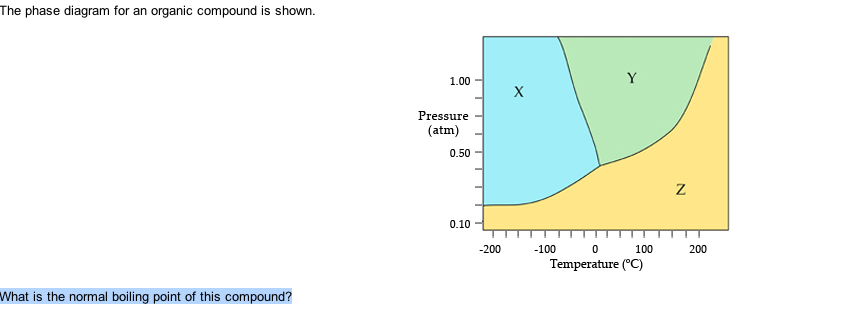
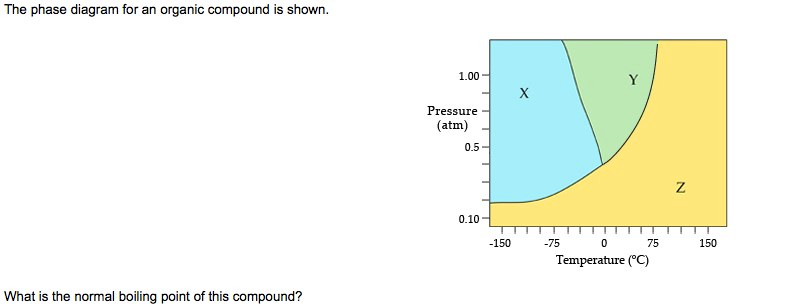
The Phase Diagram For An Organic Compound Is Shown
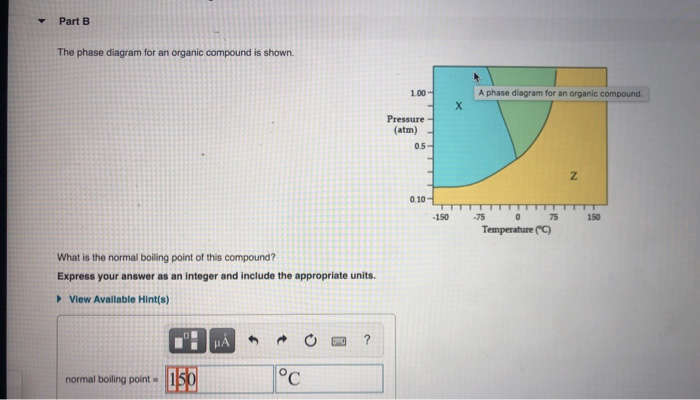
Ever looked at a complicated graph in a science textbook and thought, "Who on earth needs to know this?" Well, my friends, today we're going to dive into something that might look intimidating at first glance, but trust me, it's actually a super cool way to understand how everyday stuff behaves. We're talking about the phase diagram for an organic compound. Don't let the fancy name scare you! Think of it as a secret map to the hidden life of molecules.
So, what exactly is this mysterious phase diagram? Imagine you're trying to figure out the perfect conditions for, say, making homemade caramel. You need to know the right temperature and pressure, right? A phase diagram is kind of like that, but for a single substance – like a sugar molecule, or even water (though we're focusing on organic ones today!). It tells you, at any given temperature and pressure, what state that substance is going to be in. Is it a solid? A liquid? A gas? Or maybe something even more exciting?
Think of it as a personality test for molecules! Each molecule has its own unique phase diagram, its own set of rules for when it wants to be stiff and solid, when it feels like flowing freely as a liquid, and when it gets so energetic it zips around as a gas. Isn't that neat? It’s like understanding their moods!
Let’s break it down. On this diagram, you’ll usually see two main axes. One is temperature – that’s pretty straightforward, hotter or colder. The other is pressure – how much squishing is going on. These two factors are the ultimate conductors of the molecular orchestra, dictating whether our organic compound will be chilling out in a crystal lattice, dancing around as a liquid, or doing the cha-cha as a gas.
Now, the really fun part begins when you look at the lines on the diagram. These aren't just random scribbles, oh no. These lines represent the magic moments where two phases can coexist. They’re the tipping points, the boundaries. For instance, there’s a line where solid meets liquid. This is your melting point or freezing point. Below this line, it's solid; above, it's liquid. Simple, yet profound!
Then you have the line where liquid meets gas. This is your boiling point or condensation point. Cross that line, and your liquid decides it's ready for a more spacious existence. And sometimes, you’ll even see a line where solid goes directly to gas – that’s called sublimation. Think of dry ice disappearing into thin air! It's like a magic trick performed by physics.
But here’s where it gets really interesting. The lines on a phase diagram don't just go on forever. They often meet at a special point. This is called a triple point. And what’s so special about the triple point? It’s the exact temperature and pressure where all three phases – solid, liquid, and gas – can exist together, in perfect harmony! Imagine a tiny, magical microcosm where ice, water, and steam are all having a party at the same time. How cool is that? It’s like finding a secret hideout where all the different sides of a personality can get along.
Why should you care about this, you ask? Well, understanding phase diagrams is like having a superpower for understanding the world around you. Think about cooking. When you boil water for pasta, you're working with its phase diagram. When you freeze ice cream, you're dealing with phase transitions. Even something as simple as how a candle burns involves changes in the phase of the wax.
For organic compounds, this is even more crucial. These are the building blocks of life, the stuff that makes up plastics, medicines, fuels, and even the flavors in your favorite foods. Knowing their phase diagrams helps scientists design better materials, develop more efficient chemical processes, and even create new drugs that work exactly as intended. It’s not just abstract science; it's the blueprint for innovation!
Imagine a chemist trying to synthesize a new drug. They need to know at what temperatures and pressures their compound will be stable, easy to handle, and pure. The phase diagram is their guide, their roadmap to success. It tells them when to heat it up gently, when to apply pressure, and when to just let it be. It's like having a wise advisor whispering the best way to handle delicate things.
And it's not just for the lab coats! Thinking about phase diagrams can make you more observant of the world. The next time you see dew forming on the grass, or fog rolling in, you're witnessing the beautiful dance of phase transitions. You're seeing molecules making up their minds, shifting from one state to another based on temperature and pressure. It’s a constant, fascinating show happening all around us.
Learning about phase diagrams might seem like a deep dive into the technicalities of chemistry, but it's really about appreciating the subtle, yet powerful, forces that govern matter. It’s about seeing the hidden order and logic in what might appear chaotic. It's about realizing that even the most complex substances have predictable behaviors if you just know how to read their map.
So, the next time you encounter a phase diagram, don't groan. Smile! You're looking at a condensed story of how a substance lives and breathes, how it transforms and adapts. You're peering into the very heart of molecular behavior, and that, my friends, is nothing short of inspiring. It’s a reminder that the world is full of elegant principles waiting to be discovered, and with a little curiosity, you can unlock some truly fascinating insights. Keep exploring, keep wondering, and who knows what amazing connections you’ll make!
