The Product From The Alpha Decay Of 235 92u Is

Alright, buckle up, buttercups, because we're about to dive into the wacky, wonderful world of radioactive decay! Today's star of the show is a rather famous element, a true heavyweight in the nuclear arena: Uranium-235, or as its cool kids call it, 235U. This energetic atom, with its atomic number of 92 (that’s a whopping 92 protons, mind you!), is feeling a bit… unstable.
Imagine Uranium-235 as a balloon that's just a tad overinflated. It's got all this energy packed inside, just itching to escape. So, what does it do? It decides to shed some of its heft, like a celebrity dropping a few pounds before a big red carpet event. This grand exit is called alpha decay, and it’s a pretty dramatic way to go!
Now, when 235U undergoes this magnificent alpha decay, it doesn't just poof into thin air. Oh no, that would be far too simple! It actually ejects a tiny, but significant, particle. Think of it like this: if Uranium-235 were a grumpy old man complaining about his aches and pains, he'd be tossing out a very specific handful of marbles.
This ejected particle is none other than an alpha particle. And what, pray tell, is an alpha particle? It’s basically a tiny, energetic bundle consisting of two protons and two neutrons. It’s like a miniature helium nucleus, zipping away with all the gusto of a race car leaving the starting line. Vroom!
So, our overinflated balloon, 235U, lets go of its alpha particle. It’s a moment of great relief for the uranium atom, a sigh of nuclear contentment. But what's left behind? This is where the real magic happens, folks!
The Uranium-235, now lighter and much more serene, transforms into something completely new. It’s like a caterpillar munching its way through the world, and then, voilà, a beautiful butterfly emerges! Our former radioactive heavyweight is now a different element altogether.
The product of this alpha decay is a brand new atom, a descendant born from the energetic shedding of its parent. This new element is called Thorium. Yes, you heard that right, Thorium! It’s a name that sounds like it belongs in a superhero comic book, doesn't it?

Specifically, the Thorium that pops into existence is Thorium-231, or 231Th. It’s got two fewer protons than Uranium-235 (remember, the alpha particle took two protons with it!), and its atomic mass has also decreased accordingly. It’s like losing a couple of kilos and feeling fabulous about it!
So, to recap our thrilling tale: 235U, feeling a bit antsy and ready for a change, decides to perform an alpha decay. It bravely shoots off an alpha particle, which is basically two protons and two neutrons having a grand old adventure. And what do we get as the glorious result? A brand spanking new element called Thorium-231!
Think of it like baking a cake. You start with a bunch of ingredients (protons and neutrons), and when you bake it (decay), you get a delicious cake (Thorium-231). Except in this case, the "baking" involves a tiny bit of radiation and a whole lot of atomic rearrangement. Way more exciting than my grandma's fruitcake, I promise!
And the beauty of it all is that this process is happening all the time, all over the universe. These atomic transformations are the universe's way of constantly remixing and reinventing itself. It’s a cosmic dance of elements, and 235U and Thorium are just two of the main dancers!

It’s pretty mind-boggling to think that a simple act of radioactive decay can lead to the creation of entirely new substances. It’s like a magic trick, but with real science behind it! The universe is full of these amazing, invisible transformations, and we're just peeking behind the curtain to see one of them.
So, the next time you hear about 235U, remember its role as the energetic parent. And when you think about the product of its alpha decay, give a little cheer for Thorium-231, the awesome offspring that emerges from this atomic fiesta. It’s a testament to the ever-changing, ever-surprising nature of matter!
It’s like watching a toddler give away a toy they’ve outgrown. They’re a little sad for a moment, but then they find a new, even more exciting toy to play with! In this case, 235U happily transitions to its new, stable form, and Thorium-231 gets to start its own atomic journey.
This whole process is a beautiful illustration of how elements can change and evolve. It’s not just about sticking around forever; sometimes, it’s about letting go and transforming into something different. And that, my friends, is a lesson we can all learn from, whether we’re talking about atoms or life itself!
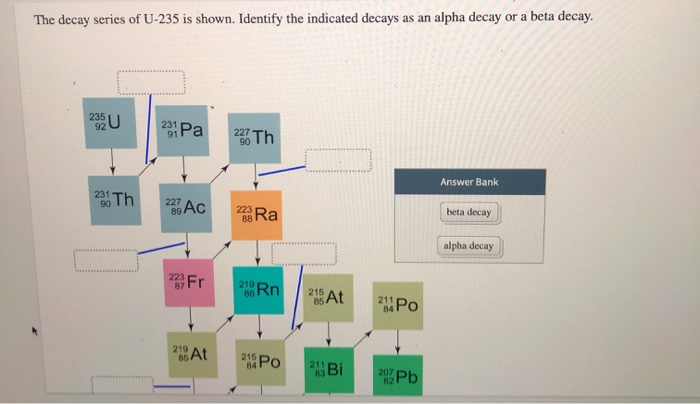
So, there you have it! The product of the alpha decay of 235U is Thorium-231. It’s a bit of an atomic family reunion, with a whole lot of energy involved. Pretty neat, right? The universe, as it turns out, is one giant, exciting science experiment!
Isn't it just fantastic? We start with a heavy-duty element like Uranium, it sheds a bit of its atomic baggage, and poof – we have a different element, ready to take on its own destiny. It’s a cycle of creation and transformation, happening on a scale so tiny it’s almost impossible to comprehend, yet its impact is colossal.
Consider it the ultimate atomic makeover! Uranium-235 is like that old outfit you’ve had for ages, and alpha decay is the brave decision to donate it to someone who can use it. And the result? A fresh, new look in the form of Thorium-231, ready to strut its stuff on the nuclear stage.
This isn't just some dusty old scientific fact; it’s a peek into the very building blocks of our universe and how they interact. It’s a reminder that everything is in constant flux, always changing, always becoming something else. And that, in my book, is downright exhilarating!

So, let’s give a round of applause for 235U and its magnificent transformation into Thorium-231. It's a stellar example of nuclear alchemy in action, and it happens every single moment, reminding us of the dynamic and ever-evolving nature of the cosmos. It’s a party, and everyone’s invited, even if you can’t see it!
The universe is indeed a playground of particles, and 235U is one of the most exciting toys. When it decides to share its energy, it gifts us with Thorium-231, a new player ready to join the atomic games. It’s a beautiful, energetic exchange that keeps the universe humming with activity.
So, the next time you're pondering the mysteries of the universe, just remember this simple, yet profound, transformation. It's a story of shedding the old to make way for the new, powered by the incredible forces within the atom. And the star of this particular chapter? Our wonderful Thorium-231!
It’s like a cosmic shedding of skin, a renewal that’s fundamental to how matter behaves. And the result of this particular shedding is a strong, new element, ready to embark on its own atomic adventure. It’s a continuous cycle of change, a testament to the vibrant and dynamic nature of reality.
So, the product from the alpha decay of 235U is, in fact, Thorium-231. It's a delightful example of nuclear transmutation, a process that’s both fundamental and endlessly fascinating. It's a little bit of atomic magic, making the universe an even more wondrous place to explore!
