The Polar End Of The Phospholipid Plasma Membrane Is _____.
Hey there, fellow humans navigating the glorious chaos of modern life! Ever stopped to wonder what’s really going on inside you, at that super-tiny, fundamental level? No, not like, “Did I leave the oven on?” tiny, but microscopic. We’re talking about the unsung heroes of your cells: the phospholipids. And specifically, we’re diving into the wonderfully weird world of the polar end of the phospholipid plasma membrane. Drumroll, please...
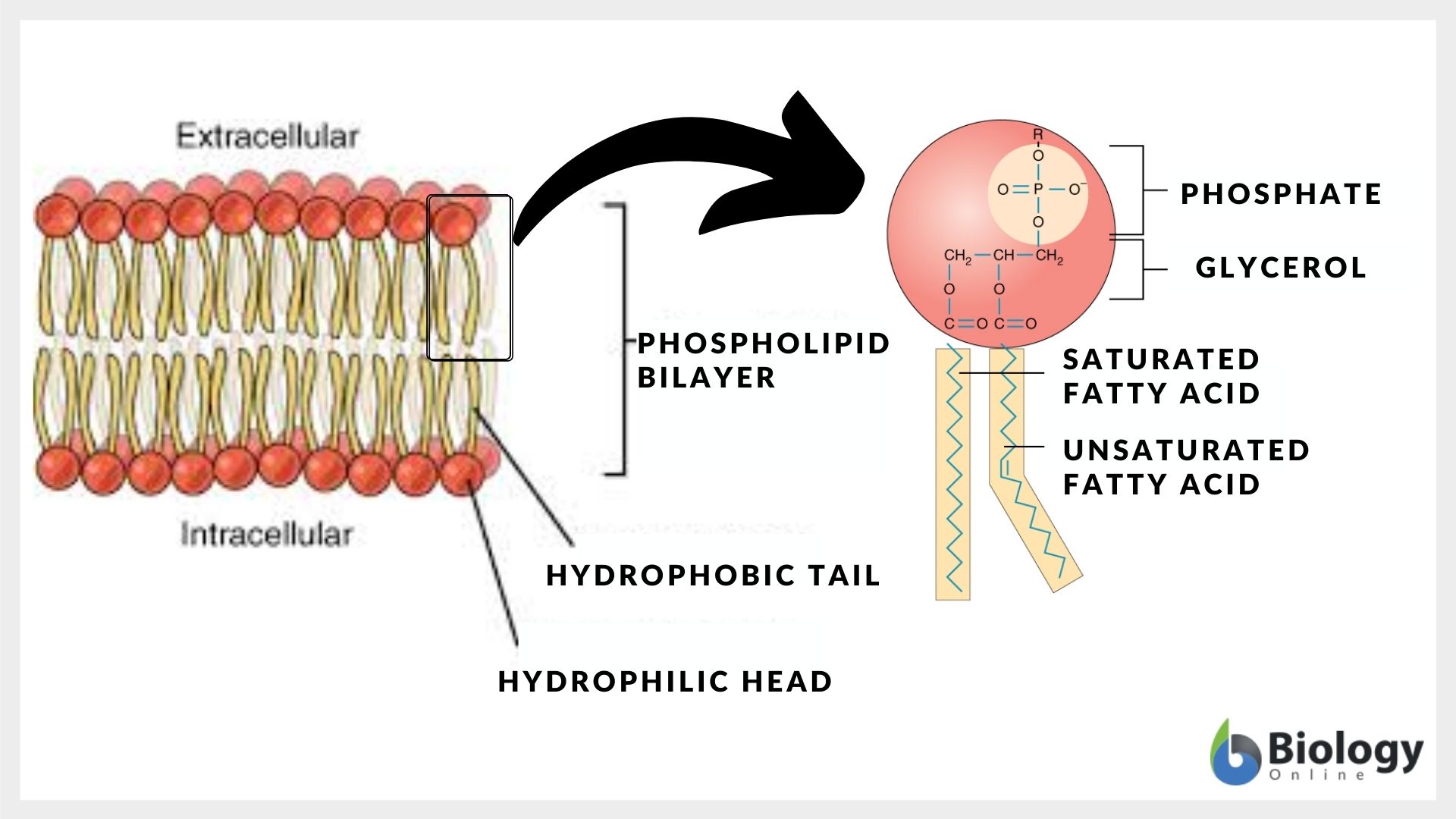
The polar end of the phospholipid plasma membrane is... the hydrophilic part! Yep, that’s the technical term, but let's unpack it, shall we? Think of it as the “loves water” bit of the phospholipid molecule. It’s the part that’s all about being friendly with H₂O. And since our bodies are basically walking water balloons, this is a pretty big deal.
More Than Just a Pretty Face (of a Molecule)
So, what exactly is this hydrophilic end? Imagine a phospholipid as a tiny, two-pronged party animal. It’s got two fatty acid tails – those are the hydrophobic, or “water-fearing,” guys. They’re the introverts of the molecule, always trying to hide from water. But then, there’s the head. And this head? It’s polar. This means it has an uneven distribution of electrical charge, making it attracted to water molecules. It’s like the gregarious, outgoing friend who’s always ready for a pool party.
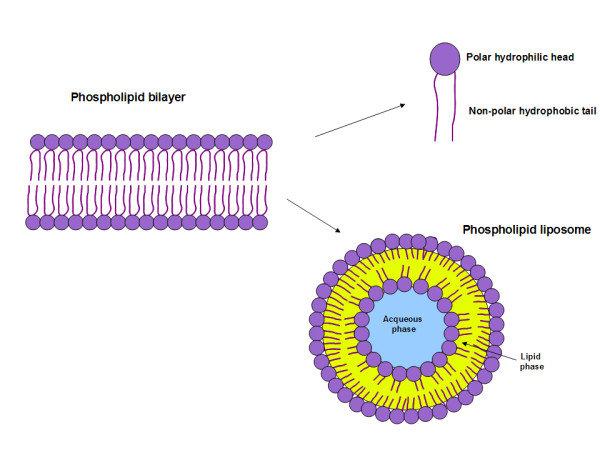
This polarity is crucial. It dictates how these phospholipids arrange themselves when they’re in an environment like your cell. They naturally form a bilayer, a double layer, where the hydrophilic heads all face outwards, towards the watery environment outside the cell and the watery cytoplasm inside. The hydrophobic tails, meanwhile, huddle together in the middle, away from all that water, creating a protective, oily core.
Why Should You Care About Water-Loving Heads?
This seemingly simple arrangement is the foundation of your cell’s boundary, the plasma membrane. It’s what separates your precious internal cellular machinery from the sometimes-harsh external world. It’s like the velvet rope at a super exclusive club, only instead of bouncers, it’s science!
This hydrophilic exterior isn’t just for show. It allows the cell to exist in the watery soup that is our internal environment. Think about it: if your cell membrane were entirely hydrophobic, it wouldn’t be able to interact with anything! It would be like a fish trying to survive in a desert. Not ideal.
Moreover, the hydrophilic nature of the membrane surface allows for the transport of water-soluble substances. It’s a gateway, but a very selective one. Certain proteins embedded within the membrane act as channels and transporters, helping specific molecules – like sugars, ions, and amino acids – to cross this barrier, guided by their interaction with the polar heads.

A Splash of Science Fun: The Amphipathic Nature
The term for a molecule that has both a hydrophilic and a hydrophobic part is amphipathic. Phospholipids are the rockstars of amphipathic molecules. This dual nature is their superpower. It’s what allows them to self-assemble into these vital structures. It's like having a Swiss Army knife of molecules!
Think of it like this: you’re at a beach party. The hydrophobic tails are the folks who brought their beach towels and found a shady spot under a palm tree, away from the direct sun. The hydrophilic heads are the ones splashing in the ocean, absolutely loving the cool water. They’re coexisting, making the whole beach experience work.
Cultural Cameos: Membrane Metaphors
We see this amphipathic principle in action all around us, even if we don’t always realize it. Consider soap! Soap molecules are amphipathic. They have a hydrophobic tail that can grab onto greasy dirt and a hydrophilic head that can dissolve in water. That’s why soap is so good at cleaning. It’s science in action, making your dishes sparkle and your hands feel squeaky clean.
Or think about salad dressing. Oil and vinegar don’t naturally mix, right? The oil is hydrophobic, and the vinegar (mostly water) is hydrophilic. But when you whisk them together, the emulsifier (often lecithin, derived from soybeans, which is a phospholipid!) helps them to temporarily combine, creating a homogenous dressing. The lecithin molecules surround the oil droplets with their hydrophilic heads facing out, allowing them to mingle with the vinegar. Science, art, and deliciousness, all in one bottle!
The Hydrophilic Head: What's Inside the 'Head'?
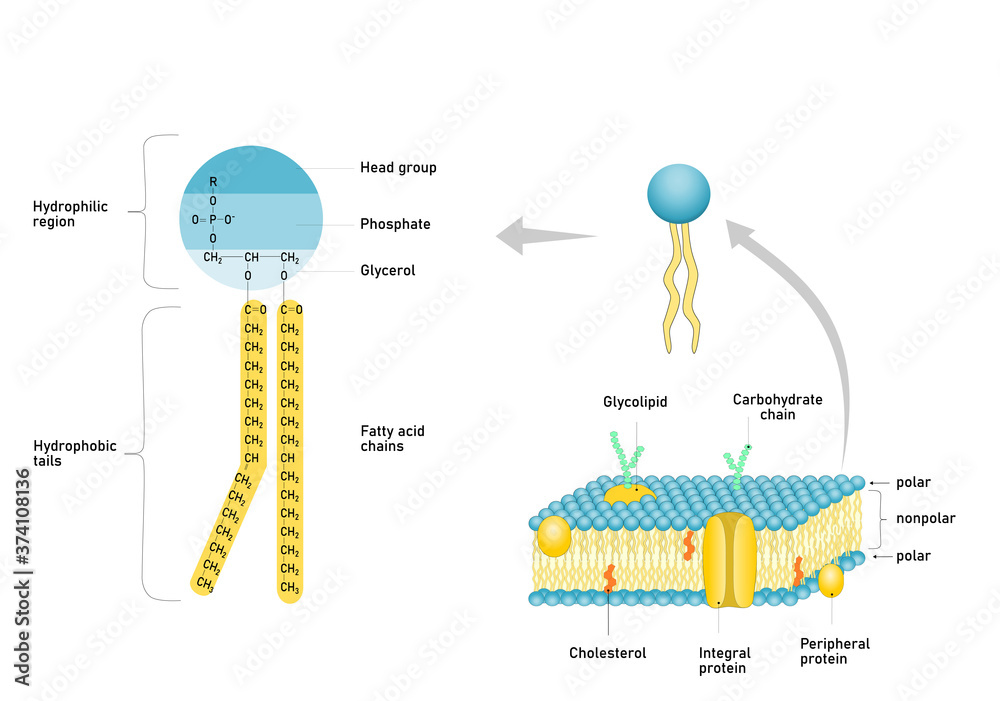
While we often simplify it to "the head," the actual composition of this hydrophilic part can vary and is fascinating in itself. It typically consists of a phosphate group linked to a small, organic molecule. This "head group" can be choline, serine, ethanolamine, or inositol, among others.
The specific head group influences the properties of the phospholipid and the overall function of the cell membrane. For instance, the charge on the head group can affect how the membrane interacts with other molecules and how easily it can be disrupted. It's like different people having different personalities – some are more bubbly, some are more reserved, but they all contribute to the social dynamic.
From Cell Walls to Your Skin
This fundamental structure, driven by the hydrophilic heads and hydrophobic tails, is responsible for everything from maintaining the shape of your cells to regulating what enters and leaves them. It’s the silent guardian of your internal homeostasis. It ensures that the delicate balance within your cells is maintained, allowing your organs to function, your muscles to move, and your brain to think.
And it’s not just about cells. The principles of phospholipid membranes are applied in fields like nanotechnology and drug delivery. Imagine tiny, artificial vesicles designed to carry medication directly to diseased cells, mimicking the natural way our own cells are structured and interact. It’s biomimicry at its finest, inspired by the humble phospholipid.
The Hydrophilic Hug: A Constant Connection
The hydrophilic nature of the polar end means that the outer surface of your cell membrane is constantly interacting with water. This is essential for cell signaling, nutrient uptake, and waste removal. It’s a constant exchange, a dynamic dance between the cell and its watery environment. It’s never static; it’s always in flux, always communicating.
Think about when you get a paper cut. The immediate response of your body is to send fluids to the area – that’s water, carrying all sorts of healing factors. This interaction with water is fundamental to life, and the hydrophilic heads of your phospholipids are the gatekeepers of this interaction.
Fun Fact: The Fluidity of It All!
Despite its structured appearance, the phospholipid bilayer is surprisingly fluid. The individual phospholipids can move laterally within their layer, kind of like dancers on a stage. This fluidity is crucial for many membrane functions, such as cell movement and division. It’s not a rigid wall, but a dynamic, ever-shifting dance floor.
The hydrophilic heads, being exposed to the water, contribute to this fluidity by allowing for relatively easy movement within the aqueous environment. It’s a beautiful balance between structure and movement, a testament to the elegance of biological design.
The Great Divide: Inside vs. Outside
The very existence of a hydrophilic and hydrophobic side creates an impermeable barrier for most water-soluble molecules. This is a critical survival mechanism. Without this barrier, your cells would simply dissolve in the surrounding fluid. It’s the ultimate “keep out” sign for anything that doesn’t have the right credentials to enter.
This selective permeability is managed by specialized proteins embedded within the membrane. These proteins act as bouncers, letting in what’s needed and keeping out what’s not. The hydrophilic nature of the membrane’s surface is what allows these protein gatekeepers to be embedded and function correctly, facilitating their interaction with both the internal and external aqueous environments.

When the Heads Get It Wrong (Rarely!)
While the hydrophilic nature is generally a good thing, in certain disease states, the way these molecules interact can go awry. For example, some genetic disorders can affect the lipids in cell membranes, altering their structure and function. This can lead to a cascade of problems within the cell and the organism. But for the most part, these hydrophilic heads are the silent heroes of our cellular health.
It’s a reminder of how intricate and precisely balanced our biological systems are. Every component, from the smallest phospholipid to the largest organ, plays a vital role. And when one part falters, the whole system can be affected. It’s a powerful lesson in interconnectedness.
A Daily Dose of Membrane Magic
So, the next time you’re sipping on a glass of water, or feeling that refreshing splash on your skin, take a moment to appreciate the unsung heroes. The hydrophilic ends of your phospholipid plasma membrane are working tirelessly, every second of every day, to keep you alive, to keep you functioning. They are the welcoming committee, the first responders, and the gatekeepers, all rolled into one.
It's a beautiful reminder that even in the most fundamental aspects of our existence, there's a complex and elegant design at play. This seemingly simple attraction to water is the bedrock of cellular life, allowing for the intricate dance of molecules that keeps us, well, us. Pretty neat, right?
Reflection: Thinking about the hydrophilic nature of our cell membranes makes me realize how fundamentally connected we are to water. We are, in essence, beings that thrive in a watery world, and our very structures are built to embrace and interact with it. It’s a gentle nudge to remember to stay hydrated, to appreciate the feel of water, and to recognize that this simple element is intrinsically linked to our existence, right down to the molecular level. It's a small piece of science that can inspire a mindful appreciation for our own bodies and the liquid lifeblood that sustains us.
