The Nitride Ion In Lithium Nitride Is Composed Of

Ever heard of Lithium Nitride? It sounds a bit fancy, maybe something you’d find in a science lab with bubbling beakers and serious-looking scientists. But trust me, this stuff is way cooler than it sounds. It’s like a secret ingredient with a personality, and at its heart is something truly special: the nitride ion.
Now, when you think about common things, you probably think about things like water (H₂O) or salt (NaCl). We’re all pretty familiar with those. But Lithium Nitride is a bit more of a rockstar. It’s an ionic compound, which means it’s made of two types of charged particles that are best buddies, sticking together like magnets. You’ve got your lithium ions, which are small and zippy, and then you’ve got our star of the show: the nitride ion.
So, what’s so amazing about this particular nitride ion in Lithium Nitride? Well, it’s not just any old nitrogen. Usually, when we think of nitrogen, we think of the air we breathe. That’s about 78% nitrogen gas (N₂). In that form, the nitrogen atoms are happily paired up, being pretty chill and stable. They’re like a perfectly matched couple, not looking for any drama.
But in Lithium Nitride, things get a bit more exciting. The nitride ion is actually nitrogen that’s decided to go solo and take on a negative charge. Think of it as nitrogen saying, “You know what? I’m going to be a bit more energetic today!” This extra charge makes it a really interesting character in the compound.
Imagine the lithium ions as tiny, positive little fireflies, and the nitride ion as a slightly larger, glowing orb that’s collected a lot of negative energy. They’re drawn to each other because opposites attract, right? This strong attraction is what holds Lithium Nitride together. It's a beautiful dance of positive and negative forces, all thanks to the nitride ion.

What makes the nitride ion in Lithium Nitride so entertaining is its readiness to react. Because it has that extra electron, it’s a bit like a magnet looking for things to grab onto. This makes Lithium Nitride a super useful material for all sorts of cool applications. It’s not just sitting around being pretty; it’s actively participating in chemistry!
One of the most fascinating things about Lithium Nitride is its potential for storing hydrogen. Yes, you read that right – storing hydrogen! Imagine a super-efficient way to hold onto this clean fuel. The nitride ion plays a crucial role here. It has this unique ability to interact with hydrogen in a way that’s really promising for things like fuel cell technology. It’s like the nitride ion has a special handshake with hydrogen atoms, making them stick around until we need them.
This makes Lithium Nitride a hot topic in the world of energy research. Scientists are super excited about it, and you can see why! It's not just a boring chemical compound; it’s a potential game-changer. The nitride ion is the unsung hero, the secret sauce that gives Lithium Nitride its superpowers.

Think about it: a material that can help us store clean energy more effectively. That's pretty darn inspiring! The nitride ion, with its negative charge and its ability to form strong bonds, is the key to unlocking these possibilities. It’s a tiny particle with a massive impact.
The way the nitride ion is structured, with its extra electron, is what makes it so reactive. It's always looking for a positive partner or a way to share its energy. In Lithium Nitride, it finds its perfect match with the lithium ion. Together, they form a stable but highly functional compound.
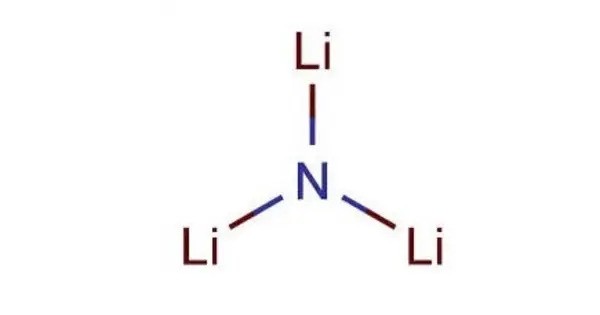
The nitride ion in Lithium Nitride is essentially a nitrogen atom that has gained an extra electron, giving it a negative charge. This seemingly small detail makes all the difference!
It’s like giving your favorite toy a little upgrade. Suddenly, it can do all sorts of new and amazing tricks. The nitride ion is the upgraded version of nitrogen, and it’s ready to perform. The strong electrostatic attraction between the positively charged lithium ions and the negatively charged nitride ions creates a crystal lattice structure that is both robust and full of potential.
So, next time you hear about Lithium Nitride, remember the star of the show: the nitride ion. It’s more than just a chemical component; it’s a tiny powerhouse of reactivity and potential. It’s what makes this compound so special and so exciting for the future of technology and energy. It’s a little bit of chemistry magic, all thanks to a charged-up nitrogen atom.
Isn’t that cool? A simple element, nitrogen, transformed into this energetic nitride ion, and then combined with lithium to create a material that could change the way we think about energy storage. It’s a testament to how even the smallest parts of the universe can have the biggest effects. The nitride ion is a perfect example of this, a tiny but mighty player in the world of materials science. So go ahead, get curious about Lithium Nitride and its fantastic nitride ion!
