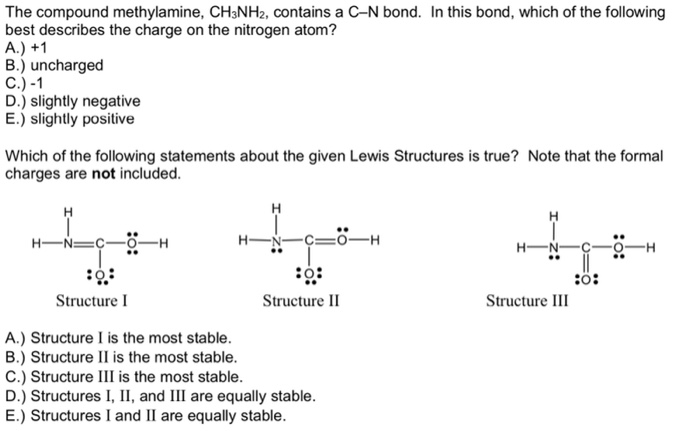
The Compound Methylamine Ch3nh2 Contains A C-n Bond

Hey there! Grab your coffee, settle in. We're gonna chat about something super small, but honestly, kind of a big deal. You know those molecules chemists are always talking about? Well, today, we're diving into one that’s got a pretty neat trick up its sleeve. It’s called methylamine. Fancy name, right? But stick with me, it’s actually pretty cool. Think of it like a tiny building block for… well, a lot of stuff. Like, really a lot of stuff. And the star of the show, the little secret handshake that makes it all work, is this thing called a C-N bond. Yeah, I know, sounds like something you’d find in a textbook. But let’s break it down, shall we?
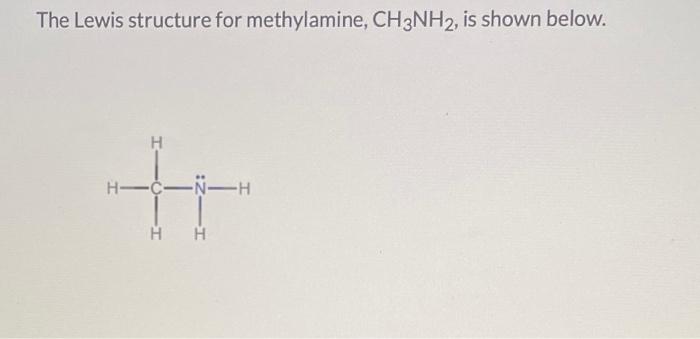
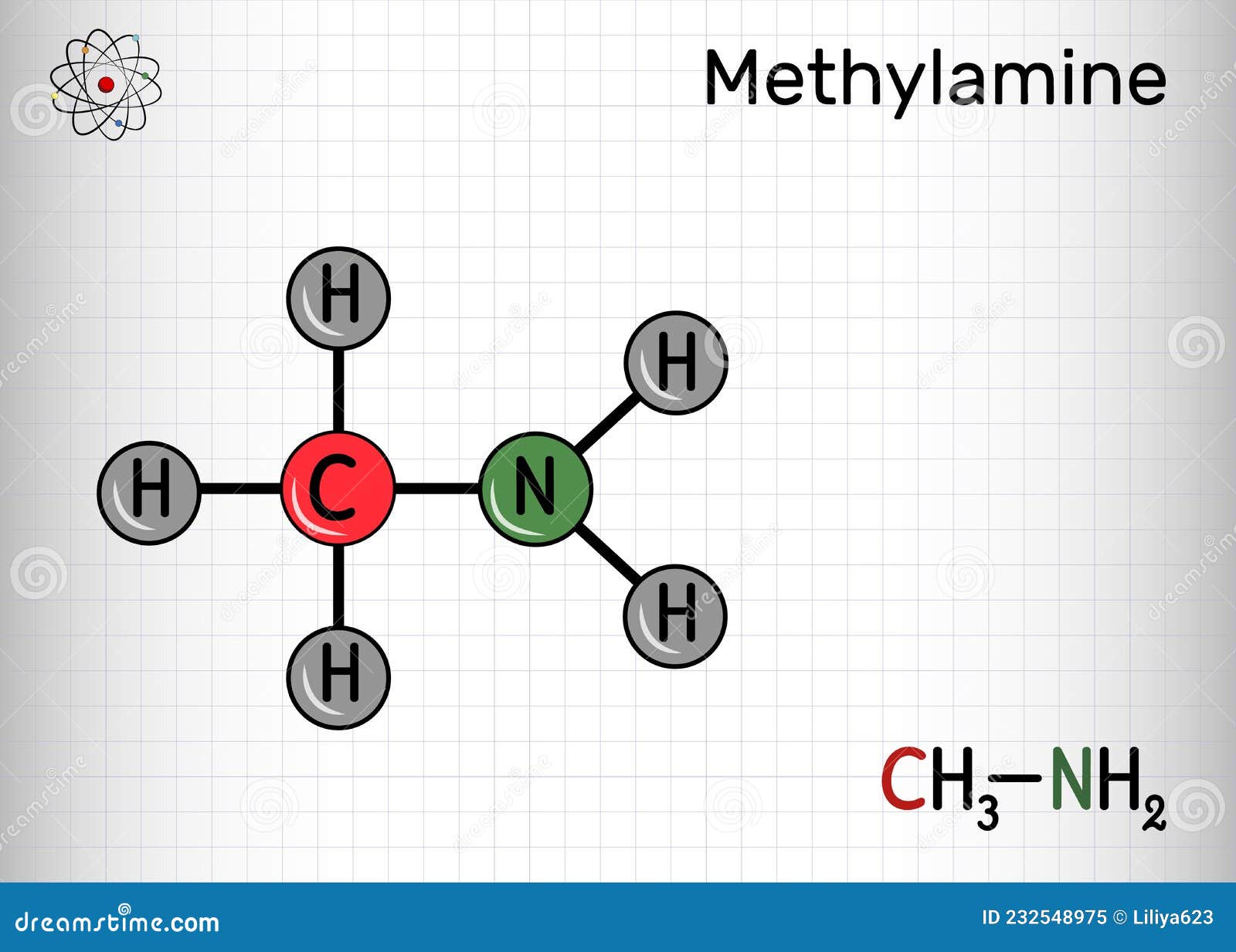
So, methylamine. Its chemical formula is CH3NH2. See that? C for carbon, H for hydrogen, N for nitrogen. It’s like a miniature family reunion happening at the atomic level. And the most exciting part, the absolute raison d'être if you will, is that carbon atom (the C) and that nitrogen atom (the N) are holding hands. They're bonded. They’re a team. They’re basically BFFs in the molecular world. It’s this connection, this carbon-nitrogen bond, that gives methylamine all its personality. Without it, well, it’d just be a sad pile of separate atoms, wouldn't it? And who wants that?
Imagine it like this: you’ve got a carbon atom, right? It’s kind of a social butterfly, loves to bond with things. Then you’ve got a nitrogen atom, which is also pretty keen on making connections. When these two get together, and they decide to really commit, they form this C-N bond. It’s not just a casual hello; it's a firm handshake. And in methylamine, this is happening with a carbon that’s already got three hydrogens attached to it, making it a methyl group (that’s the CH3 part). And that nitrogen? It’s got two hydrogens hanging out with it (that’s the NH2 part). So, you’ve got the CH3 crew, and the NH2 crew, and they’re united by this glorious C-N bond. It’s like a miniature, super-stable social club. The best kind of club, obviously.
Now, why is this C-N bond such a big deal? I mean, atoms bond all the time, right? True, but the carbon-nitrogen bond is a bit special. It’s got this… oomph to it. It’s strong enough to keep the molecule together, but flexible enough to participate in all sorts of cool chemical reactions. Think of it as the chassis of a really versatile car. You can soup it up, paint it a new color, add spoilers – it’s still the same basic car, but it can do so much more. That C-N bond is like the foundation that allows for all these chemical adventures.
And where does this methylamine, with its trusty C-N bond, pop up? Oh, you’d be surprised! It’s everywhere. Well, maybe not everywhere like air, but it’s a foundational molecule for so many important things. Ever heard of amino acids? Those are the building blocks of proteins, which are, like, essential for literally everything your body does. Think muscles, enzymes, your very existence. And guess what? Many amino acids have a C-N bond, often within an amino group. So, indirectly, that little methylamine molecule is a distant cousin to your biceps. How cool is that? You’re basically made of molecules that are related to methylamine. Mind. Blown.
But it’s not just about us, fleshy humans. Methylamine is also a key player in making pesticides. Yeah, I know, not the most glamorous application. But think about it: keeping crops healthy so we can eat them. It’s a tough job, but someone’s gotta do it, right? And methylamine, thanks to its versatile C-N bond, is good at forming those complex structures needed for pest control. It’s like the unsung hero in the fight against tiny, crop-munching villains. We’ll take it.
What else? Oh, pharmaceuticals! That’s a big one. Many medicines, the ones that make you feel better when you’re sick, have a C-N bond somewhere in their structure. It’s a common feature in drug molecules, and methylamine is a simple starting point for building these complex therapeutic agents. So, next time you pop a pill, remember that somewhere in its ancestry, there might be a little methylamine. It’s like a molecule with a healing touch, all thanks to that humble C-N bond. Who knew chemistry could be so medicinal?
And let’s not forget about things you might find in your everyday life. Dyes, for example. Those vibrant colors on your clothes? Many of them involve molecules with C-N bonds. It’s the nitrogen atom, often in a group like an amine, that helps the molecule absorb and reflect light in just the right way to create those amazing colors. So, that bright red t-shirt you love? It owes a little something to the chemistry that methylamine represents. It's like a tiny molecular artist, painting the world with its C-N bond.
So, we’ve established that methylamine, with its signature C-N bond, is a pretty important little molecule. But let’s get a little more into the why. What makes this bond so special? Well, carbon and nitrogen are both in the same row of the periodic table, which is neat, but they have different electronegativities. Ever heard of that? It’s basically how much an atom wants to hog electrons. Nitrogen is a bit greedier than carbon. It’s like a slightly more dominant personality in the relationship.
Because nitrogen pulls the shared electrons in the C-N bond a little closer to itself, it creates a slight partial negative charge on the nitrogen and a corresponding partial positive charge on the carbon. This is called a polar bond. And this polarity? It’s like a superpower for chemists! It means that the molecule can interact with other molecules in specific ways. It can be attracted to positive things, or repel negative things. It’s this subtle electrical imbalance that makes the molecule reactive and allows it to participate in all those fascinating reactions we talked about.
Think of it like a tiny magnet. Not a strong magnet, mind you, but a subtle one. The carbon end has a tiny bit of a positive “pull,” and the nitrogen end has a tiny bit of a negative “push.” This allows it to be attracted to other charged molecules or to participate in reactions where electrons need to be transferred or shared in specific ways. It’s all about those little tugs and pulls of electrons, orchestrated by the C-N bond. It’s the silent conductor of a chemical orchestra.

And this polarity is also key to methylamine’s ability to act as a base. Bases are molecules that can accept protons (which are just H+ ions). The nitrogen atom in methylamine, with its extra electron density thanks to the polar C-N bond, is really good at picking up a proton. It’s like it’s saying, “Come here, little proton, I’ve got room for you!” This basicity is a fundamental property that makes methylamine useful in so many different chemical processes. It’s the welcoming committee for protons, all thanks to that well-positioned nitrogen atom and its bond with carbon.
So, we’ve got the structure, we’ve got the polarity, we’ve got the basicity. What else makes this C-N bond in methylamine so darn important? It’s about its stability and its reactivity. While it’s strong enough to hold the molecule together, it’s not so strong that it can’t be broken or modified. This sweet spot of stability and reactivity is what makes it such a versatile building block. Chemists can manipulate this bond to create entirely new molecules, to build up more complex structures, or to break down larger molecules. It’s like having a molecular Lego brick that’s sturdy but also easy to connect and disconnect.
And this ability to form and break bonds is how we get those complex molecules for drugs, dyes, and all sorts of fancy chemicals. It’s a dance of electrons, a precise choreography where the C-N bond plays a starring role. Without this specific type of connection, a lot of modern chemistry would simply grind to a halt. We’d be back to drawing board, wouldn’t we? Imagine a world without colorful clothes or effective medicines. It’s a bleak thought, I tell you.
Methylamine itself is a gas at room temperature, which is kind of wild to think about. This little molecule, zipping around as a gas, is the ancestor of so many solid, tangible things. It’s like the invisible force that shapes the visible world. And its primary identity, its very essence, is tied to that C-N bond. It's the anchor, the defining characteristic. It’s what makes a methylamine molecule methylamine, and not, say, a methane molecule or an ammonia molecule, though they’re related!
Think about the sheer diversity of life. All those different creatures, plants, fungi – they all rely on complex organic molecules. And a huge chunk of those molecules contain C-N bonds. From the DNA in your cells (yes, DNA has nitrogen!) to the pigments that give flowers their colors, this fundamental bond is everywhere. It’s a testament to how simple building blocks, when arranged in clever ways, can lead to incredible complexity and variety. Methylamine, with its relatively simple C-N bond, is a prime example of this principle in action.
So, the next time you hear the word “methylamine,” or even just think about the vastness of organic chemistry, take a moment to appreciate that little C-N bond. It’s the unsung hero, the silent workhorse, the molecular glue that holds so much of our world together. It’s a simple connection, just one carbon and one nitrogen, but its impact is truly colossal. It’s proof that even the smallest things can have the biggest consequences. And that, my friends, is something to ponder over your next cup of coffee. Cheers!
