The Range In Size Of Most Atomic Radii Is Approximately

Hey there, curious minds! Ever stop to think about just how incredibly tiny things can get? We’re talking about the building blocks of everything you see, touch, and even are. Yep, we’re diving into the super-duper small world of atoms, and specifically, their sizes. Get ready to have your mind tickled, because the range in size of most atomic radii is, well, pretty mind-boggling!
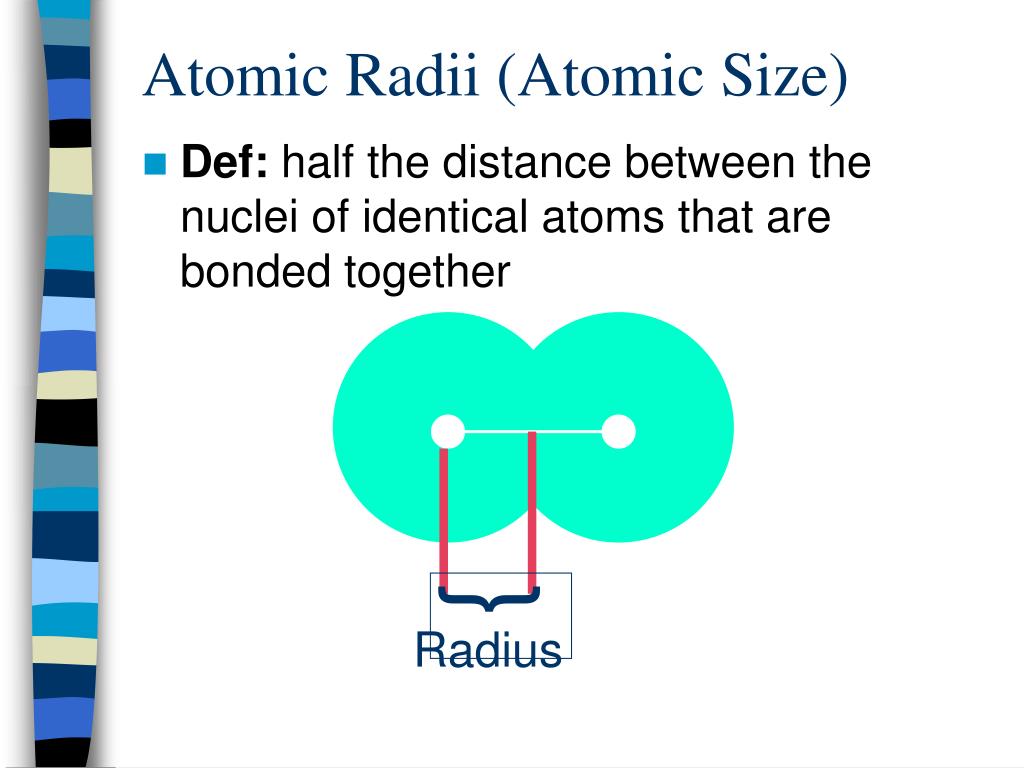
So, what exactly is an "atomic radius"? Think of it as the average distance from the center of an atom (the nucleus, with its protons and neutrons) to the edge of its electron cloud. It's basically how we measure how "big" an atom is. And when we say "big" here, we're talking about something so small it’s almost impossible to picture.
Imagine you have a super-powered magnifying glass, like, way more powerful than anything you’ve ever seen. Even then, you wouldn’t be able to see a single atom with the naked eye. They are that minuscule! And the really fun part? While they’re all unbelievably tiny, they do come in a range of sizes. It’s not like every single atom is the exact same size. Which, if you think about it, is pretty darn interesting, right?
A Universe of Tiny Differences
When we talk about the range of most atomic radii, we’re usually throwing around numbers with a lot of zeros after the decimal point. We’re talking about things like picometers (pm). Now, a picometer is one trillionth of a meter. A trillion! Can you even wrap your head around that number? It’s a 1 followed by 12 zeros. Insane!
So, the smallest atoms, like hydrogen (the simplest of the lot, with just one proton and one electron), have atomic radii around 37 picometers. That’s 0.000000000037 meters. Just let that sink in for a moment. It’s like trying to measure the distance between two grains of sand using the thickness of a single hair. It’s that level of infinitesimal!
On the other end of the spectrum, for the larger atoms you’ll find in the typical periodic table, we’re looking at radii in the ballpark of 200-300 picometers. Think elements like cesium or francium. They’re still mind-blowingly small, but they're almost eight times bigger than a hydrogen atom. That might not sound like a lot in our everyday lives, but in the atomic world, that's a colossal difference!
Why Should This Tiny Difference Make Your Day?
Okay, I hear you. "This is all well and good," you might be thinking, "but how does this tiny difference in atomic size make my life more fun?" Great question! Because understanding these seemingly insignificant differences is the key to unlocking some of the most amazing wonders of the universe. It’s where the magic happens, folks!
Think about it: the way atoms interact, bond, and form molecules – which make up everything – is hugely influenced by their size. The subtle variations in atomic radii dictate everything from the strength of a chemical bond to the color of a pigment. Seriously!
For instance, imagine trying to build with LEGOs. If all your LEGOs were the exact same size, building anything complex would be… well, less interesting, wouldn't it? But you have all sorts of different shapes and sizes of LEGOs, and that's what allows you to create incredible structures. Atoms are like the ultimate LEGOs of the universe, and their size differences are the secret sauce that lets us have things like vibrant flowers, strong metals, and even the complex molecules that make up our own bodies.

Consider the difference between diamond and graphite. Both are made of pure carbon atoms! The only difference? How those carbon atoms are arranged, and their atomic radii play a role in how they can pack together. Diamond is incredibly hard and sparkly, while graphite is soft and used in pencils. All because of how the carbon atoms, with their similar-but-not-identical radii, decide to link up! Isn’t that just fascinating?
And what about the incredible diversity of materials we use every day? From the plastics in your gadgets to the medicines that keep you healthy, they all owe their properties to the precise dance of atoms. The atomic radius, this tiny little measurement, is a fundamental factor in determining whether a substance will be a gas, a liquid, or a solid at room temperature, how it will conduct electricity, or even how it will taste or smell (in the case of molecules!).
It’s this incredible, intricate ballet of size and electron arrangement that gives us the rich tapestry of chemistry and, by extension, the material world around us. So, the next time you admire a beautifully colored gemstone, enjoy a delicious meal, or marvel at the resilience of a bridge, remember that the subtle variations in atomic radii are playing a crucial, albeit invisible, role.

A Window into the Grand Design
The fact that there's a range in atomic radii, rather than a single, universal size, is actually a testament to the elegant, yet complex, rules that govern the universe. It shows us that even at the most fundamental level, there's diversity and variation, which is the very essence of creativity and evolution.
The periodic table, that iconic chart of elements, is organized in a way that reflects these size trends. As you move across a row or down a column, you can often predict how the atomic radius will change. It's like a treasure map of atomic properties! Learning these trends is like learning the secret handshake of the universe’s smallest builders.
And this knowledge isn't just for scientists in lab coats! Understanding these basic principles can give you a whole new appreciation for the world. It’s like suddenly seeing the intricate workings behind a clock – you don’t need to be a clockmaker to appreciate the precision and ingenuity. Similarly, you don’t need to be a quantum physicist to find the size of atoms utterly captivating.
.jpg)
Think of the scientific discoveries made possible by understanding these tiny differences. Materials science, for example, is constantly pushing boundaries by designing new materials with specific properties, all based on how atoms will behave, which is influenced by their size. From super-strong alloys to advanced electronics, it all starts with understanding these fundamental building blocks.
So, while the numbers might seem abstract – 37 picometers here, 200 picometers there – they represent a universe of possibilities. They are the foundation upon which all matter is built, and their subtle variations are the reason for the incredible diversity and complexity we experience every single day.
Isn’t that just the most inspiring thought? The fact that something so utterly invisible, with variations so infinitesimally small, holds the key to so much of what we see and interact with? It’s a beautiful reminder that the grandest designs can be built from the tiniest components. So, don't just let these numbers pass you by. Let them spark your curiosity! Dive deeper, explore the periodic table, and see for yourself the amazing story that atomic sizes have to tell. The universe, in all its tiny, wondrous glory, is waiting for you to discover it!
