The Reactivity Of An Atom Arises From __________.
Imagine the universe as a giant playground, and atoms are the energetic little kids running around. They're not just sitting there; oh no, they're constantly itching for some action. And that "itching for action," that buzz of excitement, comes from one super important thing: the number and arrangement of their tiny electrons.
Think of electrons as the atom's social butterflies. They're the ones who decide if an atom wants to mingle, join forces, or maybe even have a little spat with another atom. It's all about making friends and finding their perfect spot in the atomic social scene.
You see, atoms are a bit like people trying to achieve that perfect sense of belonging. They're happiest when their outermost shell, their "personal space," is totally full of these little electron buddies. It’s like having a full party where everyone has a dance partner!
So, if an atom has a few too many electrons in its outer shell, it's like having extra guests at your party who don't quite fit in. It'll be super eager to give some away to a neighbor who's desperately looking for more. This is the birth of a chemical bond, folks! A beautiful exchange.
On the flip side, if an atom is missing a few electrons to feel complete, it’s like having a gaping hole in your party. It'll be practically begging to take electrons from anyone willing to share. It's a cosmic game of "catch" and "give" played at the subatomic level.
And some atoms, like our friends Helium and Neon, are the ultimate party animals. They've already got their perfect, full outer shells. They're so content, they basically wave to everyone from the sidelines, totally uninterested in joining any major shindigs. That's why they're called noble gases – they're too cool for school, too stable for drama!
But for most other atoms, this electron juggling act is the whole reason they get up in the morning (metaphorically, of course!). It’s the engine that drives all the amazing chemical reactions we see, from the baking of a cake to the blooming of a flower.
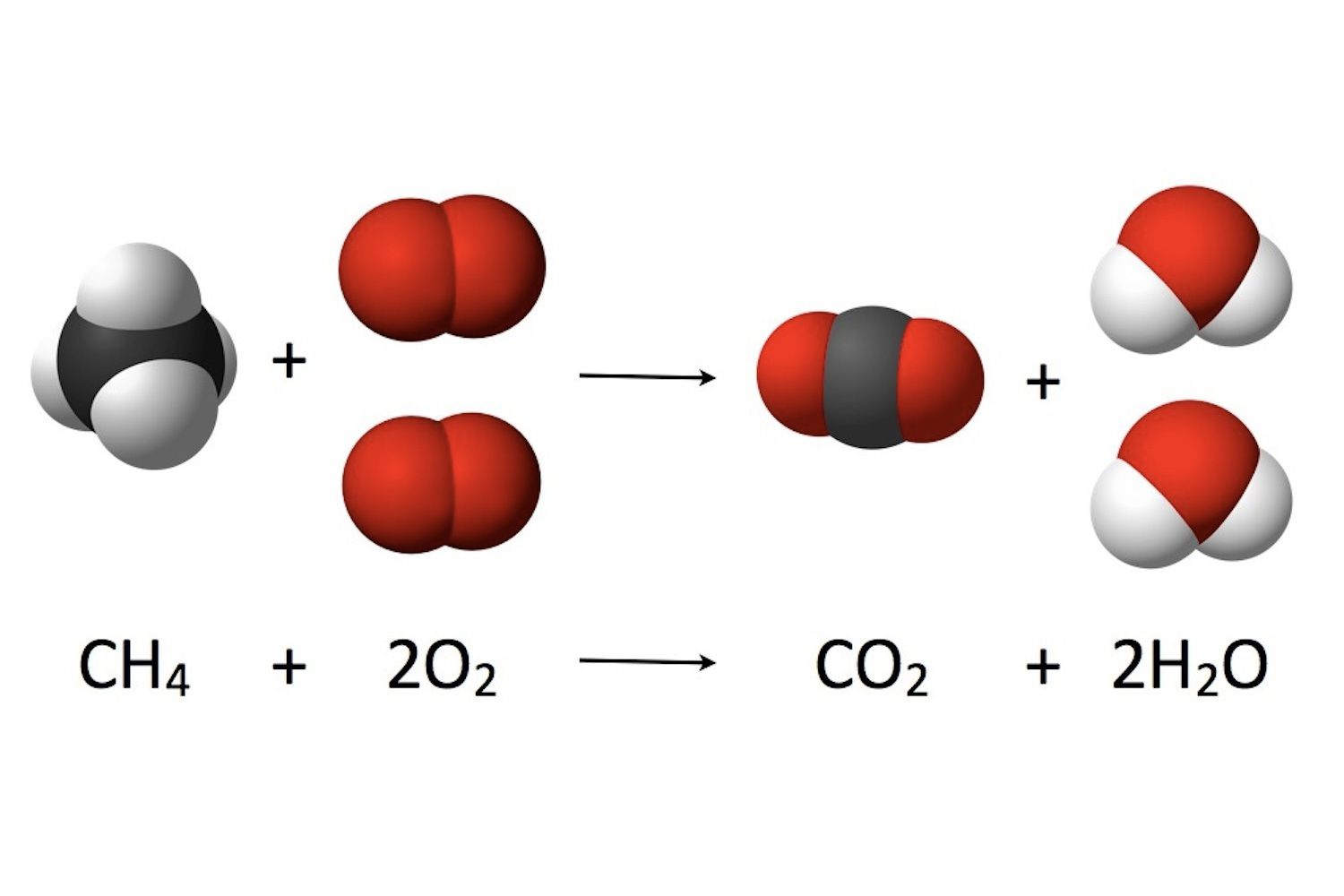
Think about water, the stuff of life itself. It's made of oxygen and hydrogen atoms holding hands, thanks to their electron arrangements. Oxygen is a bit greedy, always wanting more electron hugs, and hydrogen is happy to oblige, eager to share its one electron.
This desire to achieve that "full outer shell" feeling is what makes atoms so incredibly reactive. It’s not random; it’s a fundamental drive, a built-in quest for stability and completeness. Like us, they want to feel settled and secure.
Some atoms are super enthusiastic about making bonds. They're like those friends who are always the first to suggest a game or a new adventure. These are the highly reactive elements, ready to jump into action at the slightest opportunity.
Others are a bit more reserved, like the atoms that only react when things get really interesting. They have outer shells that are almost full, so they don't have as much of an urgent need to make a move. They can afford to be a little picky.

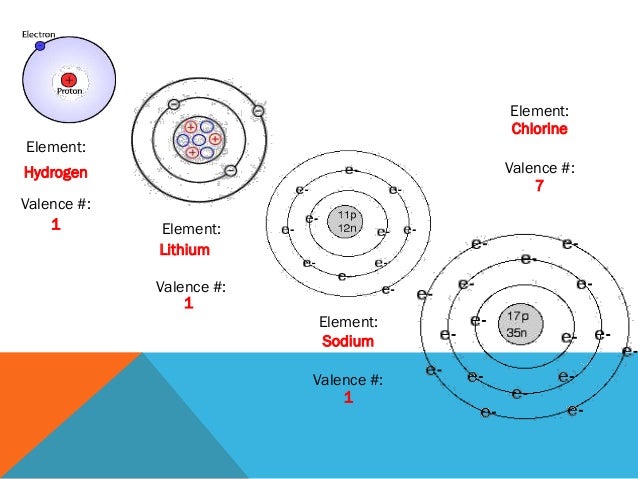
It’s all about the valence electrons, the precious ones in the outermost layer. They are the true decision-makers, the atomic matchmakers. They dictate whether an atom will be a lone wolf or a team player.
Imagine an atom with just one electron in its outer shell. It's like having just one lonely sock. It's desperate to find a mate, to join up with another atom that needs to get rid of one. This eagerness makes it incredibly reactive, like a kid with a new toy, ready to share (or maybe even trade!).
Now, consider an atom that's almost full, just needing one more electron. It's like having your favorite mug and realizing there's just a tiny chip missing. You're probably going to be on the lookout for something to fill that tiny void. This atom will be super keen to grab that missing electron from another atom.
This constant dance of electron giving and taking is what creates all the molecules we know and love. From the air we breathe to the food we eat, it’s all a testament to the reactivity driven by these eager little electrons.
So, the next time you see a chemical reaction happening, whether it's a spectacular explosion (don't try this at home, kids!) or the slow rusting of a bicycle, remember it's all thanks to those energetic little electrons and their quest for the perfect, full outer shell. They are the tiny, unseen architects of our material world!
It’s a beautiful symphony of atomic interactions, all orchestrated by the dance of electrons. They are the secret sauce, the hidden charisma, the very essence of why atoms get along, or sometimes, don't!
Think of it as the atom's way of saying, "I'm feeling a bit incomplete, or maybe a bit too crowded, and I need to do something about it!" This internal feeling, this electron-driven impulse, is the spark that ignites the vast and wondrous world of chemistry.
It’s a fundamental principle, yet so beautifully simple when you look at it this way. The universe is just a giant, interconnected network of atoms, all trying to find their perfect electron-based equilibrium. They’re all playing their part in the grand cosmic ballet.
And what's truly amazing is how this seemingly simple concept leads to such incredible diversity. The same basic rules apply, but the slight differences in electron numbers create everything from soft, fluffy clouds to the hardest diamonds. It's all in the electron arrangement!
So, to sum it all up in a fun, slightly exaggerated way: atoms get their reactivity from their inner longing for a perfectly filled outer shell, like a teenager desperate to be part of the "in" crowd, or a homeowner wanting every room perfectly decorated. They're always looking for that sweet spot, that atomic bliss!
And because of this, they’re not static objects. They are dynamic, always interacting, always changing. This constant motion and interaction is what makes our universe so vibrant and alive. It’s all about the electrons!
The reactivity of an atom arises from the number and arrangement of its electrons, especially those in the outermost shell. This simple fact is the root of all chemical reactions, the driving force behind the formation of everything we can see and touch. It’s the universe’s way of saying, “Let’s make something amazing happen!”
