The Lattice Energy For Ionic Crystals Increases As

Alright, let's talk about something a little… spicy. It’s about how certain things get more intense. You know, like a good chili or a really dramatic movie cliffhanger. In the world of tiny, sparkly things, there’s a principle that’s kind of like that. It’s called Lattice Energy.
Now, don't let the fancy name scare you. Think of it as the "stickiness factor" for those little building blocks that make up crystals. The higher the stickiness, the more energy it takes to pull them apart. It’s like trying to separate LEGO bricks that have been glued together versus ones that just snap.
So, when does this Lattice Energy crank up the volume? Well, it’s not exactly rocket science, but it does involve some tiny players. These are your ions, the charged little critters that make up ionic crystals. They’re like magnets, some with a positive charge (think tiny little protons chilling out) and some with a negative charge (imagine tiny little electrons having a party).
The first big player that makes our Lattice Energy go “WHOA!” is the charge of these ions. Imagine you have two magnets. If they’re only a little bit magnetic, they’ll stick together okay. But if they’re super-duper strong magnets, BAM! They’re practically impossible to pry apart.
So, when the ions have bigger charges, like a +2 or a -2, instead of just a +1 or a -1, they have a much stronger attraction. It’s like going from a gentle handshake to a full-on, bear hug. This means it takes a whole lot more effort, a whole lot more energy, to break those bonds.
It's all about the pull!
Think of it like this: a tiny, shy kitten might nudge a toy. A big, powerful lion, however, will drag that toy across the savanna. The force behind the action is just… different. Same with ions. Bigger charges mean a bigger, more insistent pull.
So, if you see ions with numbers like 2 or 3 next to their little plus or minus signs, get ready. The Lattice Energy is probably going to be pretty impressive. It’s like the crystal is saying, “Nope, you’re not getting out of here easily!”

The next thing that makes our Lattice Energy party even more wild is the size of the ions. Now, this one might seem a little counterintuitive at first. You'd think bigger things would be easier to grab, right? Wrong! In the world of ionic crystals, it's often the opposite.
Imagine trying to hug someone. If they’re super tiny, like a little toddler, you can wrap your arms all the way around them easily. If they’re, say, a very tall basketball player, you can only get so close. Your arms can't quite reach all the way around.
With ions, the smaller they are, the closer they can get to each other. And when they get closer, their attractive forces get much stronger. It’s like their little charged hearts are beating right next to each other.
Tiny can be mighty!
So, when you have very small ions, the distance between their centers is small. This means the electrostatic attraction between them is super concentrated. Think of a laser beam versus a floodlight. The laser is small, focused, and incredibly intense.
This is why crystals made of smaller ions often have higher Lattice Energy. They’re packed in tight, and they’re not letting go without a serious fight. It’s a bit of an underdog story, where the little guys pack the biggest punch.

Therefore, when you’re looking at ionic crystals and wondering about their stickiness, keep an eye on the size of the ions involved. If they’re teensy-tiny, prepare for some serious Lattice Energy.
So, to sum it up, the Lattice Energy for ionic crystals increases as you get ions with bigger charges and ions that are smaller in size. It’s like a double whammy of "stay put!"
Think of it as nature’s way of saying, "If you want to break this down, you're going to have to work for it!" And honestly, sometimes, that’s kind of admirable. These crystals are really committed to their structures.
It’s a beautiful, albeit sometimes stubborn, dance of charges and proximity. And who knew that tiny, invisible things could have such a powerful grip?
It’s a bit like people. Sometimes the quietest ones have the strongest wills. Or the ones with the most passion are the hardest to dissuade.

This principle applies to all sorts of things you might encounter. From the salt on your fries to the minerals in rocks, the way these little ions hold on is fundamental.
So, the next time you see a crystal, take a moment to appreciate its internal commitment. It’s all thanks to the mighty forces of Lattice Energy, driven by those feisty ions.
It’s a concept that makes perfect sense once you stop overthinking it and just picture those little charged particles getting really, really close and holding on with all their might.
And that, my friends, is how you get a seriously sticky situation in the world of ionic crystals. Higher charges and smaller sizes mean a tougher bond.
It's a little bit like trying to get toddlers to share their favorite toys versus trying to get adults to share a parking spot. The stakes, and the forces involved, are just different.
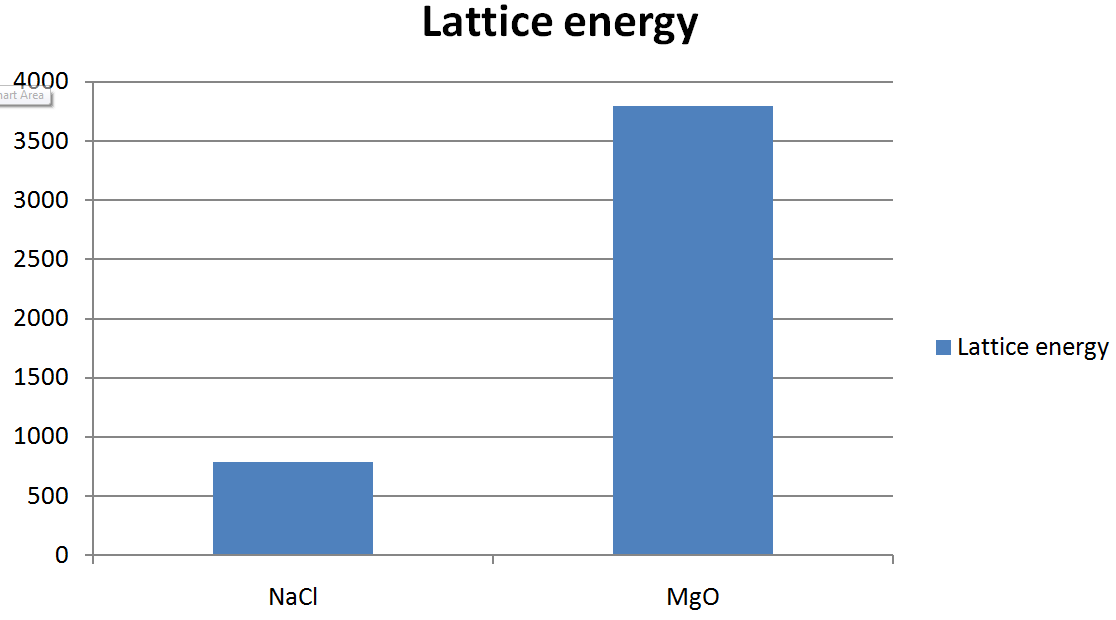
So, remember: bigger charges and smaller ions. That’s your secret code for unlocking the mystery of increased Lattice Energy. No complicated formulas needed, just a good mental image.
It’s a principle that’s both elegant and a little bit intimidating. These ionic bonds are not to be messed with!
And that, in a nutshell, is the playful truth about when Lattice Energy decides to throw a big, energetic party. It's all about the power of attraction between the smallest, most charged, and closest of friends.
So next time you marvel at a shiny crystal, give a little nod to the powerful, unseen forces holding it all together. It's a testament to the fact that sometimes, the smallest things have the biggest impact, especially when they have high charges and are up close and personal!
