The Orbital Diagram For A Ground-state Nitrogen Atom Is

Alright, settle in, grab your latte, and let's talk about something that sounds way more intimidating than it actually is: the orbital diagram for a ground-state nitrogen atom. Don't let the fancy name scare you! Think of it as the ultimate cosmic bachelor pad for our little nitrogen buddy, and we're about to get a sneak peek inside.
So, nitrogen. You know, the stuff that makes up, like, 78% of the air we breathe? It's basically the invisible bodyguard of our atmosphere, keeping things from getting too… well, o-too exciting. This nitrogen atom we're dissecting today is in its ground state, which basically means it's chilled out, totally relaxed, not doing any wild electron gymnastics. It's like that friend who’s perfectly happy just lounging on the couch, no big plans, just vibing.
Nitrogen's Atomic Crib Sheet
First things first, our nitrogen friend has seven electrons. That's its atomic number, like its social security number for the universe. These seven electrons are the residents of our orbital diagram, and they're looking for the comfiest, most energy-efficient places to hang out. They're not just randomly scattered, oh no. They follow some pretty strict real estate rules.
Think of orbitals as tiny, invisible rooms in the atom's house. These rooms have different shapes and energy levels. The lower energy rooms are closer to the nucleus (the atom's super-dense, incredibly important living room), and the higher energy rooms are a bit further out.
The Ground Floor: The 1s Orbital
Our first stop is the 1s orbital. Imagine this as the cozy studio apartment right next to the elevator. It’s got everything you need, and it’s super low energy because it’s so close to the action (the nucleus, remember?).
This 1s orbital is a sphere. Yes, a perfect, humble sphere. And it can only hold a maximum of two electrons. Why two? Because electrons are a bit like introverts at a party – they can tolerate one other person in their space, but that's about it. They also have this cool property called spin, which is like a tiny internal compass. One electron spins up, the other spins down. They have to be opposite spins to coexist in the same orbital. It’s like they’re holding hands and spinning in opposite directions to keep things balanced. Super cute, right?

So, for nitrogen, the first two electrons are going to plop right into this 1s orbital. They’re the pioneers, the first ones to claim their spot. 1s², we write that down, meaning two electrons in the 1s orbital. They’re all snug and happy.
The Second Floor: The 2s Orbital
Next up, we have the 2s orbital. This is like the slightly larger, still quite comfortable one-bedroom apartment on the next floor up. It's also a sphere, but it’s a bit bigger and has a slightly higher energy level. It’s not as close to the nucleus, so it’s a tad less desirable, but still a solid choice.
Just like the 1s orbital, the 2s can also hold a maximum of two electrons. So, our next two electrons, the third and fourth ones, are going to head over here. Again, they’ll have opposite spins. 2s². Now we've accounted for four of our seven electrons. They’re all settled in their comfy spherical digs.

The Penthouse Suites: The 2p Orbitals
Now, things get a little more interesting. We’ve got three electrons left, and they’re eyeing the 2p orbitals. These aren’t spheres anymore, oh no. These are like fancy, dumbbell-shaped rooms. And here’s the kicker: there are three separate 2p orbitals!
These three 2p orbitals are all at the same energy level. Think of them as identical penthouses, all with amazing views, but each facing a slightly different direction. We call them the 2px, 2py, and 2pz orbitals. Imagine them like three perfectly placed sculptures in a modern art gallery, each occupying its own distinct space but offering a similar experience.
Now, here’s a crucial rule, a bit like the unspoken etiquette of sharing communal spaces: Hund's Rule. This rule says that electrons will spread out into empty orbitals at the same energy level before they start pairing up. They're like, "Why share a room when I can have my own little kingdom?" It’s all about minimizing electron repulsion and maximizing personal space. They're not anti-social, they just prefer a little elbow room.
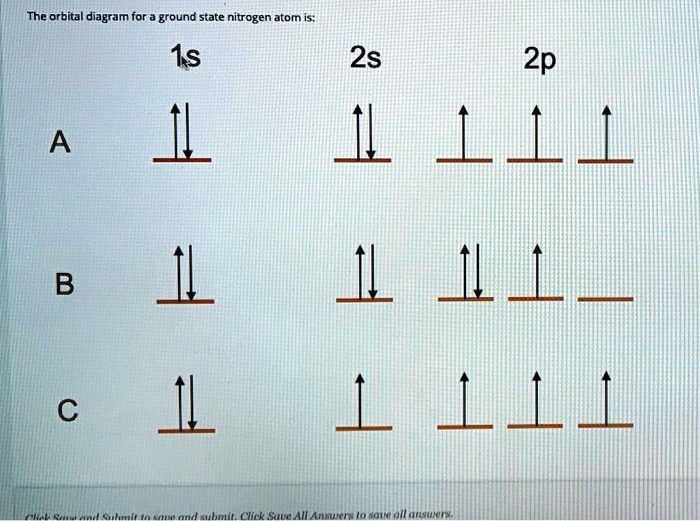
So, our remaining three electrons are going to do the following: The fifth electron goes into the 2px orbital. The sixth electron goes into the 2py orbital. And the seventh electron goes into the 2pz orbital. Each of them gets their own little dumbbell-shaped haven, and they all spin in the same direction. They’re like a synchronized swimming team, but with tiny, charged particles.
We represent this as 2p³, but it’s crucial to remember that these three electrons are unpaired and occupying separate orbitals.
Putting It All Together: The Nitrogen Orbital Diagram
So, let’s visualize this cosmic apartment complex for ground-state nitrogen:
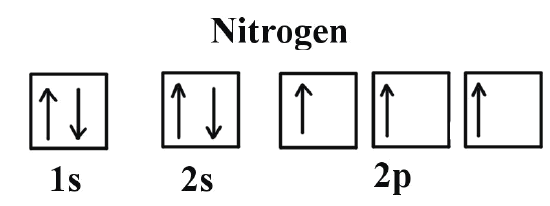
- 1s orbital: One spherical room, with two electrons inside, spinning in opposite directions (one up ↑, one down ↓).
- 2s orbital: Another spherical room, one floor up, also with two electrons, spinning in opposite directions (one up ↑, one down ↓).
- 2p orbitals: Three dumbbell-shaped rooms, on the top floor, each with a single electron inside, all spinning in the same direction (up ↑, up ↑, up ↑).
This arrangement is the most stable and lowest energy configuration for a nitrogen atom. It’s like perfectly stacked building blocks, everything in its right place, no unnecessary clutter.
Why Does This Even Matter?
You might be thinking, "Okay, that’s neat, but why should I care about where electrons are chilling?" Well, my friends, this seemingly simple diagram is the blueprint for how atoms interact. Nitrogen’s electron arrangement dictates its reactivity. Because those 2p electrons are a bit more exposed and unpaired, they’re the ones that get involved in forming chemical bonds. They’re the party starters!
Nitrogen, with its three unpaired electrons in the 2p orbitals, is itching to share or take electrons to achieve a more stable, filled outer shell. This is why nitrogen is so essential in forming molecules like ammonia (NH₃) and, believe it or not, the very DNA that makes you, you! It’s all thanks to the humble, yet incredibly important, orbital diagram of a ground-state nitrogen atom.
So, the next time you take a deep breath, spare a thought for our nitrogen friends, diligently occupying their orbital apartments, keeping the air breathable and the universe, well, chemically interesting. And remember, even the most fundamental parts of science can be a little bit like a story about cosmic roommates. Cheers!
