The Principal Force Driving Movement In Diffusion Is

Ever wondered why your amazing perfume scent fills the entire room, even though you only sprayed a little bit on your wrist? Or how that teabag magically colors your water, no teabag in every drop, of course! It's not magic, it's diffusion, and the real star behind this everyday phenomenon is surprisingly simple, yet incredibly powerful: a little something called the concentration gradient.
The Invisible Push: Why Things Spread Out
Think about it. We're surrounded by movement. From the air we breathe to the way our bodies work, things are constantly moving and mixing. Diffusion is a fundamental process that explains a huge chunk of this movement. It’s how nutrients get into your cells, how waste products leave them, and how even the flavors in your food spread throughout your meal. Understanding diffusion isn't just for scientists in lab coats; it's about understanding the world around you on a much deeper, and dare we say, cooler level.
The benefits of grasping this concept are pretty awesome. For starters, it helps demystify a lot of everyday observations. It explains why if you spill a drop of ink in water, it eventually disperses to color the whole glass. It’s also crucial for understanding biological processes. For example, how do plants absorb carbon dioxide from the air? Diffusion! How do our lungs exchange oxygen and carbon dioxide? You guessed it, diffusion! Even things like osmosis, which is vital for keeping our cells healthy and hydrated, is a special case of diffusion. So, it's not just about interesting facts; it's about the very mechanics of life and the world.
The Heart of the Matter: The Concentration Gradient
So, what's this elusive force that makes things spread out? Drumroll, please... it's the concentration gradient! Now, that might sound a bit scientific, but let's break it down in a way that's as easy as breathing (which, incidentally, involves diffusion!).
Imagine you're at a concert, and the band just played an amazing encore. Everyone in the middle of the crowd is packed in super tightly, right? There's a very high concentration of people there. But as you move towards the edges of the venue, the crowd thins out. There are fewer people, meaning a lower concentration. The difference in how packed people are between the center and the edges is what we call a gradient. In diffusion, instead of people, we're talking about particles – like molecules of perfume, or sugar, or oxygen.
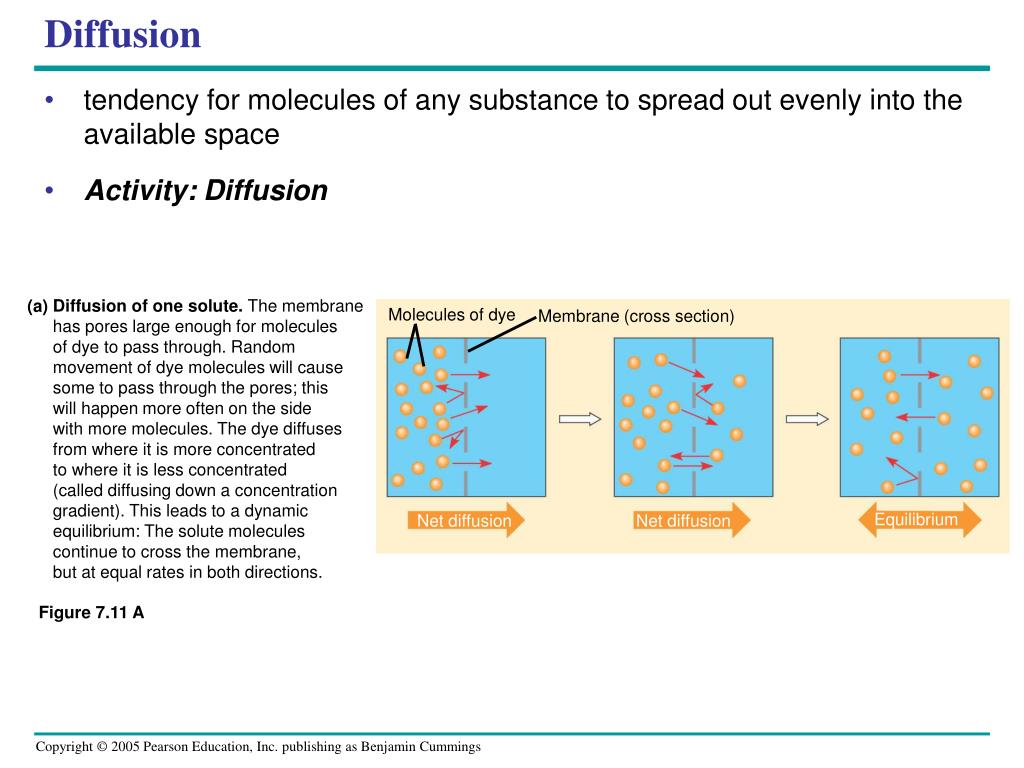
The concentration gradient is essentially the difference in the amount of a substance (like molecules) between two areas. Think of it as a "crowdedness" difference.
These particles, whether they are tiny gas molecules or larger dissolved substances, are always in motion. They jiggle and bounce around randomly. Now, picture those particles in an area where there are a lot of them (high concentration). Because they're all bumping into each other and moving randomly, they're naturally going to spread out into areas where there are fewer of them (low concentration). It's like a bunch of excited kids in a small room – they'll naturally want to explore the bigger, emptier hallway!
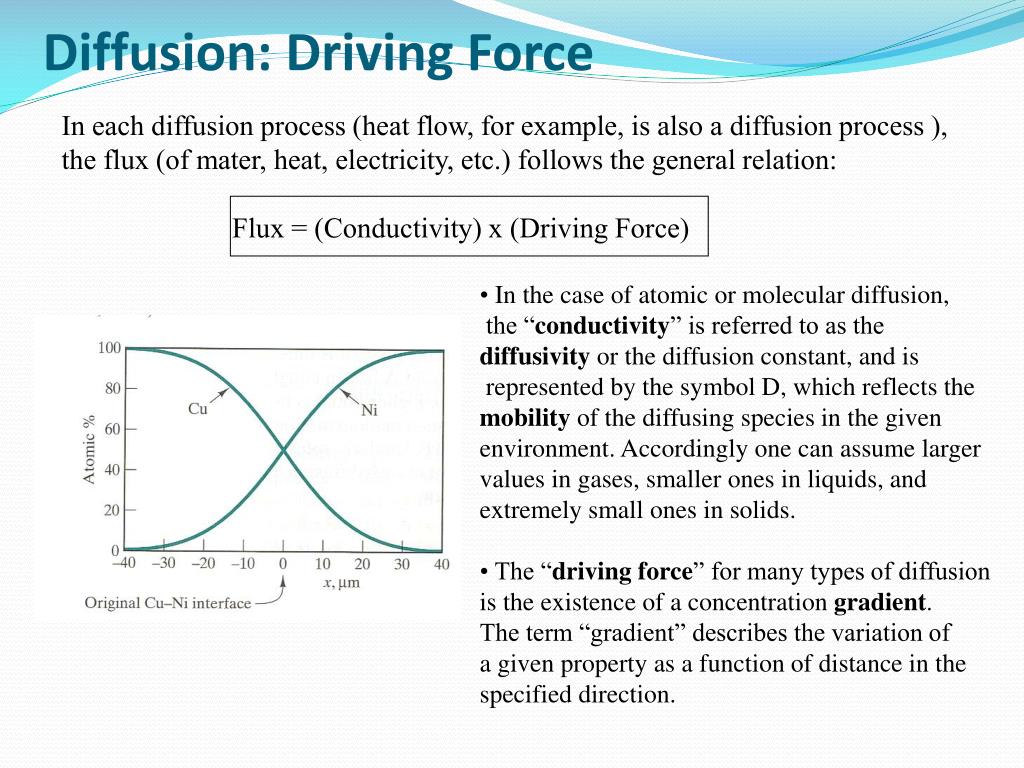
This tendency for particles to move from an area of high concentration to an area of low concentration, driven by the concentration gradient, is the principal force behind diffusion. It's an inherent property of matter, a passive process that doesn't require any extra energy input. The "gradient" provides the push. The greater the difference in concentration between two areas, the steeper the gradient, and the faster the diffusion will happen. So, if you spray a lot of perfume in a small, enclosed space, the concentration gradient is huge, and the scent will spread rapidly!
Everyday Examples of the Gradient in Action
Let's look at some more relatable scenarios where the concentration gradient is the unsung hero:
- Sugar in your coffee: When you add sugar to your hot coffee, the sugar molecules are highly concentrated where you dropped them in. The rest of the coffee has very little sugar. The concentration gradient drives the sugar molecules to spread out from the clump at the bottom until the entire cup of coffee tastes sweet.
- Breathing: Inside your lungs, the air you breathe has a high concentration of oxygen. Your blood, on the other hand, has a lower concentration of oxygen. This difference, this gradient, causes oxygen to move from your lungs into your bloodstream. The opposite happens with carbon dioxide – it moves from your blood (where its concentration is higher) into your lungs (where its concentration is lower) to be exhaled.
- Food coloring: If you put a drop of food coloring into a glass of water, the dye molecules are initially clustered together. The water has no dye. The concentration gradient pushes the dye molecules outwards until they evenly color the entire glass.
- Smell of baking cookies: When cookies are baking, delicious fragrant molecules are released into the air. Inside the kitchen, the concentration of these molecules is very high. As they move into other parts of the house where the concentration is lower, you start to smell those yummy cookies from afar!
It’s truly fascinating to realize that such a fundamental and widespread process is powered by this simple concept of particles moving from where there are many to where there are few, all thanks to the invisible hand of the concentration gradient. It's the gentle, persistent nudge that keeps the molecular world in motion and, by extension, keeps our own world functioning!
