The Number Of Pi Bonds In The Molecule Below Is

Hey there, science curious folks! Ever find yourself staring at a molecule, maybe one you sketched out in a chemistry class or saw in a cool infographic, and wonder, "What's really going on in there?" Today, we're going to dive into a tiny, but super important, aspect of molecular structure: pi bonds. And we'll figure out how many of these awesome things are hiding in a particular molecule. Don't worry, we're keeping it super chill, no complicated jargon overload here!
So, what even is a pi bond? Think of it like this: when atoms decide to share electrons to become friends (that's what a covalent bond is, by the way), they don't always just do it in a straightforward, one-on-one handshake. Sometimes, they get a little more creative, and that's where different types of bonds come into play. We've got your standard sigma bonds, which are like a direct, head-on hug between atoms. They're the foundation, the strong, stable connection that holds most of the molecule together.
But then, you have these special, more intimate connections called pi bonds. Imagine those sigma bonds are the main roads connecting cities. Pi bonds? They're more like the scenic side roads, the ones that twist and turn and offer a different kind of view. They form from the sideways overlap of atomic orbitals, kind of like two balloons brushing past each other instead of bumping head-on. This sideways overlap means pi bonds are generally weaker than sigma bonds, and they often appear in pairs or triples when atoms decide to really get cozy.
You’ll typically find pi bonds in what we call double bonds and triple bonds. A single bond is just one sigma bond. But a double bond? That’s where the magic happens. A double bond is made up of one sigma bond and one pi bond. It's like a stronger, more committed handshake. And a triple bond? That’s even more intense! It’s comprised of one sigma bond and two pi bonds. That’s a seriously strong connection, like superglue for atoms!
So, why should we care about these pi bonds? Well, they’re a big deal! They're often responsible for a molecule's reactivity. Think of them as the spots where chemical reactions are most likely to occur, like a particularly interesting intersection where many roads meet. They can also influence the shape and flexibility of a molecule. Molecules with lots of pi bonds can sometimes be a bit more rigid, while those with only single bonds can twist and turn more freely. It's all about how the electrons are distributed!
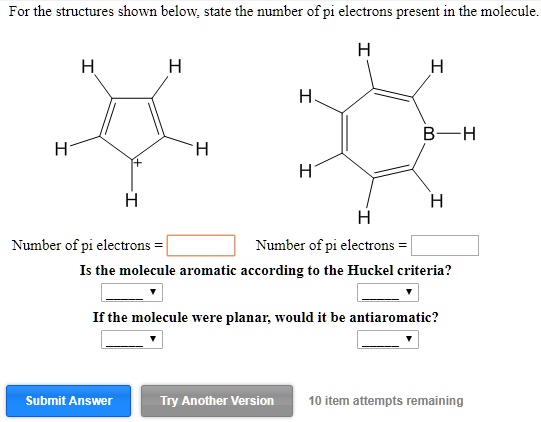
Now, the big question: how do we find the number of pi bonds in a specific molecule? This is where it gets fun. You usually need to know the molecule's structure. Sometimes, you're given a chemical formula (like C2H4), but that doesn't tell you the arrangement of atoms. For that, you might need a structural diagram or to draw it yourself using some basic chemistry rules. But let's imagine we've got our molecule right in front of us, or at least its picture!
Let's take a super common example. What about something like ethene (also known as ethylene)? Its formula is C2H4. If you draw it out, you'll see two carbon atoms are connected to each other with a double bond, and each carbon is also connected to two hydrogen atoms with single bonds. So, we have that double bond between the carbons. Remember what we said about double bonds? One sigma bond and one pi bond. And all the carbon-hydrogen bonds? Those are just single bonds, so they're all sigma bonds. Therefore, ethene has exactly one pi bond.
How about something a bit more complex, like ethyne (acetylene)? Its formula is C2H2. When you draw ethyne, you'll see the two carbon atoms are joined by a triple bond, and each carbon is also bonded to one hydrogen atom with a single bond. Now, a triple bond is where we get more pi bonds! A triple bond is made up of one sigma bond and two pi bonds. So, in ethyne, we have those two pi bonds between the carbons. The carbon-hydrogen bonds are single bonds, so they're sigma bonds. This means ethyne has two pi bonds.
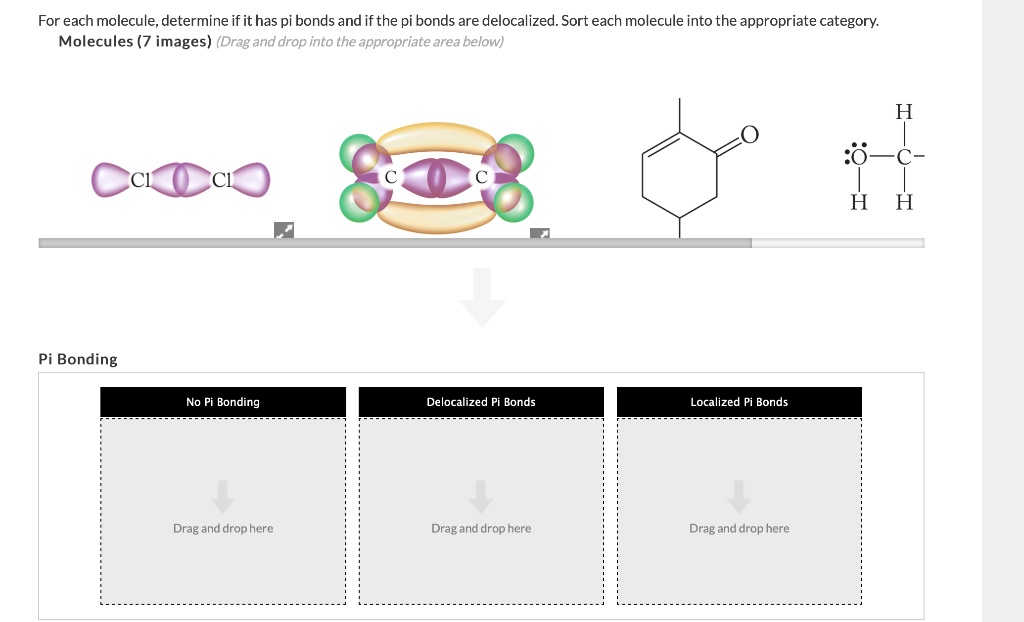
Okay, so let’s try to visualize a molecule that might be presented to you. Imagine a molecule that looks like this: a central carbon atom, and it's double-bonded to one oxygen atom, and then it's single-bonded to two hydrogen atoms. This would be formaldehyde. The double bond between the carbon and the oxygen? That's one sigma and one pi bond. The single bonds between the carbon and the hydrogens? Those are sigma bonds. So, formaldehyde has just one pi bond.
What if we had a molecule with a benzene ring? Benzene is a really famous and important molecule, with the formula C6H6. It's a ring of six carbon atoms, and it has a special structure called "resonance," which means the electrons in its double bonds are delocalized, spread out. But if we think about the simplified structure, it has alternating single and double bonds around the ring. Each double bond contributes one pi bond. Since there are three double bonds in the basic representation of benzene, you could say it has three pi bonds. These delocalized pi electrons are a huge reason why benzene is so stable and has its unique properties!

The trick, really, is to look at the types of bonds connecting the atoms. * Single bonds = 1 sigma bond (0 pi bonds) * Double bonds = 1 sigma bond + 1 pi bond (so, 1 pi bond) * Triple bonds = 1 sigma bond + 2 pi bonds (so, 2 pi bonds)
So, if you're given a molecule and asked to count its pi bonds, the first thing you'd do is look for any double or triple bonds. For every double bond you see, add one pi bond to your count. For every triple bond, add two pi bonds. Ignore all the single bonds; they don't have any pi bonds in them. It’s like a little treasure hunt for those sideways overlaps!
Let's imagine a hypothetical molecule now, just to make sure we've got it. Picture a chain of four carbon atoms. The first carbon is single bonded to the second. The second carbon is double bonded to the third. And the third carbon is triple bonded to the fourth. The other bonds are to hydrogens, but we’re just focusing on the carbon backbone for this pi bond count. * Carbon 1 to Carbon 2: Single bond (0 pi bonds) * Carbon 2 to Carbon 3: Double bond (1 pi bond) * Carbon 3 to Carbon 4: Triple bond (2 pi bonds)
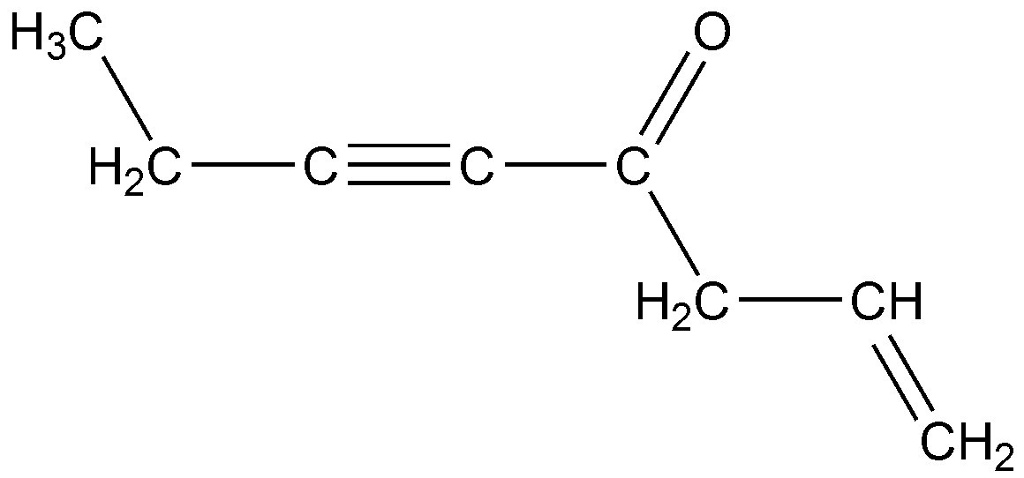
Add them up: 0 + 1 + 2 = 3 pi bonds in this imaginary carbon chain. Pretty neat, right?
Understanding pi bonds helps us understand so much about the world around us. They're crucial in things like the colors we see (many colored molecules have extensive systems of pi bonds), the way plastics are formed, and even how our DNA is structured (though DNA is more complex, the principles of bonding are still at play!). It's these subtle differences in how atoms connect that give molecules their unique personalities and functions.
So, next time you see a molecule, whether it's in a textbook, a magazine, or even a cool animated movie, take a moment to look for those double and triple bonds. They’re the tell-tale signs of pi bonds, the energetic, sideways hugs that add so much character and reactivity to the molecular world. It's a simple concept, but it unlocks a deeper appreciation for the intricate beauty of chemistry. Keep exploring, and happy molecule counting!
