The Elements In Group 7a Are Known By What Name

Alright, gather ‘round, you lovely lot, and let me tell you about a little secret the periodic table has been keeping from us. You know, that big chart of all the building blocks of everything? It’s got its quirks, like a grumpy old uncle with a hidden soft spot. Today, we’re diving into one of its more… lively neighborhoods, specifically, Group 7A.
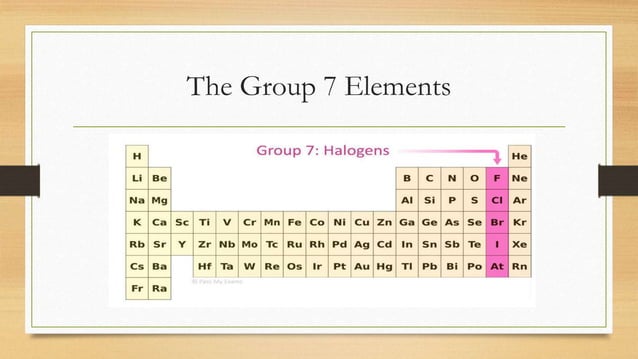
Now, if you’re picturing some dusty old room where element buddies hang out, you’re not entirely wrong, but these guys are more like the rock stars of the periodic table. And what do you call a group of rock stars? You call ‘em something cool, right? Something that makes you go, “Ooh, yeah!” Well, the elements in Group 7A have a name that’s just as catchy. They are known, my friends, as the Halogens!
Halogens. Doesn't that just sound a bit… mysterious? Like they’re about to blast off in a spaceship or perform some ancient ritual? Well, in a way, they kind of do! The name itself comes from Greek, meaning “salt-former.” And believe me, these guys are excellent at forming salts. Like, Olympic-level salt-formers. So good, in fact, that they’ve pretty much salted the earth (and your dinner table) forever.
Let’s break down who these salty celebrities are. We’ve got Fluorine, Chlorine, Bromine, Iodine, and Astatine. And then there’s Tennessine, which is so new and flashy it’s practically still in its paparazzi phase. Don’t worry, we’ll get to all of them, but let’s start with the heavy hitters, the ones you’ve probably bumped into, even if you didn’t realize it.
First up, we have Chlorine. Ah, Chlorine. The element that makes your swimming pool smell like a fancy spa (or, you know, a chlorinated puddle). It’s also a bit of a superhero in disguise, keeping our water safe and our kitchens smelling… well, clean. But don’t let its household fame fool you. Chlorine can be a real drama queen. In its pure form, it’s a greenish-yellow gas that’s, shall we say, unpleasant to inhale. Think of it as the element equivalent of that one friend who talks way too loud after a couple of drinks. You appreciate them, but maybe from a distance.
Then there’s Fluorine. Now, Fluorine is the undisputed heavyweight champion of the halogens, the one element that scares even the other halogens a little. It’s so reactive, it makes Chlorine look like a sleepy kitten. Seriously, Fluorine is like the energetic puppy that chewed through the sofa, the walls, and possibly your car keys before you even finished your morning coffee. It’s the most electronegative element on the periodic table, which is a fancy way of saying it’s really, really good at stealing electrons from anything it comes into contact with. So good, it’s basically a cosmic kleptomaniac.
This extreme reactivity is actually what makes it useful, though. It’s what gives your toothpaste its superpower to fight cavities. Yes, that little minty freshness in your mouth? It’s got a bit of a wild child, super-powerful halogen in it, working to keep your pearly whites intact. Imagine that! You’re brushing your teeth with a tiny, controlled explosion of atomic goodness. Pretty neat, huh?

The Shiny and the Slightly More Shadowy
Moving on, we encounter Bromine. Bromine is… interesting. It’s a liquid at room temperature, which is already a bit of a party trick. It’s a reddish-brown, volatile liquid that also has a rather pungent odor. So, if you ever smell something that makes you want to open all the windows and question your life choices, it might be Bromine paying a visit. Historically, it was used in things like flame retardants and photographic film. Think of it as the element that used to get all the cool, edgy jobs before the digital age.
Next up, the regal Iodine. Ah, Iodine. This one is probably the most familiar to you in a friendly way. It’s that purplish-brown liquid that your mom or grandma used to dab on scrapes and cuts, warning you, “This will sting!” And boy, did it sting! But that stinging was Iodine doing its job, acting as an antiseptic. It’s also essential for your thyroid gland to function properly. So, in a weird way, Iodine is the element that helps you… well, be you. Without enough Iodine, you might feel a bit sluggish, like a deflated balloon. It’s like the elemental equivalent of a good cup of coffee for your metabolism.
Now, things start to get a little more… exotic. We have Astatine. This one is the mysterious stranger at the party. It’s incredibly rare, so rare that you could spend your entire life and never see a pure chunk of it. It’s radioactive, and it decays really, really quickly. Scientists have estimated that at any given moment, there’s less than an ounce of Astatine on the entire planet. Less than an ounce! That makes it rarer than a politician’s promise of a tax cut. Because it’s so unstable and hard to get, its uses are still mostly theoretical, but imagine the bragging rights if you did manage to get your hands on some!
And finally, the brand new kid on the block: Tennessine. This is the element that’s so new, it’s practically still teething. It was first synthesized in 2010, and it’s highly radioactive and unstable. Like, really unstable. It probably exists for less time than it takes to blink. Scientists are still trying to figure out exactly what it is and what it does. It’s like the periodic table’s latest iPhone model – super high-tech, incredibly expensive to create, and nobody is entirely sure what it’s good for yet. It's a theoretical celebrity, a rumor in the scientific world.
Why Are They So Special (and a Little Scary)?
So, what’s the common thread that ties these seemingly diverse elements together? It’s their electron configuration, specifically, they all have seven valence electrons. Think of valence electrons as the outermost layer of an atom, the ones that get to mingle and mingle and mingle with other atoms. Having seven out of a possible eight spots filled means these guys are desperate to get that eighth electron. They’re like that person at a party who’s trying way too hard to complete a set of something.
This desperation makes them incredibly eager to react with other elements, especially those who have an extra electron to spare (like the Alkali Metals in Group 1A – but that’s a story for another day!). When a halogen grabs an electron, it becomes a negatively charged ion, and when it gives one away (though they’re not usually keen on that!), it becomes positively charged. This dance of electron exchange is what forms those ionic bonds, the glue that holds many compounds together.
And remember that salt-forming business? When a halogen reacts with a metal, especially an alkali metal, you get a salt! Table salt, for instance? That’s Sodium Chloride (Na + Cl). Sodium is the eager giver, and Chlorine is the delighted receiver. It’s a perfect match made in chemical heaven, or at least, on your dinner plate.
So, the next time you’re at the beach, or swimming in a pool, or even just brushing your teeth, take a moment to appreciate the Halogens. They’re the salty, reactive, sometimes a bit aggressive, but ultimately essential building blocks that make our world a little cleaner, a little healthier, and definitely a lot more interesting. They’re the rock stars of Group 7A, and they’ve earned their name!
