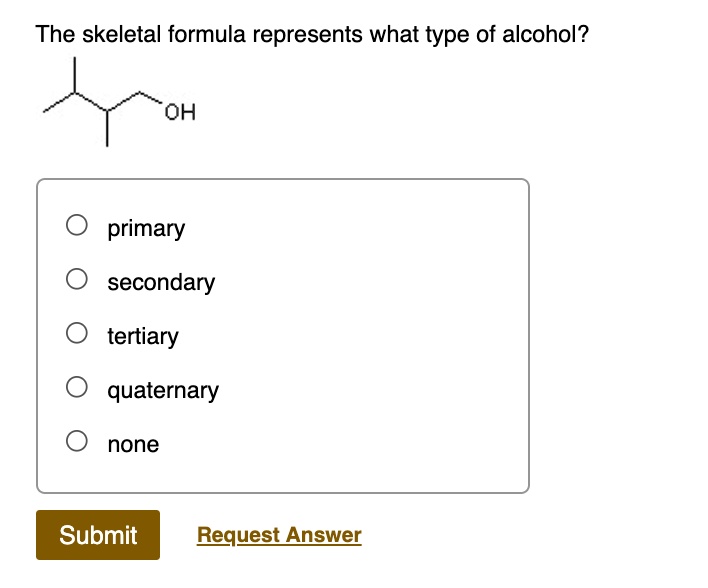
And finally, the most connected of the bunch: tertiary alcohols. Here, the carbon atom bearing the '-OH' group is attached to a whopping three other carbon atoms. This carbon is the ultimate social butterfly, surrounded by carbon friends on all sides! These are often found in more complex molecules and can have different chemical properties compared to their primary and secondary cousins.
Why Does This Even Matter?
You might be thinking, "Okay, so it's primary, secondary, or tertiary. Big deal!" But it's actually a really big deal in the world of chemistry! These classifications tell chemists a lot about how the alcohol will behave. Different types of alcohols react differently in chemical reactions. It's like knowing if you're talking to a shy person or an extrovert – you'd approach them differently, right? Similarly, chemists use the type of alcohol to predict how it will interact with other molecules, what products it might form, and even how stable it will be.

For example, primary and secondary alcohols can be oxidized (think of it as being "burned" or reacting with oxygen in a specific way) to form aldehydes or ketones. Tertiary alcohols, on the other hand, are much more resistant to this kind of oxidation. This difference is super important for synthesizing new compounds in labs and understanding biological processes.
So, next time you see one of those skeletal formulas, don't feel intimidated! You've got the secret decoder ring now. Just look for that '-OH', check out the carbon it's clinging to, and count its carbon buddies. Is it one? Two? Three? That’s your answer! You’re not just looking at lines anymore; you’re seeing the structure, the relationships, and the potential behavior of a molecule. It’s pretty neat, don't you think? You’re basically seeing the personality of the alcohol!
It’s like being able to tell if a character in a book is the quiet scholar, the friendly neighbor, or the energetic leader just by looking at a few key details. The skeletal formula is the character sketch of the alcohol. And understanding this simple classification is the first step to appreciating the vast and fascinating world of organic chemistry. So go forth and decode those stick figures – you’re officially an alcohol detective!
