The Equilibrium Constant Kc For The Reaction Pcl3 + Cl2

Ever found yourself staring at a chemical reaction, particularly one involving PCl3 and Cl2, and wondered about its secret little balancing act? Yeah, me neither. Until recently. But then I stumbled upon this thing called the equilibrium constant, or Kc, and let me tell you, it’s like the universe’s way of saying, "It’s complicated, but we’ll settle for a decent compromise."
Imagine you're at a party. Some people are chatting excitedly, others are chilling by the snacks, and a few are nervously eyeing the exit. The party never really ends, does it? People mingle, conversations start and stop, some folks leave, new ones arrive. It's a constant flux. That’s sort of like a chemical reaction at equilibrium. It’s not that the reaction just gives up and stops. Oh no. It’s just that the forward reaction (PCl3 and Cl2 teaming up to form PCl5) and the reverse reaction (PCl5 deciding it’s had enough and breaking apart back into PCl3 and Cl2) are happening at the exact same speed. It's a perfectly choreographed dance, with everyone doing their own thing but somehow staying in sync.
Now, this Kc thing. It’s basically a number that tells us how this dance is going. Is it a wild rave where everyone is forming PCl5 like there’s no tomorrow? Or is it more of a quiet soirée where the PCl3 and Cl2 are just politely mingling, with barely any PCl5 showing up?
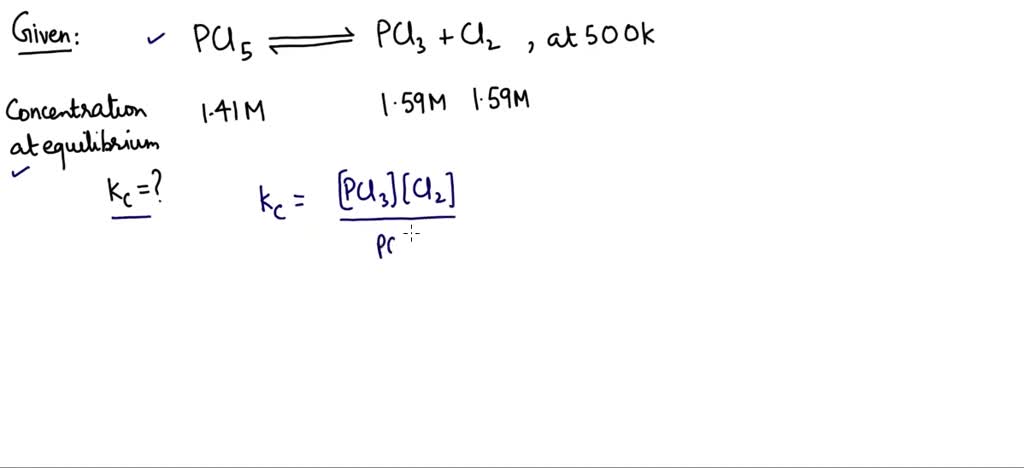
For our specific pals, PCl3 and Cl2, forming PCl5, the equation looks like this: PCl3 + Cl2 ⇌ PCl5. See that little double arrow? That’s the universal symbol for "we’re not sure what’s going to happen, but we’ll keep trying."
The Kc is calculated by taking the amounts of the stuff you make (the products) and dividing it by the amounts of the stuff you start with (the reactants). It’s like saying, "Okay, how much of the fancy new gadget did we produce, and how much of the raw materials did we use up?" So, for our reaction, it's basically [PCl5] divided by [PCl3] multiplied by [Cl2]. (Don't worry, the brackets just mean "the amount of").

My unpopular opinion? This whole "equilibrium constant" thing is just chemistry's way of acknowledging that sometimes, in life, you just can't get everything you want. You aim for perfect, but you often end up with "good enough." And you know what? That's okay!
A big Kc number means that when things finally settle down, you'll have a lot more PCl5 hanging around. The reaction really likes making PCl5. It’s like a baker who always makes way too many cookies because, well, who doesn't love cookies? The forward reaction is practically doing a victory lap.
A small Kc number, on the other hand, means that even after all the hustle and bustle, you'll still have a good chunk of PCl3 and Cl2 chilling in the reaction vessel. The reaction isn't super enthusiastic about churning out PCl5. It’s more like a reluctant teenager being told to clean their room – they’ll do it, but don't expect a spotless floor anytime soon.

So, why does this number matter? Well, it’s a chemist's crystal ball. It helps them predict what will happen when you mix these chemicals. If they know Kc is huge, they can be pretty confident they’ll end up with a lot of PCl5. If Kc is tiny, they know they’ll have a lot of leftover ingredients.
It’s like knowing whether your favorite restaurant is famous for its pizza (high Kc for pizza formation!) or its salads (low Kc for salad formation, meaning they’ll probably have a lot of lettuce left over). You can make your ordering decisions based on that.
And get this, Kc can change! It’s not etched in stone forever. If you mess with the temperature, you can actually nudge this equilibrium. Think of it like turning up the music at that party. Some people might start dancing more wildly (forward reaction speeds up), while others might decide it's too much and head home (reverse reaction might be affected). It’s a delicate balance, and temperature is like the DJ controlling the vibe.
It’s kind of funny, really. These molecules, PCl3 and Cl2, are just trying to find their happy medium. They react, they un-react, and eventually, they land on a state where things are just… steady. It’s not necessarily "happy" in the human sense, but it’s a stable state. A bit like how after a long day, you just want to flop on the couch and have things be quiet. The molecules just want their own version of couch-flopping.
So, next time you hear about the equilibrium constant Kc for the reaction PCl3 + Cl2 ⇌ PCl5, just picture that never-ending party. It’s a dynamic situation, always moving, but with that one magic number, Kc, giving you a hint about the overall mood. And maybe, just maybe, it’ll make you appreciate the subtle, complex, and sometimes perfectly imperfect ways the world works. After all, who needs perfect when you can have equilibrium?"
