The Following Picture Of An Aqueous Solution Best Represents
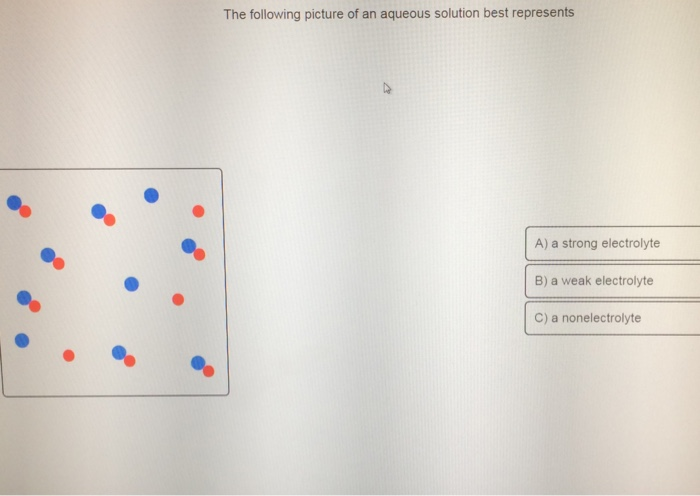
Hey there, science explorers and curious cats! So, you’ve probably seen a picture, or maybe even a diagram, of an aqueous solution and wondered, "What is this visual jazz all about?" Don't worry, we're not diving into a super complex chemistry textbook here. Think of this as a friendly chat over coffee, where we break down what that picture is really trying to tell us in a way that's as easy as making instant ramen. (And arguably more interesting, depending on your ramen flavor preference.)
First off, let’s get our heads around what an "aqueous solution" even means. Forget the fancy jargon for a sec. It’s basically something dissolved in water. Yup, that’s it! Water is like the ultimate party host, and when something else joins the party, it becomes an aqueous solution. Think of it like dissolving sugar in your tea. The tea is your water, and the sugar is your solute. Voilà! You’ve got an aqueous solution. Pretty neat, right? It's like a tiny microscopic rave happening in your cup.
Now, when we’re talking about what a picture best represents, we're usually looking at how those dissolved bits, the solute particles, are behaving. Imagine you’ve got a bunch of tiny dancers (the solute) and a huge dance floor (the water). The picture is basically showing us how these dancers are spread out and interacting on the floor. Are they all huddled together? Are they doing the cha-cha solo? Or are they having a full-on disco inferno?
Let's break down the most common scenarios you’ll see in these aqueous solution pictures. Sometimes, you'll see a bunch of little circles or spheres. These are our solute particles. And then you'll see a whole lot of other, even smaller circles or squiggly lines representing the water molecules. The key is how these two groups are interacting.
The "All Chummy-Chummy" Picture
One common visual shows the solute particles, let's call them "solute buddies," all hanging out together. They're clumped up, almost like they're whispering secrets to each other or forming a tiny, exclusive club. This usually represents a situation where the solute hasn't fully dispersed. Think of trying to dissolve a big chunk of something in water. It might start off like this, with the edges of the chunk slowly breaking away and dissolving.
In these pictures, you'll often see the solute buddies sticking together, and the water molecules are kind of milling around the edges, trying to coax them out. It’s like the water is saying, "Come on out, guys! The dance floor is great!" But the solute buddies are a little shy, or maybe they just really like each other's company. This can happen when you haven't stirred enough, or the solute just doesn't dissolve super easily. It’s the chemical equivalent of someone taking forever to get out of their cozy blanket.
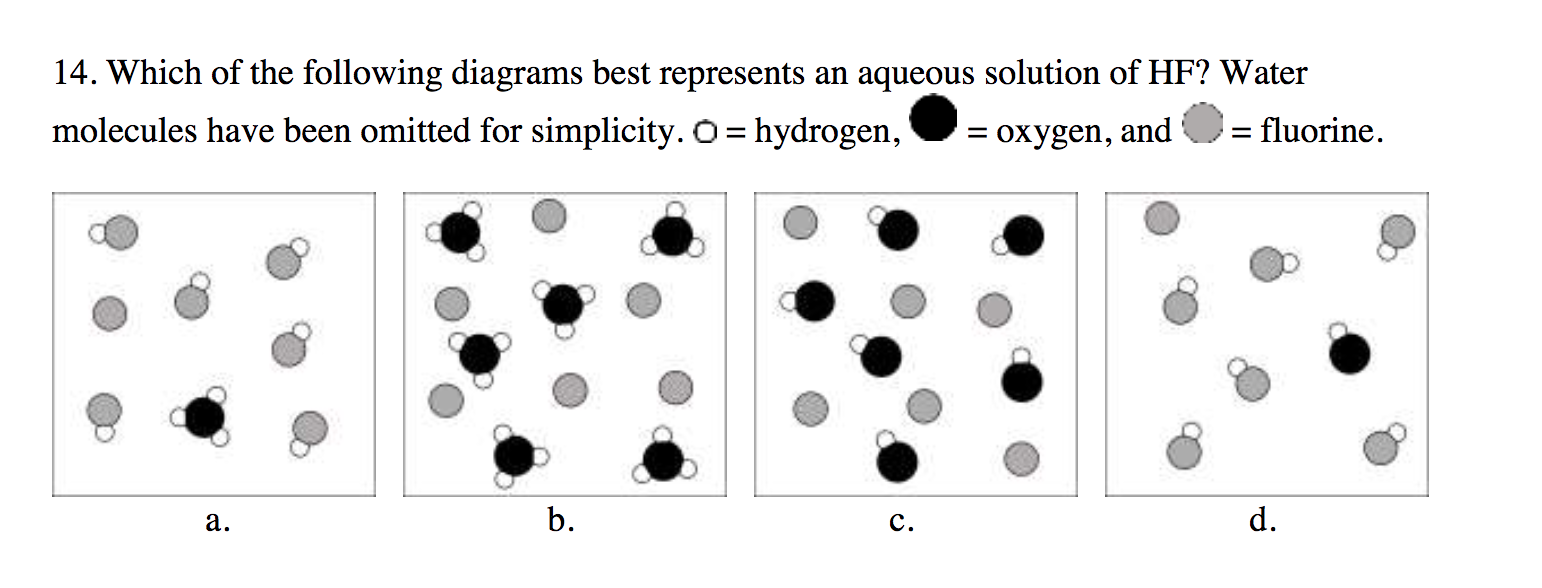
These pictures are good for showing us what happens before everything is perfectly mixed. It’s the messy middle stage, the awkward silence before the music starts. And hey, even awkward silences have their own charm, right? They’re a reminder that sometimes, things take a little time to get going. Just like that perfect cup of coffee – you can’t rush perfection, can you?
The "Independent Sprinkles" Picture
Then there’s the picture where the solute buddies are all spread out, like tiny, happy, independent sprinkles on a giant cake. Each solute buddy is doing its own thing, zipping around and interacting with the water molecules. They’re not huddled together; they're enjoying their personal space, but still very much part of the solution.
This is usually what we mean by a true solution. The solute has completely dissolved and dispersed evenly throughout the water. Imagine adding a spoonful of fine sugar to your water and stirring. After a bit, you can’t see the sugar anymore, right? It’s vanished, but it's there, spread out like a silent disco. The picture will show these solute particles scattered everywhere, with water molecules surrounding them, keeping them company.
These images are fantastic for illustrating homogeneity. That's a fancy word meaning "the same all the way through." It means if you took a tiny sip from the top of your sugary tea, it would taste just as sweet as a sip from the bottom. The solute buddies are so well-distributed, they're like the most popular kids at the party, talking to everyone. They've embraced the water's hospitality and are having a blast!
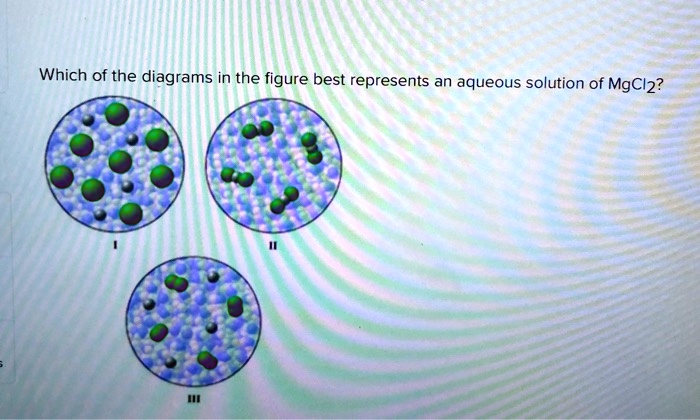
The "Oh No, It's Separating!" Picture
Sometimes, you’ll see a picture that looks a bit… unsettling. It might show the solute buddies starting to clump together again, or maybe forming distinct layers. This is the visual equivalent of the party starting to wind down, or worse, the guests having a bit of a disagreement. This can happen in a few different ways.
One possibility is that the solution is saturated. Imagine you’ve tried to dissolve as much sugar as possible in your tea. Eventually, no matter how much you stir, some sugar will just sit at the bottom, refusing to dissolve. The picture will show some solute buddies dissolved and spread out, but others are still in their solid, clumped-up form, or they're starting to recrystallize and reform. It's like the water's capacity to host has been maxed out. It’s throwing its hands up and saying, "Okay, that's all I can handle!"
Another scenario this picture might represent is instability. Some solutions aren't meant to stay mixed forever. They might separate over time, forming distinct phases. Think of oil and vinegar in salad dressing before you shake it up. The picture would show the solute buddies gathering in one area and the water molecules in another, or perhaps forming a cloudy, unappealing mess. This is the visual of a solution saying, "You know what? I'm just not feeling this anymore. I'm going my own way."
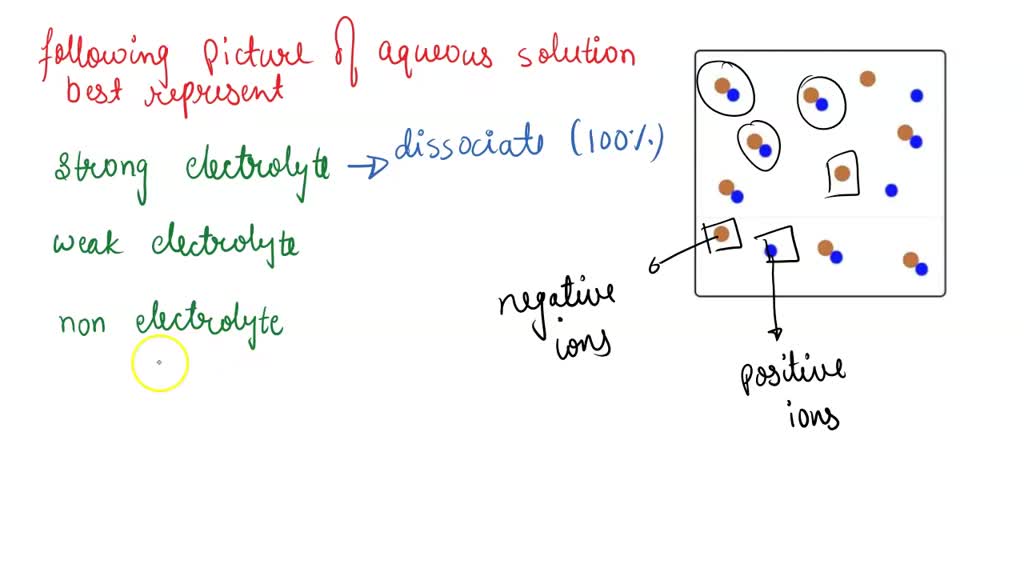
The "Shocked! It's Ions!" Picture
Now, here's where things get a little more electrifying – literally! Some solutes, when they dissolve in water, don't just remain as whole molecules. They break apart into charged particles called ions. These are like tiny electrically charged superheroes, each with their own positive or negative aura. When you dissolve something like salt (sodium chloride, NaCl) in water, it doesn’t just sit there as NaCl. It breaks into positive sodium ions (Na+) and negative chloride ions (Cl-).
Pictures representing these solutions will show the original solute splitting into these charged ions, each surrounded by water molecules. The water molecules, being polar (meaning they have a slightly positive and a slightly negative end), act like little magnets, clustering around these ions and stabilizing them. You’ll see the solute buddies broken into smaller, oppositely charged pieces, and the water molecules strategically positioned around them. It’s like a perfectly orchestrated dance of attraction and repulsion.
This is super important in chemistry and biology. Think about the electrolytes in your body – they are ions that help conduct electrical signals. So, a picture showing these separated ions is representing a solution that can conduct electricity. It’s like the difference between a dull party and a party with a killer sound system and flashing lights – the ions bring the energy!
The "Cloudy Conundrum" Picture
Finally, let's talk about the pictures that look a bit… foggy. You might see a uniform cloudiness throughout the liquid, or perhaps tiny particles suspended but not truly dissolved. This usually represents a suspension or a colloid, not a true solution. In a suspension, the particles are too big to dissolve completely, and they might even settle out over time if left undisturbed. Think of sand in water. If you don’t stir it constantly, the sand will sink to the bottom.

In a colloid, the particles are larger than in a true solution but smaller than in a suspension. They stay dispersed, but they make the liquid look cloudy or opaque. Milk is a classic example of a colloid. The picture would show these medium-sized particles spread throughout the water, making it look hazy. They haven't dissolved, but they're not sinking to the bottom either. They're just… hanging out, making things look a bit mysterious.
So, when you see a picture, your mission, should you choose to accept it, is to observe the solute particles. Are they clumped? Are they scattered as individual units? Have they broken into charged bits? Or are they creating a misty, cloudy effect? Each visual tells a different story about how that substance is interacting with its watery environment.
The beauty of these representations is that they’re simplified windows into a complex microscopic world. They help us understand everything from how medications are absorbed by our bodies to how the oceans maintain their delicate balance. It’s all about those little particles doing their thing in the vast ocean of water!
So, the next time you encounter one of these aqueous solution pictures, don't just glance at it. Take a moment to appreciate the story it's telling. Is it a chaotic dance party? A serene scattering of individuals? Or a tightly packed gathering? Whatever it is, it’s a little snapshot of the incredible, invisible world that makes up so much of our reality. And that, my friends, is pretty darn awesome. Keep exploring, keep questioning, and keep smiling at the wonders of science – one dissolved particle at a time!
