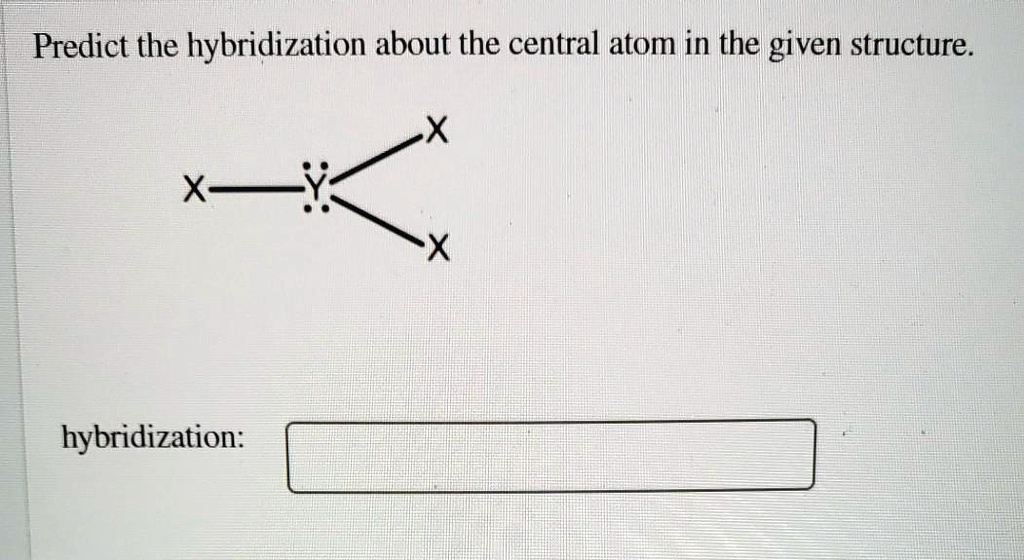
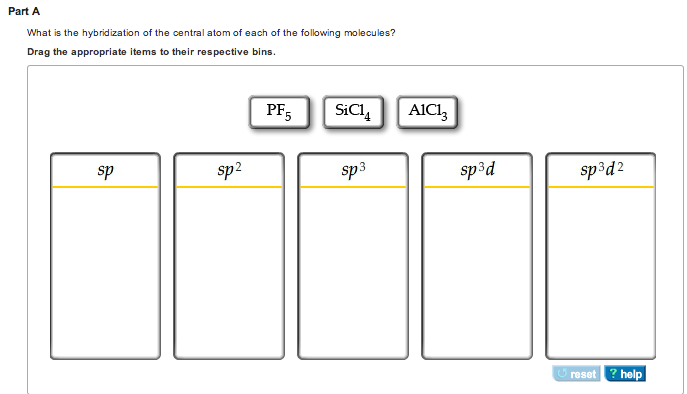
The Hybridization Of The Central Atom In O3 Is
Hey there, science enthusiasts and curious cats! Ever wonder what makes O3, you know, O3? We're talking about ozone, that fancy gas floating around up in our atmosphere. It's super important, but today, we're going to chat about something a little more behind-the-scenes. We're diving into the wacky world of how its central atom gets its vibe on. Yep, we're talking about hybridization.
Think of it like this: atoms are like Lego bricks. They have these neat little slots for electrons, their building blocks. But sometimes, these slots get a little… awkward. They're not quite the right shape or energy to make perfect connections. So, what do they do? They get creative!
This is where the magic of hybridization comes in. It's basically the atom's way of saying, "You know what? Let's mix and match these electron slots to make some really awesome, stable connections." It's like taking a bunch of different-sized Lego bricks and molding them into one super-versatile brick. Pretty neat, right?
Now, O3, our star molecule, has one central oxygen atom. This oxygen atom is feeling a bit social. It's got two other oxygen buddies it wants to hang out with. To do that, it needs to get its electron game on point.
So, what's the deal with O3's central oxygen atom? The answer is sp2 hybridization. Sounds a bit like a robot trying to order coffee, doesn't it? "I'll have one sp2, please!"
Let's break down what "sp2" actually means. Imagine our central oxygen atom has three main types of electron slots, or orbitals: an 's' orbital and three 'p' orbitals. The 's' orbital is like a perfectly round, cozy little home for electrons. The 'p' orbitals are more like dumbbells, a bit more complex.

Normally, these orbitals are separate. But for O3, the central oxygen atom decides to have a little mixer. It takes one 's' orbital and two of its 'p' orbitals and mashes them together. Poof! They become three new, identical orbitals. These are called sp2 hybrid orbitals. They're all the same shape and energy, ready for some serious bonding action.
Why is this cool? Because now the central oxygen has three of these sp2 hybrid orbitals to work with. And guess what? It needs to connect with two other oxygen atoms. That leaves one sp2 orbital free for… drumroll… a lone pair of electrons!
Ah, the lone pair! Electrons are like little social butterflies. They love to pair up. When they have a partner, they're happy. But sometimes, they're just chilling solo in an orbital. These are lone pairs. In O3, the central oxygen has one lone pair hanging out in one of its sp2 hybrid orbitals. It's like the atom's personal space.
So, we've got three sp2 hybrid orbitals on our central oxygen. Two of these are used to form bonds with the other two oxygen atoms. And the third sp2 orbital? It's happily housing that lone pair of electrons.
But wait, there's more! Remember those 'p' orbitals we started with? We used two of them for the sp2 hybridization. But there was one 'p' orbital left over! This leftover 'p' orbital is different. It didn't join the hybridization party. It remains unhybridized.
This unhybridized 'p' orbital is super important. It's oriented in a way that it can overlap with a 'p' orbital on one of the other oxygen atoms. This overlap creates a special type of bond called a pi bond. And when you have a sigma bond (formed by the direct overlap of orbitals) and a pi bond, you get a double bond. Bingo!
So, the central oxygen atom in O3 ends up forming one single bond with an oxygen atom and a double bond with the other oxygen atom. This is why ozone has that unique structure. It's not a perfectly straight line. It's more like a bent boomerang!
The sp2 hybridization is what gives O3 its bent shape. The three sp2 hybrid orbitals spread out as far as possible, trying to avoid each other. Imagine them as three balloons tied together at the center. They'll naturally push away from each other, creating an angle. In O3, this angle is about 116.8 degrees. Not a perfect straight line, not a tight triangle, but something in between. It's just right for its molecular personality.
And that lone pair of electrons? It also takes up space. It contributes to the repulsion between the electron groups, further influencing the molecular geometry. It's like that one friend who always needs a bit more elbow room!
Why is this hybridization stuff even a big deal? Well, it explains everything about how molecules behave. It tells us why O3 is bent, why it reacts the way it does, and why it's so good at, you know, blocking harmful UV rays. It's like the secret handshake of the molecular world.
The fact that sp2 hybridization allows for both single and double bonds is a huge deal. It's what gives molecules their diverse personalities and chemical properties. Without it, our world would be a very different, and probably much less interesting, place.
Think about it: if the oxygen atom in O3 didn't hybridize, it would have to use its regular 's' and 'p' orbitals. This would lead to different bond angles and strengths, and O3 might not be the same protective shield we rely on. It's pretty mind-blowing how these tiny electron arrangements have such massive consequences.
And the best part? This isn't just about O3. This sp2 hybridization is a common theme in chemistry. Many molecules we encounter every day use it. It's like a universal tool in the atom's toolbox. Learning about it is like unlocking a secret language that helps you understand the very fabric of the universe.
So next time you hear about ozone, remember the sp2 hybridization. It's the unsung hero, the hidden architect, the reason behind the molecule's quirky charm and vital role. It’s a little bit of molecular engineering that makes all the difference. Isn't science just a blast when you look at it this way? Keep asking questions, and keep exploring the amazing world of atoms and their electron dances!
