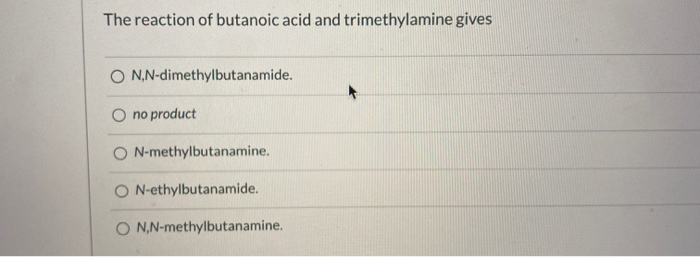
The Reaction Of Butanoic Acid And Dimethylamine Gives

Imagine a kitchen experiment, but instead of baking a cake, we're mixing two rather ordinary-sounding characters: Butanoic Acid and Dimethylamine. You might not have heard of them, but trust me, they have a story to tell, and it's a surprisingly cozy one!
Let's picture Butanoic Acid. Think of it as that slightly tangy, maybe a little bit stinky, ingredient you'd find lurking in some cheeses or even... well, let's just say some bodily odors. It’s not exactly the life of the party, but it has its place in the grand scheme of things. It's got a little bit of that "sour milk" vibe, which sounds unpleasant, but it's actually crucial for giving certain things their signature zing.
Then we have Dimethylamine. This one is a bit more of a lively character. It’s a gas, and it has a rather… distinctive fishy smell. Yes, you heard that right, fishy! So, we've got tangy and fishy characters. Doesn't sound like a recipe for a heartwarming story yet, does it?
But here's where the magic happens, or rather, the chemical reaction. When these two get together, it’s not a dramatic explosion or a fizzy mess. Instead, it’s a gentle, almost tender, coming together. They have a little dance, a chemical hug, if you will.
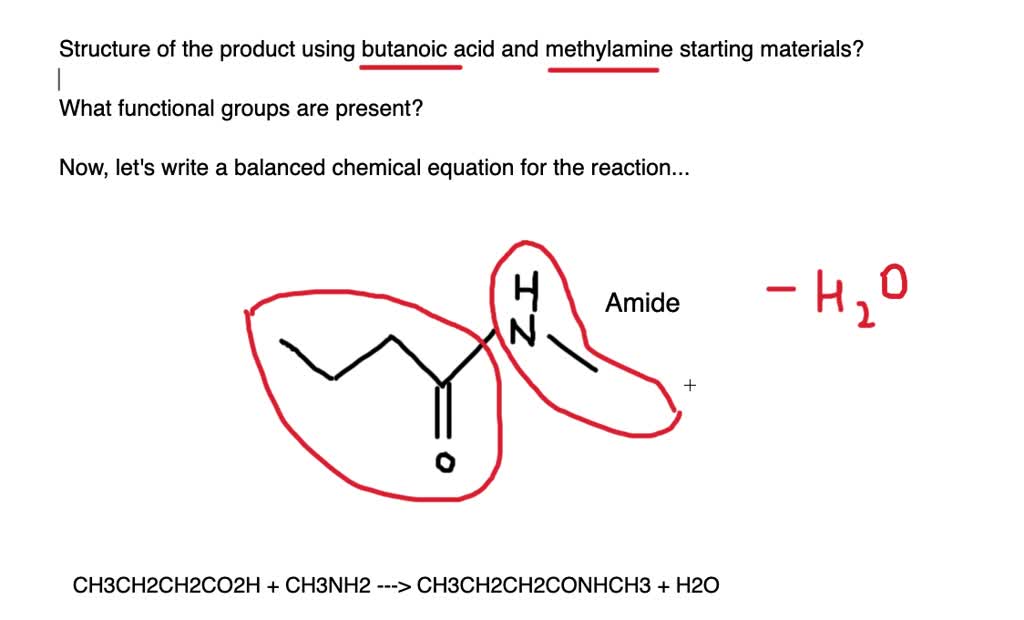
What they create is something quite special. It’s a molecule called N,N-Dimethylbutanamide. Don't let the long name scare you! Think of it as their little creation, their shared legacy.
And what does this little creation do? Well, it turns out to be incredibly useful, particularly in the world of medicine. This is where our story gets heartwarming.
N,N-Dimethylbutanamide, this offspring of tangy and fishy, is a key ingredient in making certain medications. It's like a building block, a tiny but mighty worker that helps create the very things that make us feel better when we're under the weather.
It’s fascinating to think that something derived from ingredients that aren't exactly perfume-worthy can contribute to something so beneficial. It's a reminder that even the most unassuming starting materials can lead to wonderful outcomes.
Think about it: that slightly sour note from Butanoic Acid, perhaps reminding you of a particularly pungent cheese you love, and the unmistakable aroma of a fish market from Dimethylamine, come together to form something that can help alleviate pain or fight off infections.
It's like a quirky fairytale where the grumpy old man (Butanoic Acid) and the eccentric old lady (Dimethylamine) have a surprise success story.

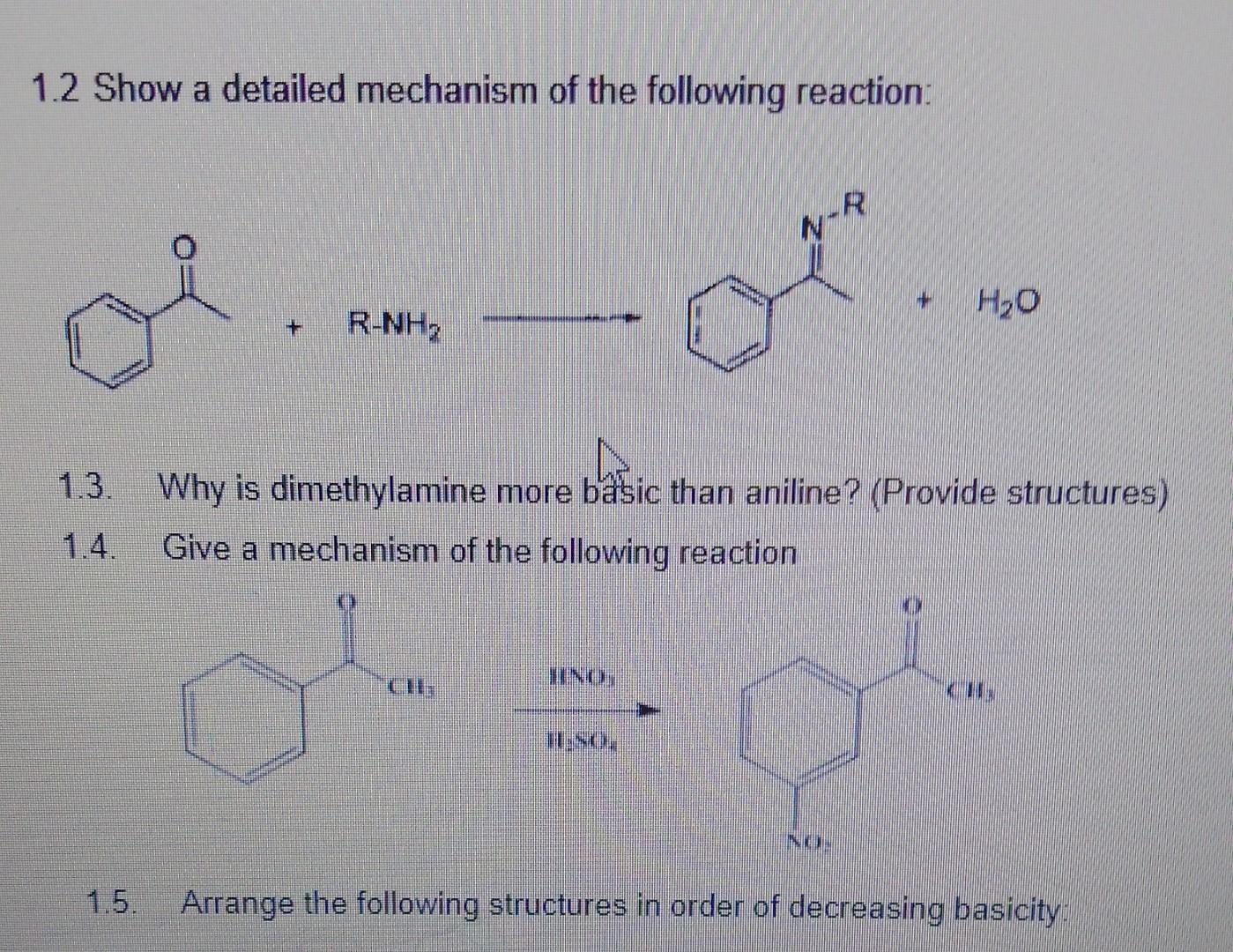
The reaction itself is a classic example of what chemists call an "amide formation." Don't worry, we're not going to dive deep into the electron swapping. Just know that they share something very important – a hydrogen atom from the acid and a part of the amine – and in doing so, they form a strong, stable bond.
This bond creates the amide structure, which is the backbone of our useful new molecule. It's like they decide to combine their best features to create something new and improved.
So, when you hear about Butanoic Acid and Dimethylamine, don't just think of unpleasant smells. Think of the quiet potential, the unexpected partnership.
Think of the journey from a sour dairy product or a bodily aroma, and a whiff of the sea, to a molecule that might be inside the medicine that helps you recover from a cold or manage a chronic condition.
It's a testament to the ingenuity of nature and chemistry. They can take the seemingly mundane and transform it into something truly remarkable.
The beauty of this reaction lies in its simplicity and its profound impact. It’s a straightforward process that yields a complex and vital compound.
This humble reaction is a small cog in a much larger, life-saving machine. It’s the unsung hero of many pharmaceutical processes.
Imagine the chemists, carefully orchestrating this encounter. They know the potential, the hidden promise within these molecules.
They are like matchmakers, bringing together seemingly incompatible partners for a greater good.
It’s a bit like those cooking shows where a chef takes humble ingredients and transforms them into a gourmet masterpiece. Here, the "chefs" are chemists, and their "masterpiece" is medicine.
So next time you encounter a strong smell that reminds you of cheese or fish, take a moment to appreciate the potential. You might be smelling the raw materials for something incredibly important.
It’s a reminder that even in the most basic chemical reactions, there can be a story of purpose and benefit.
The world of chemistry is full of these hidden gems. Reactions that sound technical and intimidating are often at the heart of things we love and rely on.
Butanoic Acid, with its pungent personality, and Dimethylamine, with its fishy charm, are a perfect example of this. They are more than just chemical formulas; they are the starting point for something that genuinely helps people.
So, while the smell might not be appealing, the outcome certainly is. It’s a chemical fairy tale with a very happy, very healthy ending.
The transformation is remarkable. From the everyday, even the slightly unpleasant, to the exceptionally valuable.
It’s a quiet revolution happening in laboratories around the world, all thanks to the dutiful pairing of Butanoic Acid and Dimethylamine.
And in this world of molecules, their union is a testament to how even the most unlikely combinations can create something truly wonderful and life-changing. It's a love story of sorts, a scientific romance that benefits us all.
Who knew that a little bit of tang and a whiff of the sea could be so powerfully good for us?
It’s the kind of surprise that makes you appreciate the intricate beauty of the world around us, even at a microscopic level.
The reaction of Butanoic Acid and Dimethylamine gives us N,N-Dimethylbutanamide, a humble hero in the making of our well-being.
It’s a gentle reminder that the most important things in life often come from the most unexpected places and partnerships.
So, the next time you think of chemistry, remember this little story. Remember the tangy and the fishy, and the beautiful thing they create together.
It’s a sweet ending to a slightly smelly beginning, and that, in itself, is pretty fantastic.
The world of science is full of these charming narratives, where simple ingredients lead to profound discoveries.
And this particular duo, Butanoic Acid and Dimethylamine, offer a heartwarming chapter in that ongoing story.
They are the perfect example of chemistry's ability to surprise and delight, proving that even the most basic reactions can have a profound and positive impact on our lives.
