The Molecular Weight Of Glucose Is 180 Daltons
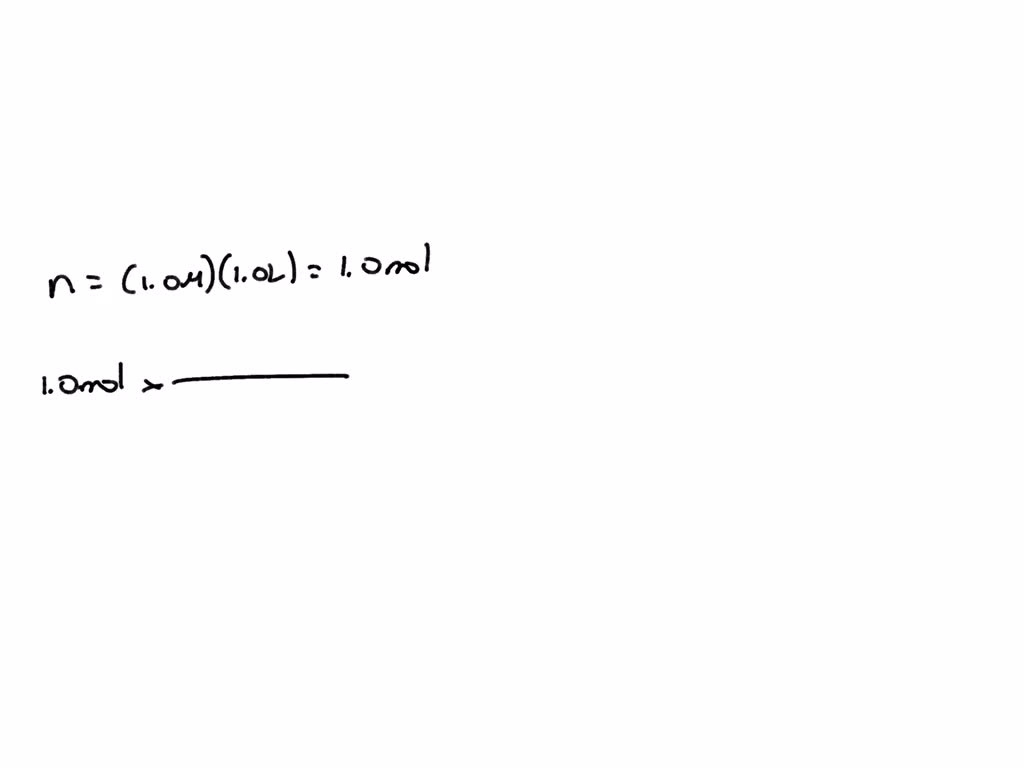
Hey there, sunshine! Ever find yourself staring at a perfectly baked cookie, a juicy piece of fruit, or even just your morning cup of coffee, and wonder what makes it all… work? You know, beyond the delicious taste and the energy boost? Well, today, we’re diving into a tiny little world, a world of molecules, to uncover a number that’s surprisingly relevant to your everyday joy: the molecular weight of glucose is 180 Daltons. Yep, we're talking about sugar, but make it science-lite and totally chill.
Now, before your eyes glaze over like a donut dipped in powdered sugar, let’s break down what “molecular weight” and “Daltons” actually mean. Think of it like this: every single substance, from the air you breathe to the water you drink, is made up of tiny building blocks called molecules. And just like you can weigh a bag of groceries, molecules have a weight too. The Dalton is just a unit of measurement for this molecular stuff, kind of like grams or pounds, but for the super-microscopic.
So, when we say the molecular weight of glucose is 180 Daltons, we're essentially saying that one mole of glucose molecules weighs approximately 180 grams. A mole, you ask? Don't sweat it! It's just a fancy scientific way of counting a huge number of molecules, specifically Avogadro's number (which is a number with 23 zeros after it – seriously, mind-boggling!). Think of it like a baker’s dozen, but for the universe.
Glucose: The Sweet Star of Our Show
Alright, so what's the big deal about glucose? It's the simplest form of sugar, and it's pretty much the go-to fuel for your body. Your cells, from your brain cells trying to remember where you left your keys to your leg muscles powering you through a spontaneous dance-off, run on glucose. It’s the primary energy currency of life, and that 180 Dalton number is its signature.
Ever heard of photosynthesis? That magical process where plants use sunlight to create their own food? Bingo! Glucose is the end product. It’s like the ultimate solar-powered snack for trees and flowers. They take carbon dioxide from the air and water from the ground, add a dash of sunshine, and voilà – they whip up glucose, a sugar molecule with that familiar 180 Dalton heft. It’s the foundation of so many food chains, which means that 180 is indirectly responsible for a lot of the deliciousness on your plate.
Think about your favorite fruits – berries, apples, grapes. They’re naturally sweet because they’re packed with glucose and other sugars. When you bite into a ripe strawberry, you’re tasting the results of plant energy production, all tied back to that elegant 180 Dalton structure. It’s pretty cool when you realize that a number you might never have encountered before is directly linked to something as simple and enjoyable as a sweet treat.
Breaking Down the 180: A Tiny Molecular Recipe
So, how do we get to 180? It's all in the ingredients, the atoms that make up the glucose molecule. Glucose has the chemical formula C6H12O6. This means each glucose molecule is made up of 6 carbon atoms (C), 12 hydrogen atoms (H), and 6 oxygen atoms (O).
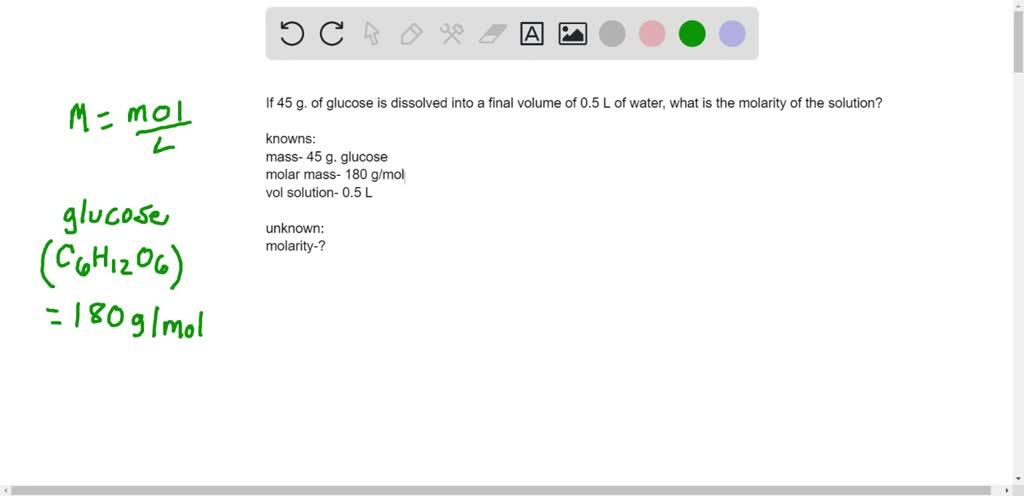
Now, each of these atoms has its own atomic weight, and we can find these values on the periodic table, which is basically a superhero lineup of all the elements. Carbon (C) weighs about 12 Daltons, hydrogen (H) weighs about 1 Dalton, and oxygen (O) weighs about 16 Daltons.
So, let’s do a little molecular math, but keep it breezy:
- 6 Carbon atoms * 12 Daltons/atom = 72 Daltons
- 12 Hydrogen atoms * 1 Dalton/atom = 12 Daltons
- 6 Oxygen atoms * 16 Daltons/atom = 96 Daltons
Add them all up: 72 + 12 + 96 = 180 Daltons! See? It’s like a tiny, perfect molecular recipe. This precise structure is what gives glucose its unique properties, allowing it to be easily broken down by your body for energy.
It’s fascinating to think that this seemingly simple number, 180, represents a specific arrangement of atoms that’s so crucial for life. It's like the molecular equivalent of a perfectly balanced chord in music or a harmonious color combination in art. Nature’s got a knack for elegant simplicity, wouldn’t you agree?

Glucose in Your Daily Grind (and Why It Matters)
You might be thinking, "Okay, that's neat, but how does this 180 Dalton fact actually help me?" Well, understanding glucose is key to understanding your own energy levels and making smarter food choices. When you eat carbohydrates, your body breaks them down into glucose. This glucose then enters your bloodstream, signaling your pancreas to release insulin, which helps usher that sugar into your cells for fuel.
This is where things get interesting. Different types of carbohydrates are digested at different rates. Simple sugars, like those found in candy or sugary drinks, are broken down quickly, causing a rapid spike in blood glucose. This can lead to a sugar rush followed by a crash, leaving you feeling tired and sluggish. Think of it as a short, intense burst of energy, like a sprinter.
Complex carbohydrates, like those in whole grains, vegetables, and legumes, are digested more slowly. This provides a steady, sustained release of glucose into your bloodstream, giving you more consistent energy throughout the day. This is more like a marathon runner – a steady pace that lasts.
So, that 180 Dalton molecule is central to this whole energy dance. When you choose foods rich in complex carbs, you’re managing the rate at which these 180 Dalton units are delivered to your cells, leading to a more balanced and enjoyable energy experience.
Sweet Tips for a Balanced Life
So, how can you channel this knowledge for a more energized and happy life? Here are a few easy-going tips:

- Embrace Whole Foods: Opt for fruits, vegetables, and whole grains. They’re naturally packed with the goodness that your body can use efficiently. Think of them as slow-release energy powerhouses.
- Hydration is Key: Sometimes, when you feel sluggish, you might just be a bit dehydrated. Water is essential for all sorts of cellular processes, including how your body uses glucose. Keep that water bottle handy!
- Listen to Your Body: Pay attention to how different foods make you feel. Do you get a sudden energy boost and then a crash after a sugary snack? Or do you feel sustained energy after a balanced meal? This is your body giving you clues.
- Mindful Sweetness: It's okay to enjoy sweet treats! The key is moderation and awareness. Savor that cookie or a square of dark chocolate. It’s about enjoying the moment, not overdoing it.
- The Power of Protein and Fat: Pairing carbohydrates with protein and healthy fats can help slow down the digestion of glucose, leading to a more stable blood sugar level. So, an apple with a handful of almonds? Smart move!
It’s not about deprivation; it’s about making conscious choices that support your well-being. Think of it as curating your energy sources, ensuring you have the steady fuel you need to tackle your day with a smile.
Beyond the Bite: Cultural and Fun Facts
The significance of glucose extends far beyond our immediate physical needs. For centuries, humans have been fascinated by sweetness and its effects. The very concept of “sugar rush” is a testament to our intuitive understanding of how certain foods impact our mood and energy.
Did you know that honey, one of nature's oldest sweeteners, is primarily a mixture of fructose and glucose? It’s been used for both sustenance and medicinal purposes for millennia, valued for its quick energy boost. Ancient Egyptians even used it in their embalming practices – talk about a sweet legacy!
And what about the science fiction of it all? In popular culture, from sci-fi stories where futuristic societies might have advanced ways to synthesize or metabolize energy, the concept of fuel is always central. While we're not quite at the point of beaming up our energy needs, the fundamental principle remains: our bodies need fuel, and glucose, with its 180 Dalton weight, is a primary source.
Consider sports drinks. They're formulated to replenish glucose and electrolytes lost during strenuous activity, providing a quick energy boost. The science behind them is all about delivering those 180 Dalton molecules efficiently when your body needs them most. It’s a modern-day application of an ancient biological process.
Even in art and literature, the idea of sweetness and its allure is a recurring theme. Think of the descriptions of decadent feasts or the symbolic use of sugar to represent pleasure and indulgence. It’s a universal language that, at its core, relates back to the simple joy and energy that glucose provides.
A Little Something to Chew On
It's amazing how a single, seemingly dry number like "the molecular weight of glucose is 180 Daltons" can be a gateway to understanding so much about ourselves and the world around us. It's a reminder that even the most complex systems, like our own bodies, are built from these fundamental, elegant units.
Every time you enjoy a piece of fruit, a slice of bread, or a sweet treat, you're interacting with this fundamental molecule. It's the engine that keeps you going, the spark that fuels your laughter, and the energy that powers your dreams. It’s a little bit of science woven into the fabric of everyday life, adding a layer of appreciation to the simple act of being alive and energized.
So, the next time you reach for something sweet or feel that familiar energy surge, give a silent nod to that humble, yet mighty, 180 Dalton molecule. It's working hard for you, in more ways than you might imagine, making your everyday just a little bit brighter and a whole lot more energetic. And that, my friends, is pretty sweet indeed.
