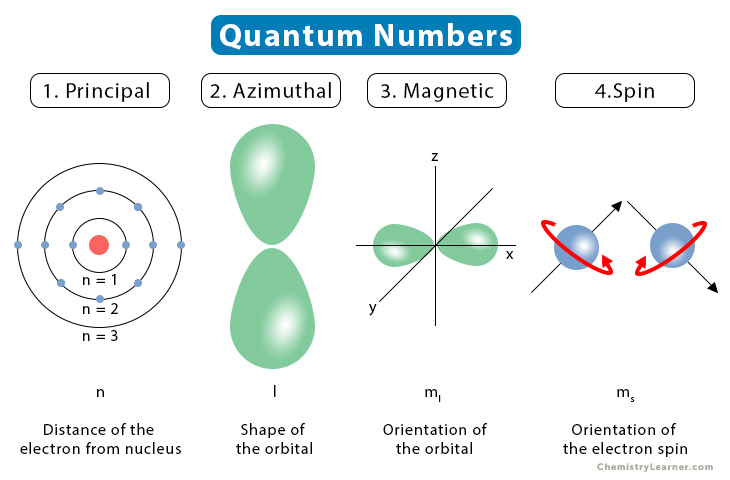
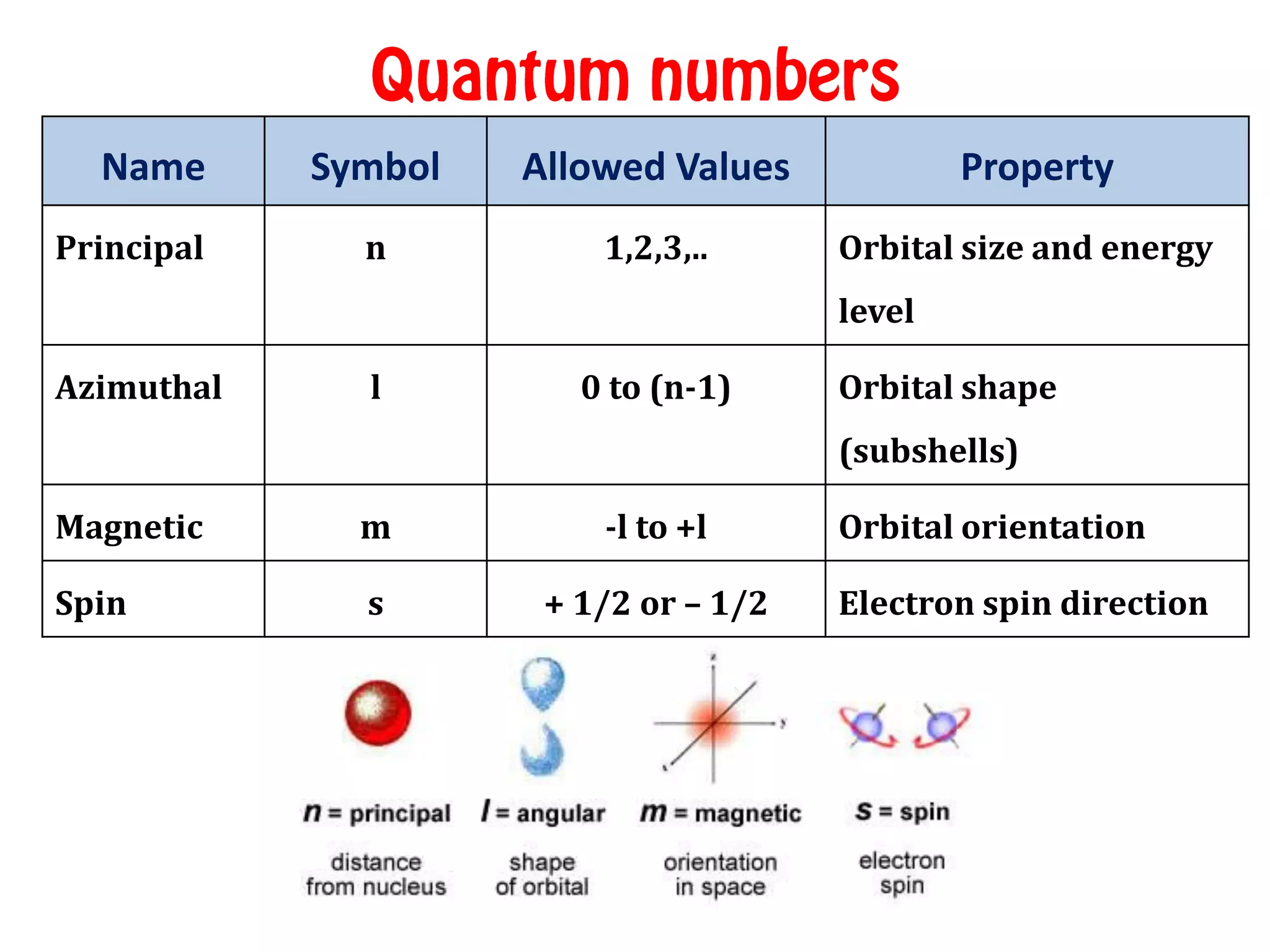
The ________ Quantum Number Defines The Shape Of An Orbital.

Hey there, ever stopped to think about what makes atoms, those tiny building blocks of everything, tick? It's a bit like trying to understand how a really, really fancy origami project is folded. We've got these things called orbitals, which are basically the "neighborhoods" where electrons hang out. And just like our own neighborhoods have different vibes and shapes – some are sprawling suburbs, others are bustling city centers – so do electron orbitals!
Today, we’re going to chat about a special number that tells us all about the shape of these electron neighborhoods. It's called the angular momentum quantum number, or as we'll affectionately call it, the L-number. Don't let the fancy name scare you; it's actually quite intuitive once you break it down.
Imagine a Balloon Party!
Think about a bunch of balloons at a party. You've got your basic, perfectly round balloons, right? Those are like the simplest orbitals, the ones with an L-number of 0. They're nice and symmetrical, like a perfect sphere. You can imagine an electron chilling in there, happily floating in the middle.
But then, as the party gets more exciting, people start twisting and shaping those balloons! You might get a balloon animal, or maybe a couple of balloons tied together in a cute little shape. That's where our L-number starts to change.
So, what’s this L-number all about? It’s a number that can be 0, 1, 2, and so on. Each of these numbers tells us about a different, and increasingly interesting, shape for our electron’s home. It’s like having a remote control for orbital shapes!
The L=0 Orb: The Humble Sphere
When the L-number is 0, we’re talking about what scientists call an 's' orbital. And guess what? It’s that perfectly round, spherical shape we talked about. Imagine a cozy, little ball pit where an electron can play. It’s the most basic shape, the foundation for all the other fancy ones. Think of it as the atom’s version of a comfy armchair – simple, reliable, and gets the job done.

Why should we care about a sphere? Well, even the simplest shapes are super important! The way electrons are arranged in these spherical orbitals dictates how atoms interact with each other. It's the bedrock of chemistry, the reason water is wet and sugar is sweet.
The L=1 Duo: The Dumbbell Dance
Now, let’s crank up the L-number to 1. This is where things get a bit more exciting. These are called 'p' orbitals, and they’re not round anymore! Imagine taking two of those spherical balloons and squishing them together at one end, then pulling them apart at the other. What you get is a shape that looks a bit like a dumbbell, or sometimes like a tiny, two-lobed peanut.
These p orbitals come in three different orientations, pointing along the x, y, and z axes of a 3D space. So, instead of one cozy armchair, our electron might have three little rooms to choose from, each shaped like a dumbbell. It's like having three separate dance floors, each with its own beat!

Why is this dumbbell shape so cool? These p orbitals are crucial for forming chemical bonds. When atoms want to share electrons and stick together to make molecules, these dumbbell-shaped orbitals are the ones doing a lot of the heavy lifting. Think about how Lego bricks connect – the p orbitals are like the special connectors that allow atoms to build all sorts of complex structures, from the air we breathe to the proteins in our bodies.
The L=2 Delights: The Four-Leaf Clovers and More!
Ready for some real artistry? When the L-number is 2, we enter the realm of 'd' orbitals. And oh boy, are they diverse! These shapes are much more complex. Some of them look like four-leaf clovers, with four lobes. Others look like a dumbbell with a donut around the middle.
There are five different 'd' orbitals, each with its own unique and beautiful shape. Imagine a garden with five different kinds of fancy flower beds, each designed for a specific purpose. These are the real show-stoppers in the orbital world!
These d orbitals are particularly important for transition metals – you know, those elements in the middle of the periodic table like iron, copper, and gold. The unique shapes of their d orbitals allow them to form an incredible variety of compounds and play vital roles in many biological processes. Think of the vibrant colors in a peacock's feather or the way our blood carries oxygen; d orbitals are often involved in making these amazing things happen.
The L=3 Fantasies: The G-Force of Orbitals!
And then, for the truly adventurous, we have the L-number of 3, the 'f' orbitals. These are even more intricate and complex, with shapes that start to look a bit like a jumble of lobes. They're like the abstract sculptures of the orbital universe. These are typically found in the inner electrons of heavier elements, and while they’re a bit harder to visualize, they still play their part in the grand scheme of atomic behavior.
The further up the L-number goes, the more complex and varied the orbital shapes become. It’s like a painter adding more and more colors and brushes to their palette to create increasingly detailed masterpieces.
Why Does This Shape Stuff Matter to You?
Okay, so we're talking about shapes of electron homes. Why should you, sitting there with your coffee or tea, care about the L-number and its fluffy, dumbbell-like, or clover-like creations? Because everything around you is a direct result of these shapes!
The way light interacts with materials, creating the colors you see? That's influenced by electron orbital shapes. The way drugs bind to proteins in your body to heal you? Again, it’s all about how these orbitals can fit together and interact. The very structure of DNA, the blueprint of life? It's built upon the foundation of how atoms, with their specific orbital shapes, bond together.
Understanding these shapes isn't just for the brainy folks in lab coats. It’s the secret sauce behind how the universe works at its most fundamental level. It’s about appreciating the elegant complexity hidden within the seemingly simple building blocks of our reality.
So, the next time you see a perfectly round ball, a dumbbell-shaped weight, or even a four-leaf clover, take a moment to remember the L-number! It’s the unsung hero that defines the fascinating shapes of electron orbitals, and in doing so, shapes the entire world we live in. Pretty neat, right?
