The Number Of Electrons In A Sulfide Ion S2 Is

Get ready to dive into the zany, electrifying world of atoms! We're about to uncover a little secret about a super-cool character in the atomic party scene. It's like discovering your best friend has a hidden talent for juggling flaming chainsaws – totally unexpected and incredibly awesome!
Today, we're shining a spotlight on our star player: the sulfide ion. Now, "ion" might sound a bit sci-fi, like something out of a blockbuster movie, but it's actually just a fancy word for an atom that's decided to play a little game of "give and take" with its electrons. Think of it as an atom that's either feeling super generous or a bit… well, needy!
Our particular friend, the sulfide ion, has a special designation: S2-. That little "2-" sign is the secret handshake, the wink and a nod that tells us this atom isn't playing by the usual rules. It's like seeing a cat wearing a tiny superhero cape – it just adds a certain oomph!
The Humble Sulfur Atom
Before we get to the main event, let's meet the parent, the foundation: the neutral sulfur atom. Imagine a perfectly balanced seesaw. That's our sulfur atom in its natural, chill state.
In this balanced state, a sulfur atom has a specific number of tiny, zippy things called electrons. These electrons are like little energetic sprites, buzzing around the center of the atom, which we call the nucleus. They're the life of the atomic party, always on the move!
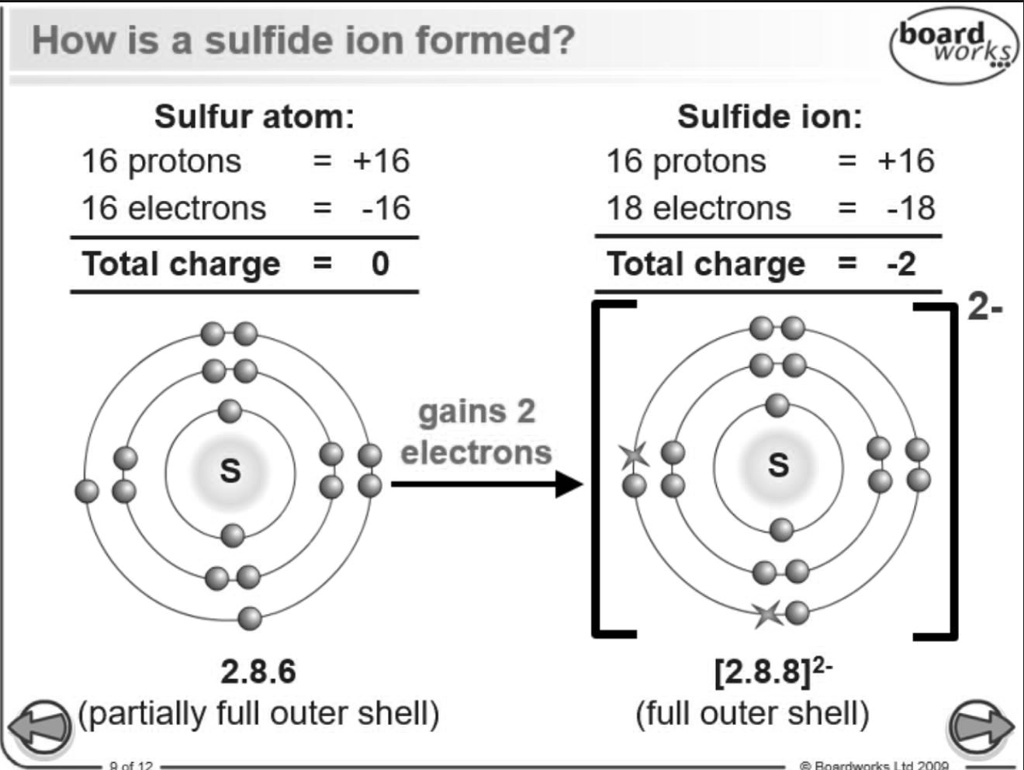
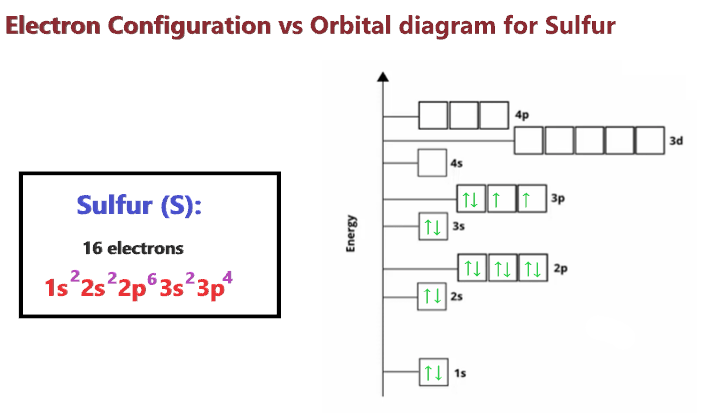
So, how many electrons are we talking about for a plain ol' sulfur atom? Drumroll, please… it's a grand total of 16 electrons! That's right, 16 tiny dancers doing the atomic cha-cha around the nucleus.
When Sulfur Gets a Little Extra
Now, here's where the fun really begins. The sulfide ion, the one with the S2- badge, isn't a plain ol' sulfur atom anymore. It's had a bit of a glow-up, a transformation that makes it… well, more interesting!
Remember that "give and take" game we mentioned? The sulfide ion has definitely been on the "take" side of things. It's like finding an extra cookie in your lunchbox when you thought you were all out – pure delight!
That little "2-" in S2- is our clue. It tells us that this sulfur atom has decided to acquire, to scoop up, not one, but two extra electrons. It's like the atom saw two lonely electrons floating around and thought, "Hey, come on over! We've got room for you here!"
The Grand Total Revealed!
So, let's do some super-simple, incredibly exciting math. We started with our neutral sulfur atom, happily rocking its 16 electrons. Then, our star player, the sulfide ion, decided it wanted a bit more zest, a bit more zing!
It welcomed those two extra electrons with open arms. Imagine adding two bouncy balls to a basket that already has 16 balls – it just makes the basket feel a little fuller, a little more exciting!
Therefore, the number of electrons in a sulfide ion (S2-) is its original 16 electrons plus those 2 extra electrons. That brings us to a magnificent, a colossal, a positively electrifying total of… 18 electrons!
Yes, you heard it here first! The sulfide ion is an electron-rich superstar, boasting a whopping 18 electrons. It’s like a party where everyone brought a friend, and suddenly, the dance floor is twice as full and twice as fun!

Think of it this way: a regular sulfur atom is like a quiet library, perfectly organized. But a sulfide ion? That's the same library, but now there's a surprise concert happening, with 18 musicians all playing their hearts out! It’s a symphony of electrons!
This extra electron baggage is what gives the sulfide ion its unique personality and its chemical "superpowers." It's these extra electrons that allow it to play so nicely (or sometimes, not so nicely!) with other atoms in the grand cosmic dance of chemistry.
So, next time you hear about a sulfide ion, remember its secret! It's not just sulfur; it's sulfur with an upgrade, a party animal that's brought two extra guests to the electron bash, making its electron count a grand total of 18. It’s a simple number, but oh, the stories it tells!

It's amazing to think that something so small can have such a dynamic life. The journey from a neutral atom to a charged ion is like a tiny atom's personal adventure story. And our sulfide ion’s story involves a little bit of electron hoarding, making it a truly fascinating character.
So, let's raise a metaphorical glass to the sulfide ion and its impressive count of 18 electrons! It's a reminder that even the most fundamental building blocks of our universe have their own quirks and their own exciting stories to tell. Isn't science just the most wonderfully energetic and entertaining thing ever?
"The number of electrons in a sulfide ion S2- is 18. It’s not just a number; it's a testament to its electron-collecting prowess!"
We've cracked the code, folks! The mystery is solved. That little S2- is a beacon of electron abundance, a true electron enthusiast. It’s a fantastic example of how atoms can change and adapt, becoming entirely new entities with their own special properties.
So, the next time you encounter the term sulfide ion, you'll know its electron secret. It's a supercharged, electron-packed atom ready for whatever chemical adventures come its way. Keep exploring, keep wondering, and keep enjoying the electrifying world of atoms!
