The Four Bonds Of Carbon Tetrachloride Is Polar

Hey there, science enthusiasts and curious cats! Ever wonder why some things jiggle and some things stick? Or why that perfectly clean glass seems to attract dust like a magnet? Well, buckle up, because today we're diving into the wonderfully weird world of molecules, and specifically, one you might have heard of: carbon tetrachloride. Now, before you imagine some scary, lab-coat-wearing situation, let's get one thing straight: we're keeping this fun, light, and maybe even a little bit sparkly. Because understanding how molecules behave? That's like having a secret decoder ring for the universe!
So, what's the big deal with carbon tetrachloride, or CCl4 as the chemists affectionately call it? It's a molecule made up of one carbon atom smack-dab in the middle, surrounded by four chlorine atoms. Think of it like a little molecular family reunion. Carbon, the host, is chilling, and his chlorine cousins are all invited to the party. And, you know, they're all holding hands, or in chemistry lingo, they're forming covalent bonds.
Now, here's where it gets interesting. We often hear about things being "polar" or "non-polar," and it sounds a bit like a personality quiz for molecules, doesn't it? But in the molecular world, it's all about how the electrons are shared. See, electrons are like the super-energetic kids at the molecular party, zipping around. In a covalent bond, these electrons are shared between two atoms. But here's the kicker: some atoms are a bit more "greedy" for those electrons than others.
Chlorine, for instance, is a bit of a hog when it comes to electrons. It's more electronegative, meaning it pulls those shared electrons closer to itself. So, in carbon tetrachloride, while the carbon and chlorine atoms are bonded, those electron-sharing parties aren't entirely fair. The electrons spend a little more time hanging out with the chlorine atoms.
This uneven sharing creates a slight "charge" on each end of the bond. The chlorine atoms, where the electrons are spending more time, get a slightly negative charge (we call this a partial negative charge). And the carbon atom, where the electrons are spending less time, gets a slightly positive charge (a partial positive charge). It's like a tiny tug-of-war happening all the time within each bond!

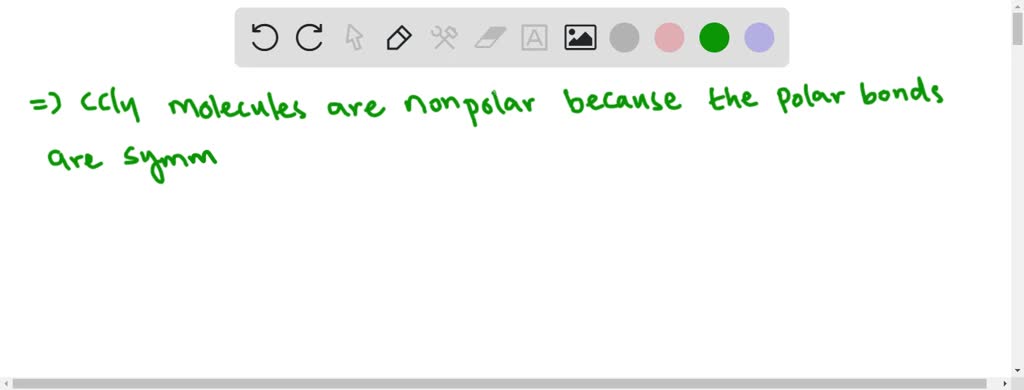
So, we've got four of these polar bonds in our carbon tetrachloride molecule. You'd think, with all these polar bonds, the whole molecule would be, well, polar, right? Like a tiny little battery? But here's the cosmic joke, the delightful twist that makes chemistry so much fun: it's actually not polar! Gasp!
Why, you ask, with all that electron-hogging and partial charges, does carbon tetrachloride decide to be non-polar? It all comes down to the shape of the molecule. Imagine our carbon atom as a central point, and the four chlorine atoms are arranged around it like the points of a perfectly symmetrical 3D star, or more technically, a tetrahedron. They are spread out as far as possible from each other, creating a beautiful, balanced symmetry.

Think of it this way: each polar bond has a little arrow pointing towards the more electronegative chlorine atom. Now, if you were to add up all those little arrows (and in science, we have fancy ways of doing this called vector addition, but let's keep it light!), they all point outwards in different directions, perfectly cancelling each other out. It's like four people pulling on a rope in exactly opposite directions – the rope doesn't move!
Because of this perfect symmetry, the slight negative charges on the chlorine atoms and the slight positive charge on the carbon atom are distributed evenly throughout the entire molecule. There's no one "side" that's significantly more negative or positive. It's like a perfectly balanced seesaw, even though the weight distribution within each individual plank might be a little uneven. The overall effect is neutral, or non-polar.
This might seem like a small detail, but it has huge implications for how carbon tetrachloride behaves. Molecules that are polar tend to attract other polar molecules, just like magnets. They love to mix and mingle. Non-polar molecules, on the other hand, are a bit more independent. They prefer the company of other non-polar molecules.
This is why oil and water don't mix! Water is a polar molecule, and oil is largely non-polar. They just don't "get" each other. But carbon tetrachloride, being non-polar, is actually a fantastic solvent for other non-polar substances. It can dissolve things like fats, oils, and greases that water can't touch. So, while we don't recommend playing with it at home (it has some health concerns and is no longer widely used in households!), understanding its properties helps us appreciate the invisible forces at play all around us.

Isn't that neat? It's like a masterclass in deception! You look at the individual bonds, and you see all this polarity, all this electron-pulling. But put them all together in the right arrangement, and the whole thing declares itself non-polar. It's a testament to the power of geometry and how the big picture can be completely different from the sum of its parts.
This stuff is everywhere! From the way your detergent works to how paint sticks to your walls, molecular properties are the unsung heroes of our everyday lives. And the fact that a molecule can be made of polar parts but behave as a non-polar whole? That's just pure scientific poetry!
So, the next time you see a clean surface, or perhaps a greasy dish, take a moment to marvel at the molecular dance happening beneath the surface. The four bonds of carbon tetrachloride, individually polar but collectively non-polar, are a perfect reminder that sometimes, it's the arrangement, the symmetry, and the overall structure that defines the outcome. It’s a lesson in how even seemingly opposing forces can create a harmonious balance. Keep asking questions, keep exploring, and let the wonders of science fill your world with endless fascination!
