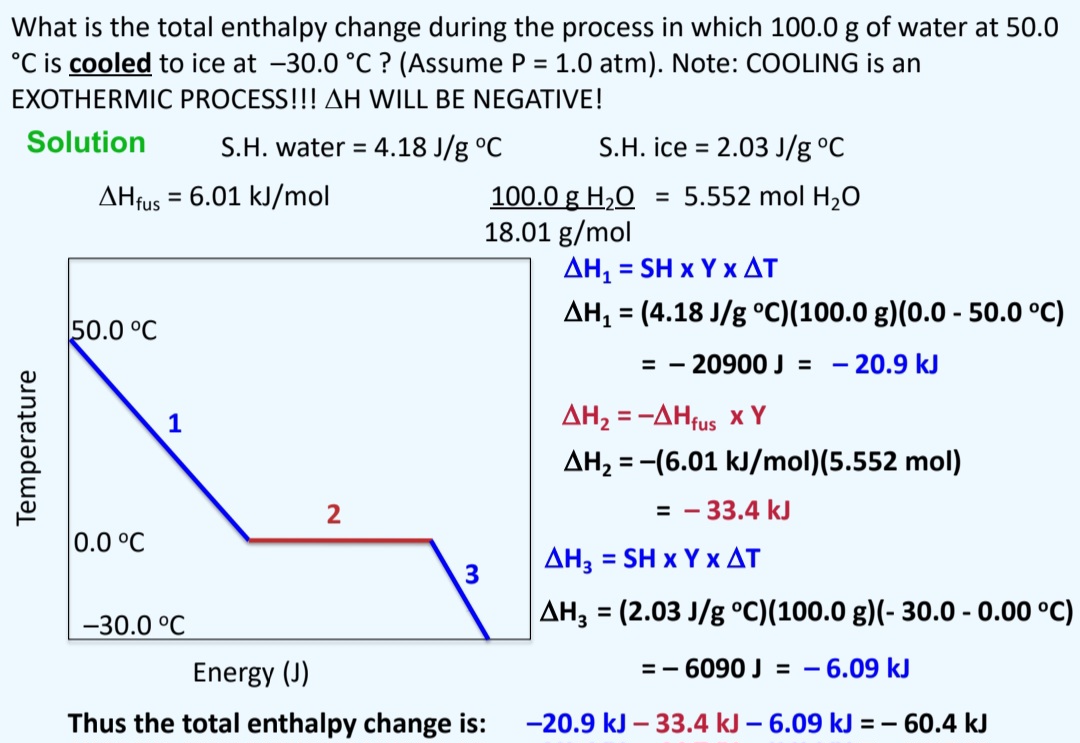
The Enthalpy Change For Converting 10.0 G Of Ice At
Okay, let's talk about something that might sound a little… intense. We're diving headfirst into the world of "The Enthalpy Change For Converting 10.0 G Of Ice." I know, I know, it sounds like the title of a particularly dry textbook chapter that makes you want to nap. But stick with me! Because this, my friends, is actually a secret gateway to understanding why we all get a little weird around frozen water.
Think about it. We've all been there. You're craving something cold, something refreshing. Maybe it's a nice, frosty glass of water. Or perhaps a delicious, brain-freezing popsicle. Whatever it is, it starts as ice. And that, my friends, is where the magic, or at least the science, begins.
Now, this whole "enthalpy change" thing? It's basically a fancy way of saying "how much energy it takes to change something's state." In our case, we're talking about changing the state of 10.0 grams of ice. That's not a whole lot, is it? It's like, what you'd find at the bottom of your soda glass after a long afternoon. Or the ice cubes in your favorite cocktail.
But even with just 10 grams, something happens. The ice, bless its little frozen heart, needs a little nudge to become that glorious liquid water we all know and love. It needs energy. And that's what the enthalpy change is all about. It's the energetic "oomph" required for the transformation.
Imagine the ice molecules are all huddled together, being all stiff and organized. They're like a bunch of introverts at a party, perfectly content in their little frozen clique. They don't want to mingle. They don't want to dance. They just want to stay exactly as they are. And that's fine, for a while.
But then, the party gets livelier. Someone turns up the music. Someone offers them a refreshing beverage (ironic, I know). They start to feel the warmth. They start to loosen up. They begin to wiggle. They begin to… melt. This melting process requires energy. It's like the ice molecules are saying, "Okay, fine. You've convinced me. Let's try this whole 'being liquid' thing."
And this is where our 10.0 grams of ice come into play. This specific amount, this tiny little chunk, needs a specific amount of energy to go from its rigid, frozen state to its free-flowing, watery state. It's not a huge demand, mind you. It's not like you're trying to power a small city with your ice cubes. But it's still a measurable, quantifiable thing.
It’s like the difference between a gentle nudge and a full-on shove. For ice to melt, it needs that gentle nudge of energy. It needs the enthalpy of fusion, which is the scientific term for the energy required to melt a substance. And for our humble 10.0 grams, this energy is… well, it's a number. A specific, calculable number.
And here's where I might get a little controversial. I think we don't give enough credit to the humble ice cube. We take it for granted. We toss it into our drinks without a second thought. But there's a whole energetic journey happening there! A mini-science experiment unfolding in real-time, right in our glasses. It's quite profound, if you think about it.
I mean, who knew that melting a few grams of frozen water could involve such fascinating scientific concepts? It’s like a tiny, everyday miracle happening before our eyes.
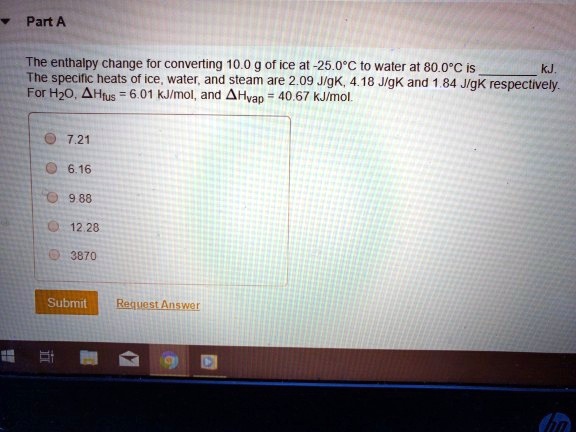
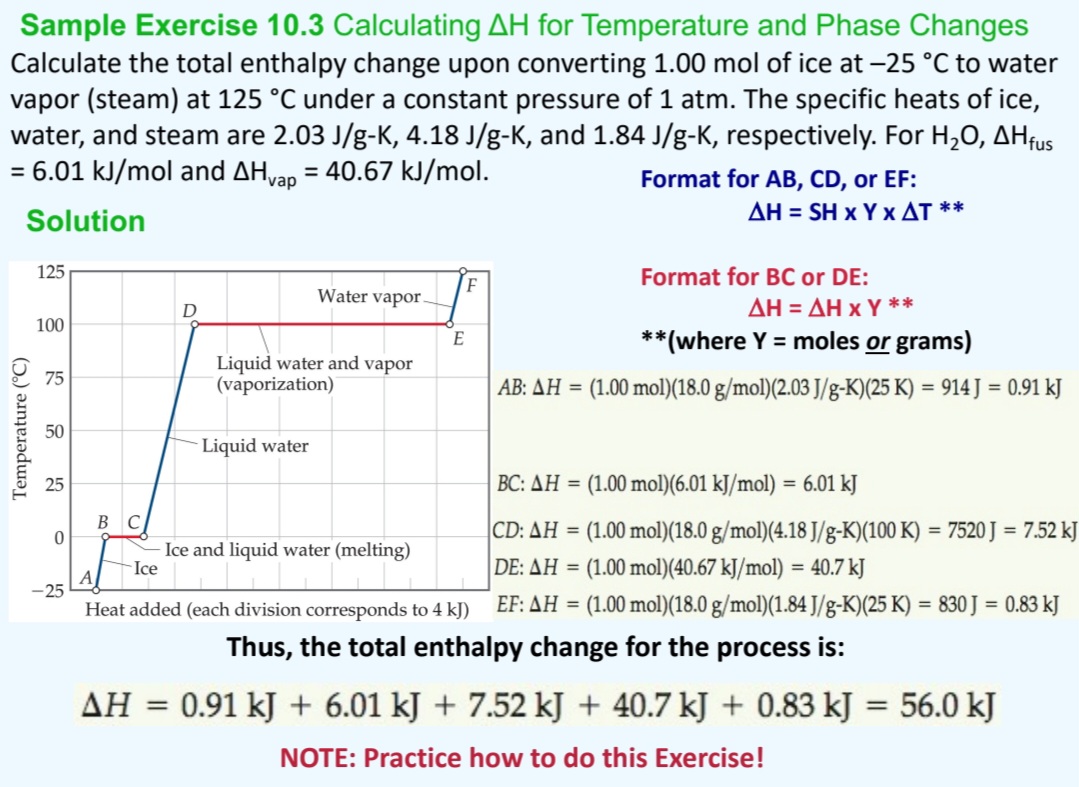
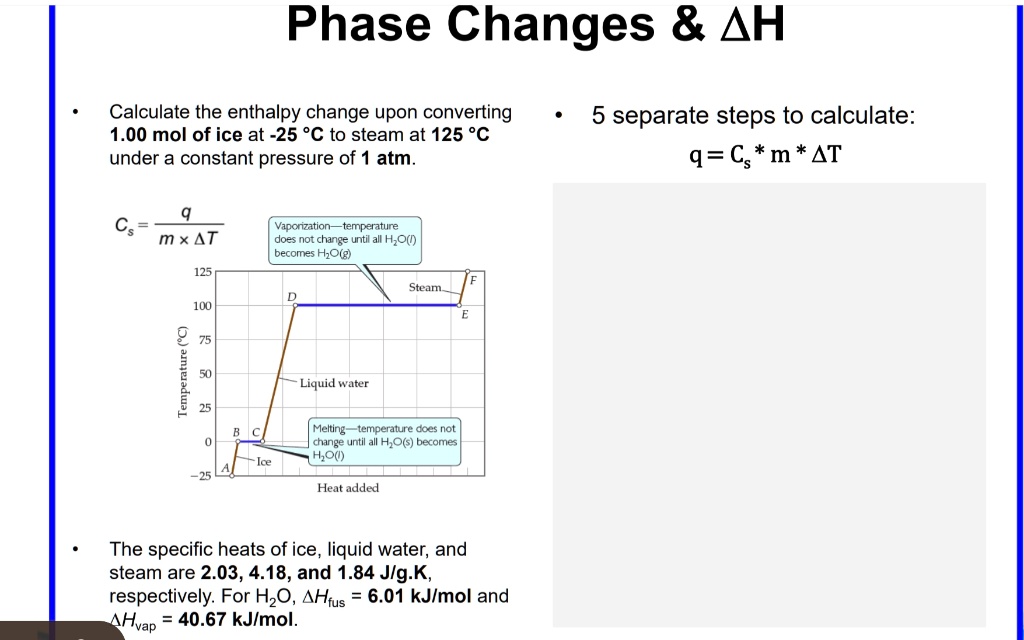
Answered: Sample Exercise 10.3 Calculating AH for Temperature and Phase
So, the next time you're enjoying an iced beverage, or watching ice melt on a hot day, take a moment. Appreciate the enthalpy change. Appreciate the energy that's being absorbed to turn that solid, stubborn ice into refreshing, flowing water. It’s a small detail, sure, but it’s the kind of small detail that makes the world, and our drinks, a lot more interesting.
And this whole process? It’s also a reminder that even the simplest things have a hidden complexity. That there's science at play in everything, from the vastness of space to the condensation on your cold drink. And for 10.0 grams of ice, that science is about a specific, measurable, and frankly, quite cool, energy change.
It’s not about the destination, the water, it’s about the journey. The journey from stiff, unyielding ice to a fluid, adaptable liquid. And that journey, that transformation, is powered by something we call enthalpy change. So, go forth, and ponder the power of melting ice. It’s more exciting than it sounds, I promise.

Think of it this way: that 10.0 grams of ice is like a shy guest at a party, needing a little encouragement to join the fun. The energy supplied is that encouragement. Without it, they'd just stand by the punch bowl, looking awkward. With it, they can become the life of the party, or at least, a pleasant liquid addition to your drink.
It's a concept that’s fundamental to chemistry, but also to our everyday experiences. We see it when we make ice cream, when we freeze leftovers, or even just when we leave a glass of water out in the sun. The enthalpy change for converting 10.0 g of ice is just a tiny snapshot of this universal phenomenon of phase transitions. It's a little piece of the puzzle that explains why things are the way they are, from the polar ice caps to the ice cubes in your gin and tonic.
And while I'm not suggesting you start calculating enthalpy changes for every ice cube you encounter, it's nice to know that there's a scientific reason behind the simple act of melting. It adds a layer of appreciation, a touch of wonder, to the mundane. So, the next time you see ice, give it a nod. It's been through a lot to get to where it is.