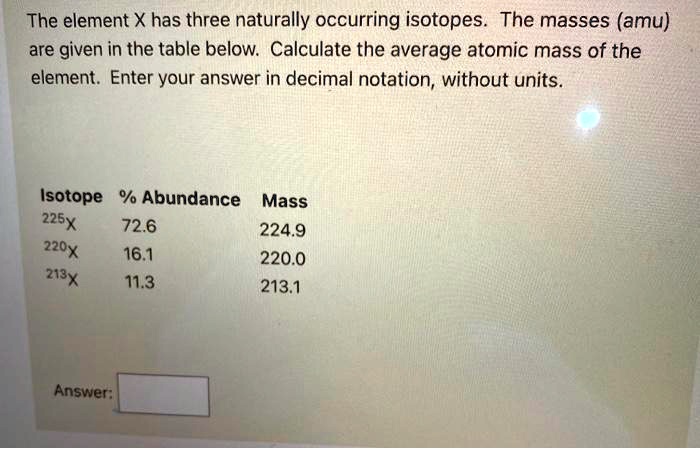
The Element X Has Three Naturally Occurring Isotopes
So, you ever think about, like, the building blocks of everything? Yeah, the periodic table and all that jazz? Well, imagine my surprise the other day when I stumbled upon this Element X. It’s a bit of a mystery, you know? Like, the cool, slightly baffling kind of mystery that makes you want to grab a cup of coffee and just talk about it.
And the coolest thing? Element X isn't just one thing. Nope! It’s like a whole family of atoms. Think of it like this: you’ve got your basic LEGO brick, right? But then, sometimes, you find one that’s just a tiny bit chunkier, or maybe a little bit lighter. That’s kind of what’s going on with Element X. It’s got three siblings, three
So, what exactly is an isotope? Don’t worry, I won’t bore you with a full-on chemistry lecture. We’re just chilling here, remember? Basically, all atoms of a specific element have the same number of protons. That’s their identity card, their fingerprint. But, and this is where it gets interesting, the number of neutrons can be different. And that’s what makes an isotope.
Think of protons as the essential personality of an atom. They define it. The neutrons? They're more like, well, the accessories. They add a bit of weight, a bit of character, but they don't change the fundamental "you." So, Element X has three different "accessory packages," if you will. How many different ways can you accessorize a LEGO brick, right?
We’re talking about Element X, isotope 1. Let’s call it X-A, just for fun. Then there's X-B, and finally, X-C. They’re all Element X, mind you. They’re all best friends, chemically speaking. They’ll react pretty much the same way. But their atomic weights? Oh, those can be a little bit different. It’s like having triplets, but one’s a little more muscular, one’s a little more lanky, and one’s just… average. Still the same person, though!
Now, why is this even a big deal? You might be thinking, "So what? They're all just X." And you're right, mostly. But those little differences in neutron count? They can have some pretty significant consequences. It’s like the butterfly effect, but with atoms. A tiny change can ripple outwards.
For instance, their masses are different. This might sound obvious, but it’s crucial for a lot of things. Imagine you're trying to sort marbles by weight. If you have a bunch of marbles that are almost the same weight, but not quite, it gets a bit tricky, doesn't it? Scientists have to deal with that when they're trying to figure out the exact composition of things.
And then there's stability. Sometimes, having too many or too few neutrons can make an atom a little… unstable. Like, it’s got extra energy it needs to get rid of. This is where radioactivity comes in. Some isotopes are perfectly happy and stable, just chilling out. Others? They're a bit more antsy, and they might decay over time, releasing all sorts of interesting (and sometimes dangerous!) particles and energy. It’s like that one friend who's always fidgeting because they drank way too much coffee.

So, when scientists talk about Element X, they're not just talking about one thing. They're talking about a mix. And the proportion of each isotope can be really important. It’s like if you were making a special blend of coffee. The ratio of Colombian to Ethiopian beans makes a huge difference to the final taste, right? Same with Element X. The relative abundance of X-A, X-B, and X-C matters.
This is where the concept of
Think about it. If you have 90% of Element X being, say, 10 grams, and 10% being 12 grams, your average isn't just (10+12)/2. It's going to be way closer to 10. This is exactly what happens with Element X’s isotopes. The universe, in its infinite wisdom (or maybe just chance!), has decided on specific percentages for each of these three siblings. And those percentages are pretty darn consistent across the board, which is pretty neat.
Why do they have these specific percentages? That’s a whole other can of worms, isn't it? It has to do with how the universe formed, with nuclear reactions in stars, and all sorts of mind-boggling stuff that happened billions of years ago. We’re talking about the cosmic recipe, and Element X just happens to have this particular mix of ingredients. Lucky us, or maybe not so lucky, depending on the isotope!
Now, let’s dive a little deeper into what makes these isotopes different on a practical level. We mentioned mass, but there are other subtle effects. For example,
Imagine you're trying to get two people to dance. If they have slightly different coordination, one might lead a fraction of a second faster than the other. It’s similar with isotopes. The heavier isotopes might be ever so slightly slower to participate in certain chemical reactions because it takes a little more energy to get those extra neutrons moving. It’s like trying to push a heavier shopping cart – it requires a little more oomph!

This can be super useful for scientists, though. If they can detect these subtle differences in reaction rates, they can actually use them to figure out the isotopic composition of a sample. It’s like being a detective, but instead of fingerprints, you’re looking for tiny variations in how quickly things react. Pretty cool detective work, if you ask me!
And what about
So, when scientists use spectroscopic techniques, they can often pick out the spectral signatures of each of Element X’s isotopes. This is another powerful tool for identifying and quantifying the different forms of Element X present in a sample. It's like having a special pair of glasses that can see the subtle differences that are invisible to the naked eye.
Now, let’s talk about abundance. How much of each isotope is out there? This is where the "naturally occurring" part really shines. It means we don't have to go out of our way (most of the time!) to create these different versions. They're just… there. In rocks, in water, in the air we breathe (if Element X is in the air, of course!).
The relative abundance of each isotope is usually expressed as a percentage. For example, you might find that X-A makes up, say, 75% of naturally occurring Element X. X-B might be around 20%, and X-C, the rarest of the bunch, could be just 5%. These percentages are crucial for calculating that average atomic weight we talked about. They are the weights in our weighted average!
And these abundances are remarkably consistent across different locations on Earth. It doesn't matter if you find Element X in your backyard or on Mount Everest (assuming it's found there!), the proportions of X-A, X-B, and X-C will be pretty much the same. This consistency is a testament to the fundamental laws of physics and chemistry that govern our universe. It's like a cosmic signature, a stamp of approval from the universe itself.

What happens if those abundances aren't consistent? Well, that can tell us something interesting too! If you find a sample of Element X where the isotopic ratios are different from the norm, it might indicate that the sample has undergone some unusual process. Maybe it’s been exposed to a lot of radiation, or it formed under very specific geological conditions. It’s like finding a perfectly ordinary-looking apple that tastes surprisingly spicy – something unusual has happened!
This is where things get really exciting for geologists and cosmochemists. They can analyze the isotopic composition of elements in rocks and meteorites to understand the history of our planet and the solar system. They can trace the origins of materials and reconstruct past events. It’s like reading a history book, but the letters are atoms and the stories are told by their isotopic ratios.
Let’s not forget the applications, because that’s where this whole isotope thing really shines. Knowing that Element X has three naturally occurring isotopes with different properties opens up a whole world of possibilities.
Think about
Or how about
Even in

So, this Element X, with its three distinct, naturally occurring isotopes, is more than just a curiosity. It’s a little window into the fundamental nature of matter. It shows us that even seemingly simple elements can have layers of complexity, and that these variations can have profound implications for our understanding of the universe and for our technological advancements.
It’s the kind of thing that makes you look at a plain old rock and wonder about the incredible atomic ballet happening within it. It makes you appreciate the subtle differences that make up the grand tapestry of reality. And it definitely makes you want another cup of coffee to ponder it all a little more!
So next time you hear about an element, especially one with multiple isotopes, take a moment to appreciate the nuance. It’s not just one entity; it’s a family, each member with its own subtle story to tell. And Element X, with its trio of siblings, is a fantastic example of this atomic diversity. Pretty neat, right?
It’s like a subtle reminder that even in the seemingly uniform world of elements, there’s always something more to discover, something to explore. And that, my friends, is why chemistry is just so darn fascinating. Who knew atoms could have so many siblings!
And the fact that these isotopes are naturally occurring? That’s the icing on the cake. It means the universe has already done the hard work of creating these variations for us. We just have to observe them, study them, and figure out all the cool things they can do. It’s a gift, really. A rather atomic gift.
So, there you have it. Element X and its three naturally occurring isotopes. A little bit of science, a lot of wonder, and a good reason to keep asking questions. Now, about that refill…
