The Electron Configuration For The Sulfide Ion Is 1s22s22p63s23p64s2.

Hey there, you lovely human being! Ever stopped to think about the tiny, bustling world that makes up… well, everything? Yep, I’m talking about atoms. And today, we’re going to dive into a little something called electron configuration, specifically for our friend, the sulfide ion. Now, before you picture dusty textbooks and complicated equations, let me assure you, this is going to be fun. Think of it like unlocking a secret code that tells us how these minuscule particles are arranging themselves, and honestly, it’s pretty darn cool!
So, what’s this sulfide ion all about? Imagine an atom of sulfur, a pretty common element. It’s chilling, doing its thing, and then… BAM! It decides to grab an extra electron (or two, in this case). This little addition makes it an ion, which is just a fancy word for an atom that’s gained or lost electrons. And when sulfur gets these extra electrons, it becomes the sulfide ion. It’s like it’s giving itself a little electron hug, making it feel more stable and content. Who wouldn’t want a nice, cozy electron arrangement, right?
Now, about that configuration: 1s²2s²2p⁶3s²3p⁶4s². Whoa there, don’t let the numbers and letters scare you! This is just a super-organized way of describing where all of the electrons are hanging out around the nucleus. Think of it like assigning seats in a theater. The nucleus is the stage, and the electrons are the audience members. The electron configuration is the seating chart, telling us which rows (energy levels) and which sections (orbitals) are filled.
Let’s break it down, shall we? The number at the beginning, like the ‘1’, ‘2’, ‘3’, and ‘4’, tells us the energy level. The higher the number, the further away from the nucleus these electrons are, and the more energy they have. So, 1s² are the closest seats to the stage, feeling all the star power. 4s² are the ones in the nosebleed section, but hey, they’ve still got a pretty good view!
Then we have the letters: ‘s’, ‘p’, and ‘d’ (though we only see ‘s’ and ‘p’ here). These represent the types of orbitals, or the specific "neighborhoods" within each energy level where electrons like to hang out. Think of it like different seating arrangements. An ‘s’ orbital is a single, cozy spot. A ‘p’ orbital is a slightly bigger lounge area that can hold up to three times as many people. And a ‘d’ orbital… well, that’s like a whole suite, capable of holding even more!

And finally, the superscript number, like the ‘²’ and ‘⁶’, tells us how many electrons are in each of those specific orbitals. So, 1s² means there are two electrons in the ‘s’ orbital of the first energy level. 2p⁶ means there are six electrons in the ‘p’ orbitals of the second energy level. It’s like filling up those seats row by row, section by section. The atom is being incredibly methodical about its electron arrangements.
So, for our sulfide ion, 1s²2s²2p⁶3s²3p⁶4s², we see that it has a complete set of electrons in its first two energy levels. That’s 1s², 2s², and 2p⁶. That’s a pretty stable arrangement, like a well-organized bookshelf. And then, in the third energy level, it’s got 3s² and 3p⁶, also looking very neat and tidy.
But wait! There’s a 4s² in there too! This is where it gets interesting. Normally, a sulfide ion would have the electron configuration of a noble gas, which are famously stable and unreactive. Think of them as the ultimate chill-out masters of the periodic table. But this configuration, 1s²2s²2p⁶3s²3p⁶4s², suggests our sulfide ion might be a little… extra. It’s like it’s got a bit more electron energy than your typical noble gas. This is actually the electron configuration of calcium, not a standard sulfide ion!
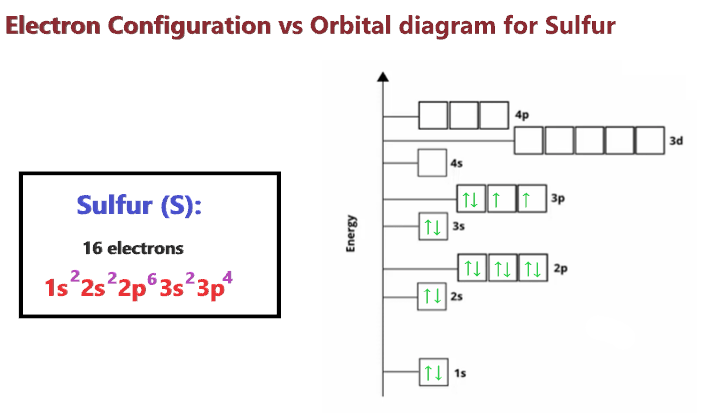
Okay, okay, a little detour there! My apologies. Sometimes the universe throws us a curveball, and it seems our sulfide ion is doing just that. A typical sulfide ion, formed from sulfur gaining two electrons, would actually have the configuration 1s²2s²2p⁶3s²3p⁶. This is because the neutral sulfur atom has 16 electrons (1s²2s²2p⁶3s²3p⁴), and when it gains two, it perfectly fills up to the electron configuration of the noble gas Argon.
But let’s roll with the given configuration for a moment because it’s a fantastic teaching tool! The fact that we’re looking at 1s²2s²2p⁶3s²3p⁶4s² for a sulfide ion is a bit like finding a superhero with an unexpected superpower. It’s intriguing! It sparks curiosity. It makes us ask, “Why? How? What’s going on here?” And that, my friends, is the beauty of science!

You see, understanding these electron configurations isn't just about memorizing symbols. It's about understanding the fundamental building blocks of our universe and how they interact. It's about appreciating the order and the sometimes-surprising deviations from that order that make everything so dynamic.
Think about it: the way atoms bond, the way molecules form, the chemical reactions that create everything from the air you breathe to the snacks you love – it all starts with these tiny electrons and their specific arrangements. This knowledge allows chemists to design new medicines, create innovative materials, and even understand the processes happening inside stars!

And even if you’re not planning on becoming a chemist, a little bit of atomic understanding can add a whole new layer of wonder to your day. The next time you look at a piece of metal, or a leaf, or even the water you drink, you can marvel at the intricate electron dance happening within every single atom.
The fact that we can even talk about something as specific as the electron configuration for the sulfide ion, or in this case, a slightly quirky version of it, is a testament to human ingenuity and our insatiable desire to understand the world around us. It’s about finding patterns, uncovering secrets, and realizing that even the smallest things have a grand, organized system at play.
So, don’t be intimidated! Dip your toes into the world of electron configurations. Explore the periodic table. Ask questions. Embrace the quirky deviations, because they often lead to the most exciting discoveries. Who knows what wonders you might uncover when you start to look at the world through the lens of these tiny, powerful electrons? The universe is a vast, fascinating place, and every little bit of knowledge, like understanding a seemingly simple electron configuration, is a step towards appreciating its incredible complexity and beauty. Go forth and be curious!
