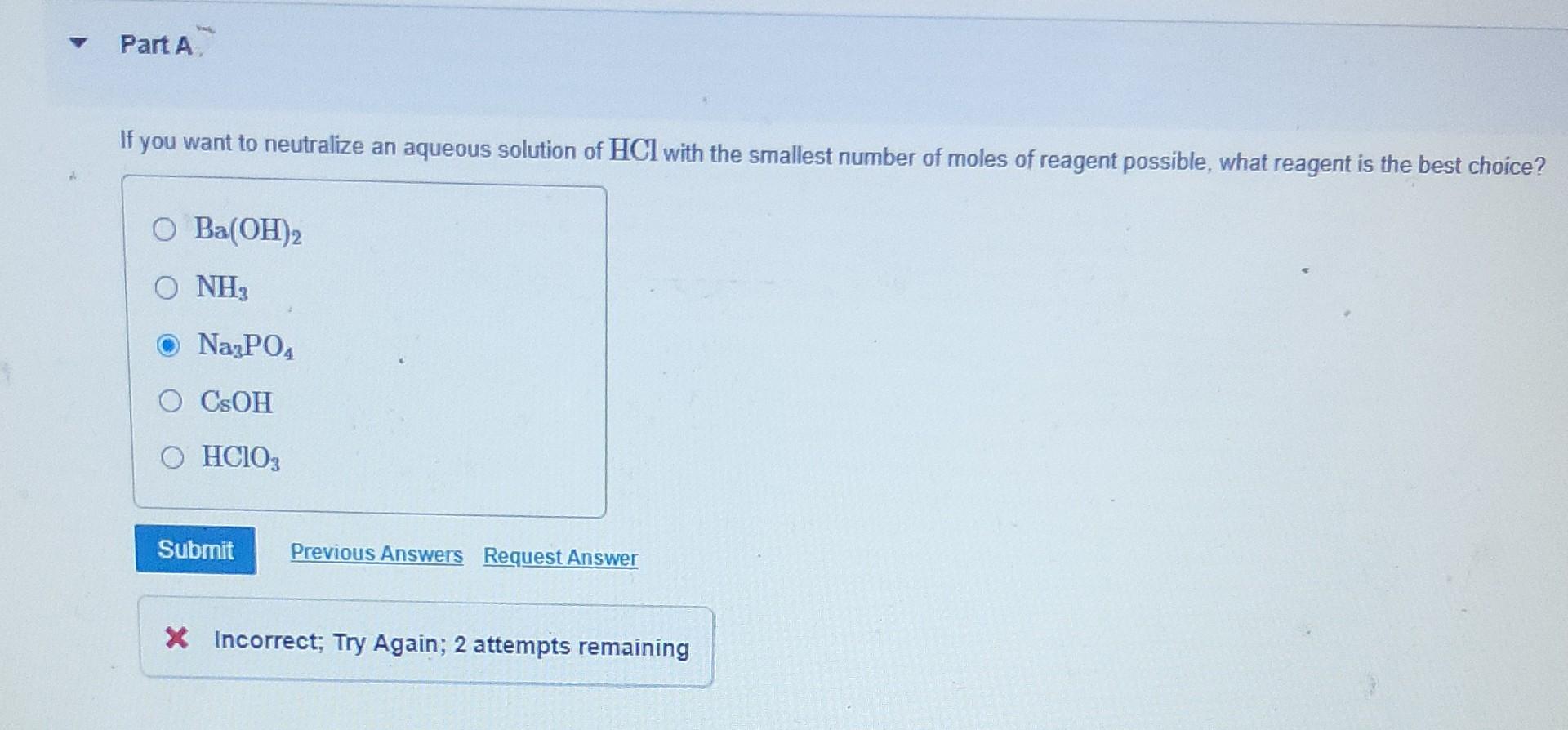
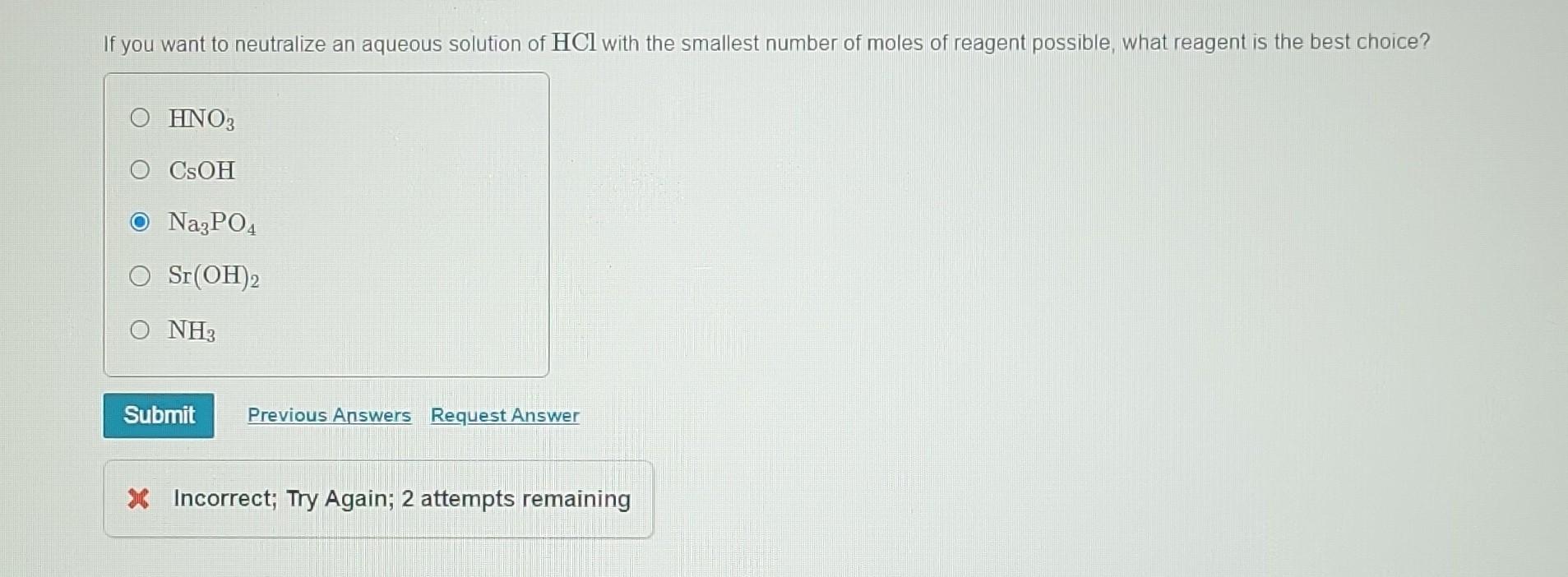
The Name Given To An Aqueous Solution Of Hclo3 Is

Ever wonder what exciting secrets lurk within seemingly simple chemical formulas? Today, we're diving into a name that might sound a little bit intimidating, but trust us, it's actually quite fascinating and surprisingly useful. It’s the name given to an aqueous solution of HClO3. While the formula itself might make you think of complicated laboratory experiments, the substance it represents plays a role in surprising places, from keeping things sparkling clean to aiding in the creation of other essential materials. Understanding this name isn't just about memorizing a chemical term; it's about unlocking a little piece of the world around us and appreciating the chemistry that makes it all work.
The Sparkle and Shine of a Familiar Friend
So, what is this mystery substance? When we dissolve HClO3, which is known as chlorous acid, in water, we get what's commonly called chlorous acid solution. While that might seem a bit redundant, it’s the most straightforward and accurate way to describe it. This solution, however, is more than just a combination of atoms. It's a versatile chemical with a range of applications that might surprise you. Think about the need for effective cleaning agents, powerful bleaching compounds, and even the creation of certain food preservatives. Chlorous acid solution quietly contributes to many of these areas, making our lives a little bit easier and a lot cleaner.
One of the primary benefits of chlorous acid solution lies in its oxidizing properties. In simple terms, this means it's really good at taking electrons from other substances. This characteristic makes it an excellent candidate for disinfection and sanitization. Imagine wanting to ensure that water is safe to drink or that surfaces in a hospital are free from harmful bacteria. Chlorous acid solution, often in controlled concentrations, can be a powerful tool in these crucial tasks. It’s not just about making things look clean; it's about making them safe.
Beyond its germ-fighting capabilities, chlorous acid solution also finds its way into the world of bleaching. Unlike some harsher bleaching agents, it can be quite effective at removing colors without necessarily damaging the material it's applied to, especially when used carefully. This makes it a valuable component in certain industrial processes and even in some household cleaning products. The controlled reactivity of chlorous acid solution is what makes it so useful in these varied scenarios.
It’s a chemical that, when handled with knowledge and care, contributes to a cleaner, safer, and more efficient world around us. The name chlorous acid solution might not roll off the tongue like "water" or "sugar," but its impact is undeniably present.
From Cleaning to Creating: The Broader Impact
The utility of chlorous acid solution doesn't stop at cleaning and disinfection. It also plays a role as an intermediate in the production of other important chemicals. For instance, it can be used to synthesize chlorites, which are salts that have their own set of applications. These chlorites can be used in various industrial processes, further extending the reach of this seemingly simple aqueous solution. It’s like a foundational brick in a larger chemical construction project, enabling the creation of even more complex and useful compounds.
Think about the food industry. While we often focus on ingredients we can eat, the processes that ensure food safety and preservation are equally vital. In certain controlled applications, compounds derived from chlorous acid can be used to help extend the shelf life of food products by inhibiting the growth of spoilage-causing microorganisms. This not only reduces waste but also helps ensure that the food we buy remains fresh and safe for consumption. It’s a behind-the-scenes hero, working to keep our food supply robust.
The beauty of chemistry often lies in its ability to perform multiple functions with elegant precision. Chlorous acid solution is a prime example of this. Its ability to act as a strong oxidizer makes it ideal for tackling a wide array of challenges, from battling microscopic invaders to aiding in the production of essential materials. Understanding what we call an aqueous solution of HClO3 – that is, chlorous acid solution – is to appreciate a chemical that contributes to our daily lives in more ways than we might initially realize. It's a reminder that even the most technical-sounding names can represent substances with practical and beneficial uses, making our world a little bit brighter, cleaner, and safer.
