The Reaction Between Ethyl Bromide And Hydroxide Ion

Imagine a tiny, bustling world inside a beaker, a place where little molecules have personalities and go on adventures. Today, we’re going to peek into one such miniature drama, starring a somewhat shy molecule named Ethyl Bromide and a rather enthusiastic character called the Hydroxide Ion.
Now, Ethyl Bromide is a bit like that friend who's always got a spare jacket but is a little hesitant to join the party. It’s made up of a couple of carbon atoms linked together, a few hydrogen atoms happily attached, and then there's Bromine. Bromine is like the slightly dramatic, sparkly accessory that Ethyl Bromide wears, and it's this very accessory that makes things interesting.
On the other hand, we have the Hydroxide Ion. Think of this one as the life of the party! It’s a small cluster of atoms, and it’s always looking for a good time, or in chemical terms, a chance to react and make something new. It’s got a bit of a negative charge, which makes it quite eager to get involved.
So, picture this: Ethyl Bromide is hanging out, minding its own business, when along comes the energetic Hydroxide Ion. It’s like a curious puppy sniffing around a new toy. The Hydroxide Ion spots that shiny Bromine atom on Ethyl Bromide and thinks, "Ooh, what's this?"
This isn't just a casual hello; it's a chemical rendezvous! The Hydroxide Ion, with its eager negative charge, is naturally drawn to the spot where Bromine is attached to the Ethyl Bromide. It’s a bit like a magnet finding its opposite pole, a dance of attraction that’s been happening in kitchens and labs for ages.
As the Hydroxide Ion gets closer, it starts to nudge the Bromine atom. It’s a gentle but persistent push. You can almost imagine the Hydroxide Ion saying, "Come on, let's try something different!"
And then, something wonderful happens. The Bromine atom, which was feeling a bit overshadowed by its more prominent position on Ethyl Bromide, decides it’s time for a change. It realizes it can find a new adventure elsewhere. It's like deciding to move to a new city for a fresh start.

With a little encouragement from the Hydroxide Ion, the Bromine atom lets go. It detaches itself from the Ethyl Bromide molecule, and off it goes, perhaps to join up with other things and make new friends. It’s a bit of a separation, but it’s a necessary step for what’s to come.
Now, what’s left behind? The Ethyl part of Ethyl Bromide is now free! It’s like the main actor after the supporting character has exited the stage. But it doesn't stay lonely for long.
The Hydroxide Ion, having successfully encouraged Bromine to move on, eagerly steps in to fill the void. It’s like the supportive friend who’s there to pick up the pieces, or in this case, to form a new bond. It's a perfect fit, a chemical embrace.
And voilà! A new molecule is born. The Ethyl group, now free from its bromine attachment, welcomes the Hydroxide Ion. Together, they form something new and exciting called Ethanol. Yes, that’s right, the very same Ethanol that we find in our favorite drinks, or even in hand sanitizers!
So, in a nutshell, this little interaction is how we get Ethanol. It’s a story of friendship, of letting go, and of forming new, wonderful partnerships. It’s a fundamental reaction, but there’s a beautiful simplicity to it, isn’t there?
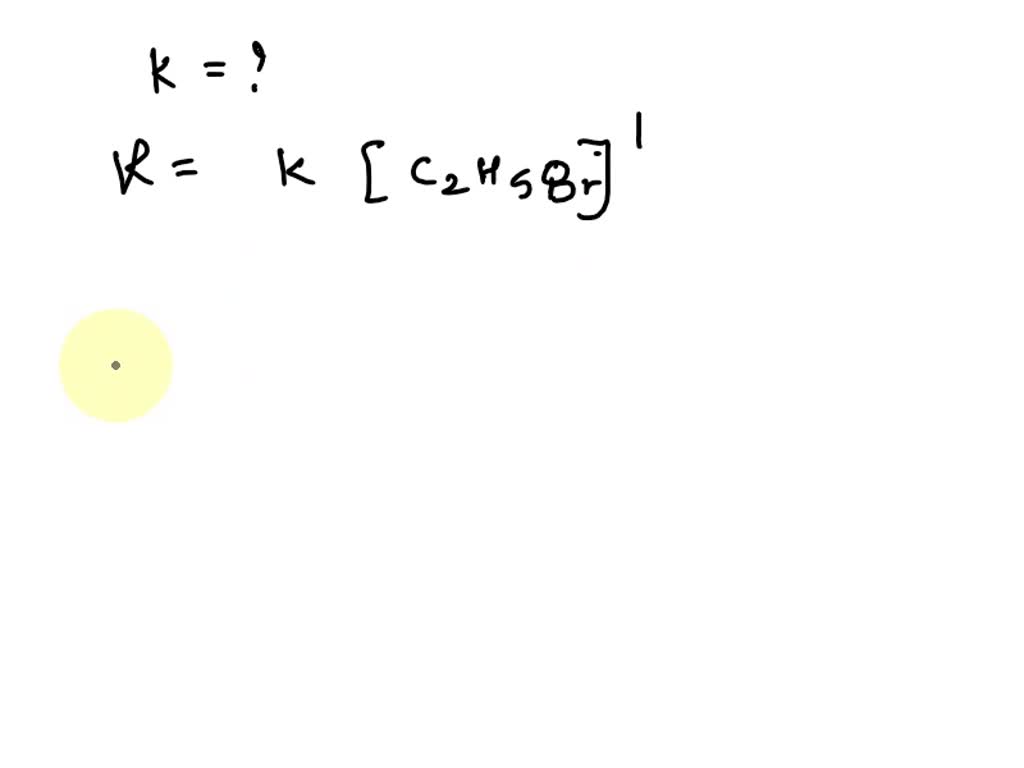
Think about your favorite alcoholic beverage. That warm, fuzzy feeling, the subtle aroma – it all starts with humble molecules like Ethyl Bromide and the ever-ready Hydroxide Ion. They’re the unsung heroes of your happy hour!
It’s also heartwarming to know that even in the microscopic world, change can lead to something so widely enjoyed. The Hydroxide Ion isn’t just a reactive species; it’s a facilitator of deliciousness and disinfectant. Talk about a helpful character!
And what about our departing friend, Bromine? Well, it doesn't just disappear. It often teams up with the ion that accompanied the Hydroxide Ion (usually a sodium or potassium ion) to form a new salt, like Sodium Bromide or Potassium Bromide. So, even the departing guest finds a happy ending!
This particular reaction mechanism is called nucleophilic substitution. Don't let the fancy name scare you. It just means that something with a lot of electron "love" (a nucleophile, like our Hydroxide Ion) attacks something else and bumps off a different group (the leaving group, like our Bromine).
It's a bit like a dance-off. The Hydroxide Ion is the energetic dancer, and the Bromine atom is the one who gracefully exits the floor, making way for the new dance partner. The Ethyl group is the stage, waiting for the next performer.

The beauty of this reaction lies in its versatility. Depending on the conditions, the outcome can be slightly tweaked. But the core story remains: Ethyl Bromide meets Hydroxide Ion, Bromine moves on, and Ethanol is made. It’s a reliable recipe for a common, useful compound.
It’s fascinating to think that the very building blocks of many things we use and enjoy are involved in such elegant, orchestrated processes. The next time you have a drink or use a cleaning product containing alcohol, you can smile and remember the adventurous tale of Ethyl Bromide and the determined Hydroxide Ion.
They might be small, but their impact is enormous. They’re like the tiny gears in a grand clockwork, each playing a vital role in creating the world we experience. This simple reaction is a testament to the intricate beauty of chemistry, unfolding silently in countless test tubes and, ultimately, contributing to the fabric of our everyday lives.
So, there you have it. A little story about a chemical reaction that’s as heartwarming as it is illuminating. It’s a reminder that even the most complex processes can be broken down into simple, relatable narratives, and that chemistry, at its heart, is about connections, transformations, and the creation of something new and wonderful.
The Hydroxide Ion is always on the lookout for an opportunity to make a difference, and the Ethyl Bromide molecule, with its slightly vulnerable Bromine atom, provides just the chance. It's a classic case of right place, right time, and the perfect chemical partners.

And the best part? This happens over and over again, all around us, powering the production of essential substances. It’s a tiny, tireless effort that has a massive effect, a constant hum of molecular activity creating the products we rely on.
So, let’s raise a metaphorical glass to Ethyl Bromide and the amazing Hydroxide Ion. They’re the dynamic duo behind so much more than you might realize, proving that even the smallest interactions can lead to the most significant outcomes.
It's a story of a goodbye and a hello, of a substitution that leads to creation. The Bromine atom leaves, and the Hydroxide Ion arrives, transforming Ethyl Bromide into the familiar and versatile Ethanol. A simple exchange, a profound result.
So, the next time you encounter alcohol, remember the little molecular dance that made it possible. It's a chemical love story, a testament to the exciting transformations that happen when molecules meet and decide to create something new together. Chemistry is all around us, and it's often much more engaging than we think!
