The Lewis Structures Of Four Compounds Are Given.

I remember when I was a kid, maybe seven or eight, and my dad brought home this ancient, dusty chemistry set. It was mostly just colored liquids in weird-shaped bottles and some metal bits that looked suspiciously like old coins. But there was this one thing… this little booklet. It had these drawings, these weird little stick-figure representations of molecules. They looked like tiny, organized explosions, or maybe even little alien spacecraft. I didn’t understand a lick of it back then, but the sheer weirdness of it stuck with me. Little did I know, those were my first, albeit highly abstract, introductions to Lewis structures. And let me tell you, they’re way cooler than any of the fizzy, colorful experiments in that old box!
Fast forward a couple of decades, and here I am, still fascinated by those same little drawings. You see, those seemingly simple diagrams are actually the secret sauce to understanding how atoms gang up to form all the stuff around us. They’re like the architectural blueprints for the molecular world. And honestly, once you get the hang of them, it’s like unlocking a new level of reality. You start seeing everything differently, from the water you drink to the air you breathe. Pretty neat, huh?
So, let’s dive into the wonderfully quirky world of Lewis structures, shall we? We’ve got four compounds to play with today, and each one is going to show us something a little bit different, a little bit special. Think of it like a molecular speed dating session. We’re going to get acquainted with some of the fundamental rules and see how they play out in practice. No need for a lab coat, just your curiosity and maybe a cup of coffee. You got this.
Let's Meet Our Molecular Guests!
Okay, so the basic idea behind a Lewis structure is to represent the valence electrons of atoms and how they’re shared or transferred to form bonds. Remember valence electrons? Those are the outermost electrons, the ones that are just itching to get involved in some chemical drama. They’re the life of the party in an atom.
We usually represent these electrons as dots. And when atoms share electrons, they form a covalent bond, which we draw as a line connecting the two atoms. Each line typically represents a pair of shared electrons, also known as a bond pair. Atoms also have lone pairs, which are valence electrons that aren't involved in bonding. These are also shown as pairs of dots, hanging out around a single atom.
The ultimate goal for most atoms in a Lewis structure is to achieve an octet, meaning they want to have eight valence electrons around them, just like the noble gases. Noble gases are the aloof, standoffish ones of the periodic table, content in their electron-filled shells. The rest of us are just trying to be like them, basically.
Now, let’s unwrap our first compound. It’s a classic, a real workhorse of chemistry: Water, H₂O.
Compound 1: Water (H₂O) - The Ubiquitous One
Water. It’s everywhere. Essential for life, good for making tea, and occasionally causes major inconvenience when it decides to freeze on the roads. But what’s going on under the hood? Let’s draw it!
First, we need to count the total number of valence electrons. Oxygen is in Group 16, so it has 6 valence electrons. Hydrogen is in Group 1, so each of our two hydrogens has 1 valence electron. Total: 6 + (2 * 1) = 8 valence electrons.
Next, we arrange the atoms. Oxygen is usually the central atom because it's more electronegative than hydrogen. So, we’ll put oxygen in the middle and the two hydrogens on either side. Looks like this: H O H.
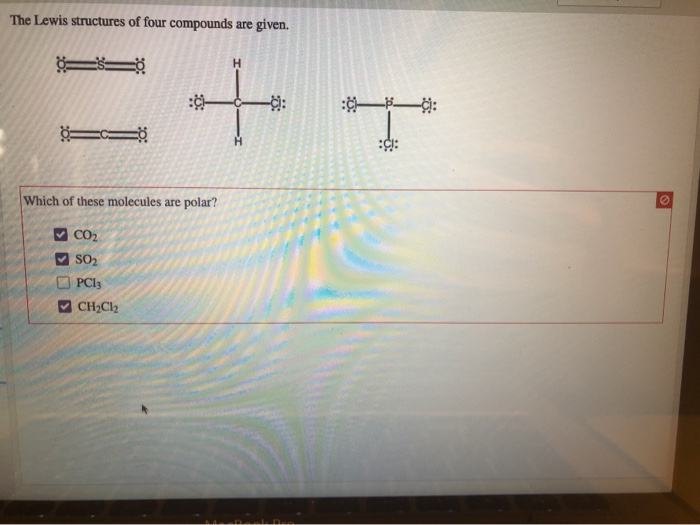
Now, let’s connect them with single bonds. We’ll use a line for each bond, representing a shared pair of electrons. So we have H-O-H. That used up 2 bonds * 2 electrons/bond = 4 valence electrons.
We have 8 total valence electrons and we’ve used 4. That leaves us with 4 electrons. Where do they go? We need to fill the octets, or at least give everyone the electrons they need. The hydrogens are happy with their single bond (2 electrons), they're already sorted. The oxygen, however, only has 4 electrons around it (two from each bond). It needs 4 more to reach its octet.
So, we’ll place the remaining 4 electrons as two lone pairs on the oxygen atom. So the structure looks like this: a central oxygen with two single bonds to hydrogens, and two lone pairs on the oxygen. See those lone pairs? They’re super important for water’s properties, like its bent shape and ability to dissolve so many things. Sneaky, right?
Let’s count again to be sure. Each hydrogen has 2 electrons (from the bond). The oxygen has 4 electrons from the two bonds, plus 4 electrons from its two lone pairs. 4 + 4 = 8. Bingo! Octet achieved for oxygen. Hydrogens are content with their duet. And that, my friends, is the Lewis structure of water. Simple, yet profound.
Compound 2: Carbon Dioxide (CO₂) - The Breath of Life (and the Greenhouse Effect)
Alright, next up, we have carbon dioxide. This one is pretty fascinating because it’s essential for plants (photosynthesis, anyone?) but also a major player in climate change. It’s a molecule with a bit of a dual personality.
Let’s tally the valence electrons. Carbon is in Group 14, so it has 4 valence electrons. Oxygen is in Group 16, so it has 6. We have two oxygens, so that’s 2 * 6 = 12. Total valence electrons: 4 + 12 = 16.
Now, who’s the central atom? Carbon is generally less electronegative than oxygen, and it also tends to form more bonds, so carbon is our central atom. We arrange it like this: O C O.
Let’s start with single bonds to get things connected: O-C-O. That uses 4 electrons (2 bonds * 2 electrons/bond). We have 16 - 4 = 12 electrons left.
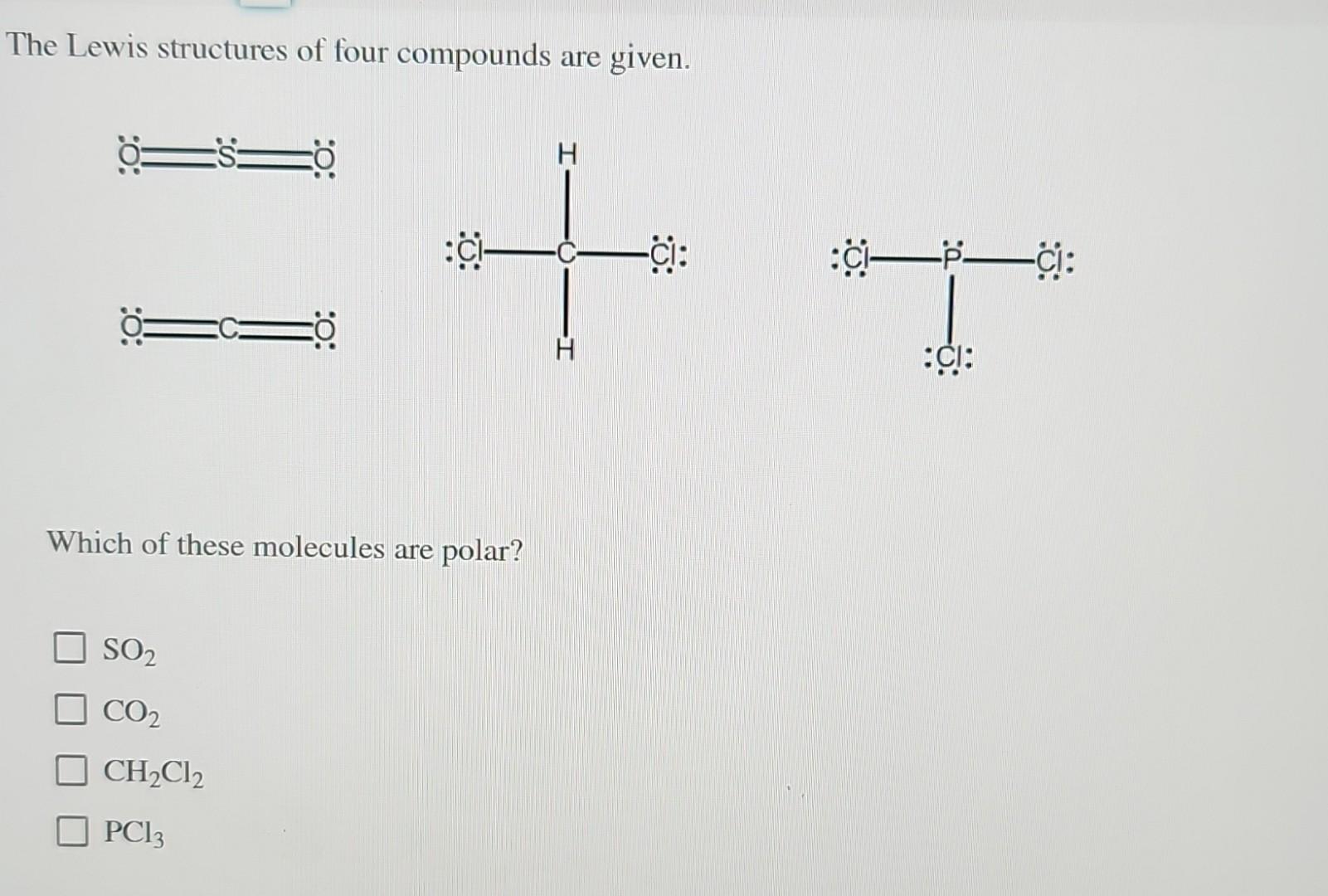
Let’s give electrons to the outer atoms first to satisfy their octets. Each oxygen needs 6 more electrons to reach 8. So, we’ll put 6 dots around each oxygen. That uses up all 12 of our remaining electrons (6 on the left oxygen, 6 on the right oxygen).
Now, let’s check our octets. Each oxygen has 2 electrons from the bond plus 6 from the lone pairs, so they have their octets. Great! But what about the carbon in the middle? It only has 4 electrons around it (2 from each single bond). Uh oh. It’s short of its octet.
This is where things get interesting. When an atom doesn’t have enough electrons, we can move lone pairs from the outer atoms to form multiple bonds. We need the carbon to have 4 more electrons. If we take one lone pair from the left oxygen and move it to form a double bond between that oxygen and the carbon, and then do the same with the right oxygen… what happens?
The structure becomes: O=C=O. Now let’s recount. Each oxygen has 4 electrons from the double bond, plus 4 electrons from its two remaining lone pairs. 4 + 4 = 8. Octet for both oxygens!
And the carbon? It has 4 electrons from the left double bond and 4 electrons from the right double bond. 4 + 4 = 8. Octet for carbon too! We’ve used all 16 valence electrons, and everyone is happy with an octet. This is the correct Lewis structure for carbon dioxide, and it shows that CO₂ has double bonds between the carbon and each oxygen. These double bonds are strong and contribute to its linear shape. Pretty cool how a molecule can be so vital and yet contribute to such big global issues, right?
Compound 3: Ammonia (NH₃) - The Pungent One
Okay, time for something a little… stronger smelling. Ammonia. If you’ve ever been near a strong cleaning product or, well, let’s just say certain animal facilities, you’ve encountered ammonia. Its pungent odor is a signature.
Let’s get our electron count. Nitrogen is in Group 15, so it has 5 valence electrons. Hydrogen is in Group 1, so each of our three hydrogens has 1 valence electron. Total valence electrons: 5 + (3 * 1) = 8.
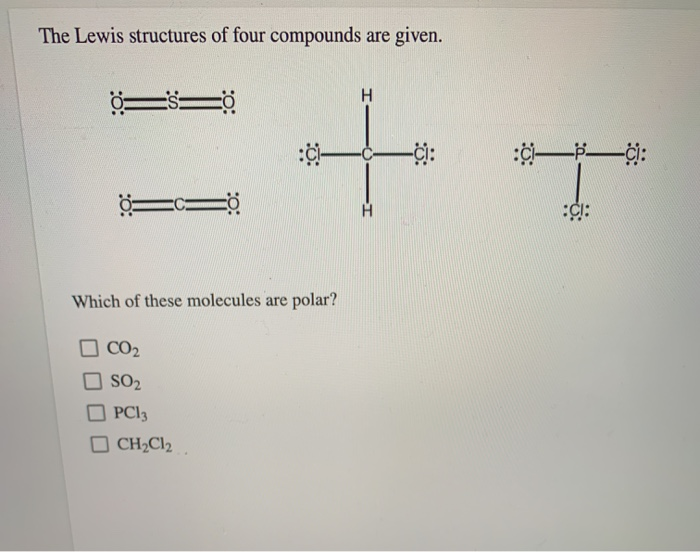
Nitrogen is usually the central atom because it’s more electronegative than hydrogen and forms more bonds. So, we put nitrogen in the middle and arrange the three hydrogens around it. N with three H’s attached.
Let’s connect them with single bonds: H-N-H with another H attached to N. This uses 3 bonds * 2 electrons/bond = 6 valence electrons. We have 8 - 6 = 2 electrons left.
The hydrogens are all happy with their single bond (2 electrons). The nitrogen, however, currently only has 6 electrons around it (2 from each of the three bonds). It needs 2 more to reach its octet.
We have exactly 2 electrons left! Perfect! We place these remaining 2 electrons as a lone pair on the nitrogen atom. So the structure is a central nitrogen bonded to three hydrogens, with one lone pair of electrons on the nitrogen. See that lone pair? That’s a really important feature of ammonia! It’s what makes ammonia a base and allows it to react with acids. It’s like a little hook ready to grab onto something.
Let’s double-check. Each hydrogen has 2 electrons. The nitrogen has 6 electrons from the three bonds plus 2 electrons from the lone pair. 6 + 2 = 8. Octet achieved for nitrogen. All good!
The presence of that lone pair on the nitrogen atom also gives ammonia a pyramidal shape, not linear like CO₂ or bent like water. It’s all about how those electron groups arrange themselves to get as far away from each other as possible. Nature loves efficiency, you know?
Compound 4: Methane (CH₄) - The Simplest Hydrocarbon
Finally, let’s look at methane. This is the simplest hydrocarbon, the primary component of natural gas. It’s also a greenhouse gas, just like CO₂, but often in higher concentrations. It’s everywhere, even in our own digestive systems – yes, really!
Time for electron counting again. Carbon is in Group 14, so it has 4 valence electrons. Hydrogen is in Group 1, so each of our four hydrogens has 1 valence electron. Total valence electrons: 4 + (4 * 1) = 8.
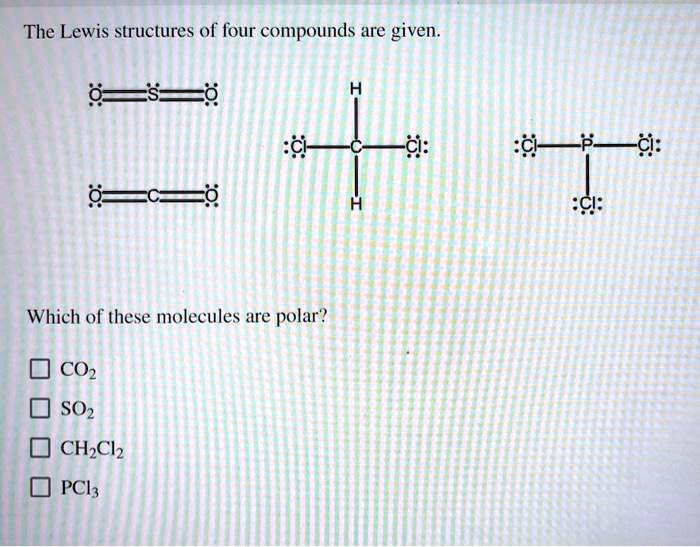
Carbon is the central atom here, as it forms more bonds than hydrogen and is less electronegative. So, we place carbon in the middle and surround it with our four hydrogen atoms. C with four H’s.
Let’s connect them with single bonds. H-C-H, with two more H’s attached to the central C. This uses 4 bonds * 2 electrons/bond = 8 valence electrons. We have 8 - 8 = 0 electrons left.
Now, let’s check octets. Each hydrogen has 2 electrons from its single bond. They’re perfectly happy. And the carbon in the middle? It has 8 electrons around it (2 from each of the four single bonds). It has achieved its octet!
So, the Lewis structure of methane is a central carbon atom single-bonded to four hydrogen atoms, with no lone pairs on any of the atoms. This is a great example of a very stable molecule where all valence electrons are used in bonding. It’s a clean, simple structure.
Methane has a tetrahedral shape. This is because the four electron groups (the four C-H bonds) around the central carbon atom repel each other and arrange themselves in a way that maximizes the distance between them, forming a tetrahedron. It’s like a perfectly balanced arrangement.
The Takeaway
So there you have it! Four compounds, four Lewis structures, and a whole lot of understanding packed into those little dot and line diagrams. From the bent beauty of water to the double-bonded efficiency of carbon dioxide, the basic lone pair of ammonia, and the tetrahedral simplicity of methane, we’ve seen how these structures reveal the arrangement of electrons and the bonds that hold atoms together.
Remember, the key steps are: count valence electrons, determine the central atom, connect with single bonds, distribute remaining electrons to outer atoms, then move to the central atom, and finally, form multiple bonds if needed to satisfy octets. It might seem a bit like a recipe at first, but with practice, it becomes second nature.
These structures aren’t just pretty drawings; they’re the foundation for understanding so much more about chemistry – molecular shape, polarity, reactivity, and even physical properties like boiling points. So next time you see water flowing, or breathe in the air, or even light a gas stove, take a moment to appreciate the elegant dance of electrons that Lewis structures so cleverly depict. It’s a tiny glimpse into the amazing molecular machinery that makes our world tick. Isn’t science just the coolest?
