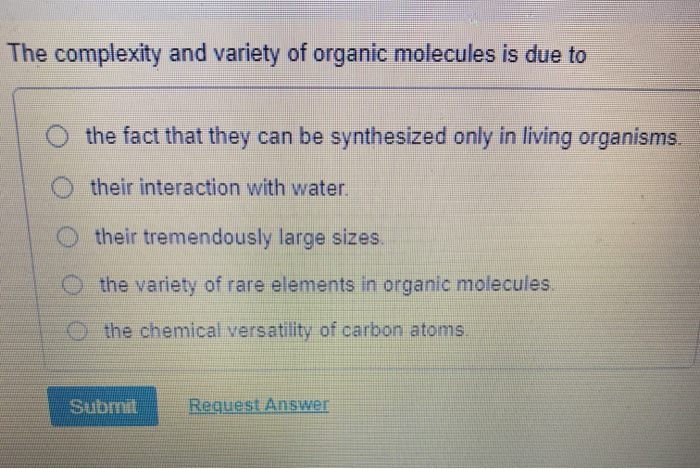
The Complexity And Variety Of Organic Molecules Is Due To
Ever wondered why life is just so… diverse? I mean, think about it. We’ve got tiny, invisible bacteria doing their thing, giant, majestic whales cruising the oceans, and us humans, with our intricate thoughts and weird cravings for pizza. What’s the secret sauce behind all this amazing variety? Turns out, it all boils down to some incredibly clever little building blocks called organic molecules.
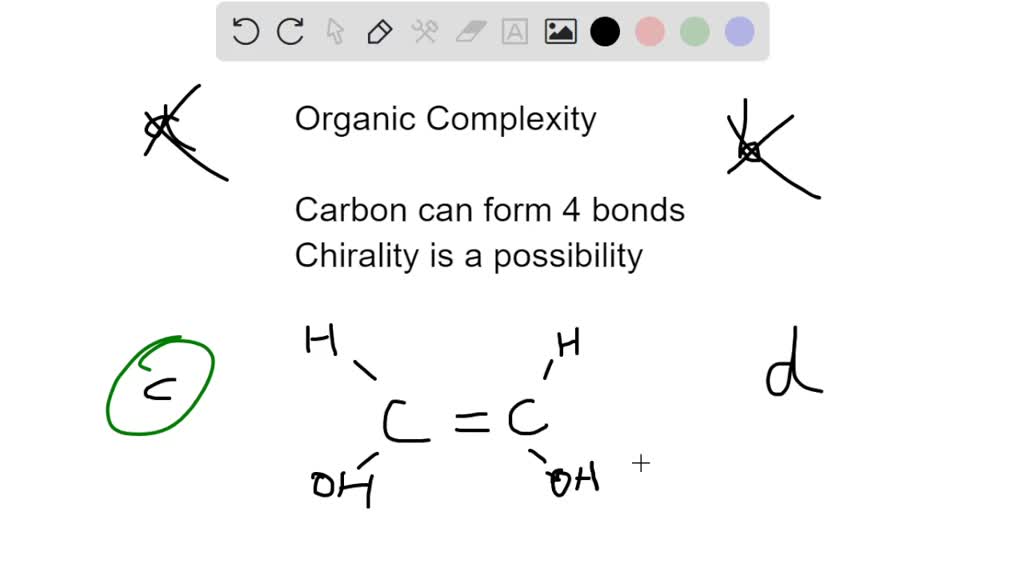
When you hear "organic," you might think of your fancy farmers market produce. And while that's a good start, in the world of chemistry, "organic" means something a bit more specific and, frankly, a lot more fundamental. It basically means molecules that have carbon at their heart.
So, why carbon? What’s so special about this particular element? Well, imagine you’re building with LEGOs. You’ve got all sorts of shapes and sizes, right? Some are basic bricks, some are special connectors. Carbon is like the ultimate LEGO brick. It’s got this amazing ability to link up with itself, forming long chains, intricate rings, and all sorts of wacky structures. It's like the universe decided to give us a molecular toolkit, and carbon is the star player.
The Carbon Connection: It’s All About Bonds
The magic really happens because of how carbon atoms bond with each other and with other elements. Carbon loves to form covalent bonds. Think of these like strong, stable partnerships where atoms share their electrons. And carbon can form up to four of these partnerships at once!
This is huge! It means a single carbon atom can reach out and grab hold of four other atoms. These could be more carbon atoms, or they could be other common elements like hydrogen, oxygen, nitrogen, sulfur, and phosphorus. And these partnerships aren't just one-offs; they can be single, double, or even triple bonds, which changes the whole personality of the molecule.
So, a simple carbon chain might look like a string of pearls. But then, you can bend that string, loop it into a circle, or even branch it off like a tiny tree. Each of these arrangements, even with the same number of atoms, leads to a different molecule with different properties. It’s like having a few basic LEGO bricks and being able to build a car, a house, or a spaceship, just by rearranging them.
Building Blocks for Life
Now, let’s talk about the real stars of the organic molecule show: the macromolecules. These are the big, complex molecules that are absolutely essential for life as we know it. Think of them as the super-powered LEGO structures.
There are four main types, and they’re like the four pillars holding up the entire edifice of biology:
Carbohydrates: The Energy Providers
These are your sugars and starches. Think of them as the fuel for your car. Simple ones, like glucose, are like quick energy bursts – that sugar rush you get from candy! More complex ones, like starch in potatoes or glycogen in our muscles, are like longer-term energy storage. They’re built from repeating units of sugars, linked together in chains or branches. It’s pretty neat how a few simple sugar units can be assembled into something that powers everything from a jog to your brain’s complex thoughts.

Lipids: The Versatile Builders and Stowers
These are your fats, oils, and waxes. They’re a bit more diverse in their structure. Some are long, straight chains, while others are more complex rings. Lipids are amazing because they’re great at storing energy for the long haul – way more than carbs. They also form the cell membranes that act as the protective barriers around every single cell in your body. Imagine them as the packaging material for all your cellular goods, keeping them safe and organized.
Plus, they’re involved in signaling and even act as insulators, keeping you warm. It’s a real do-it-all kind of molecule!
Proteins: The Workhorses of the Cell
These are arguably the most versatile. Proteins are made from long chains of smaller units called amino acids, linked together in a very specific order. Think of amino acids as different colored beads on a string. The order of these beads determines the final shape of the protein, and its shape dictates its function. And oh boy, do they have functions!
Proteins are your enzymes that speed up chemical reactions (like digesting that pizza). They’re your structural components, like collagen in your skin and bones. They’re your transporters, moving molecules around. They’re your antibodies, fighting off invaders. They’re essentially the tiny machines that make life happen. The sheer variety of shapes and functions these molecules can take on is mind-boggling.
Nucleic Acids: The Blueprint Holders
And finally, we have DNA and RNA. These are the molecules that carry your genetic information. They’re like the instruction manuals for building and operating every living thing. Their structure is a beautiful double helix (for DNA), made of repeating units called nucleotides. Each nucleotide has a sugar, a phosphate group, and one of four different bases. It’s the sequence of these bases that spells out your genes, telling your cells what proteins to make and how to do it.
It’s pretty incredible to think that all the amazing diversity of life, from the smallest amoeba to the largest redwood tree, is written in the language of these four types of organic molecules, all stemming from the unique properties of carbon.

The Infinite Potential of Carbon
So, when we talk about the complexity and variety of organic molecules, it’s all thanks to carbon’s unique talent for connecting with itself and other elements in an almost infinite number of ways. It’s like having a single alphabet that can be used to write every story imaginable, from epic poems to simple nursery rhymes.
This ability to form long, stable chains and rings, and to create branches and complex three-dimensional structures, is what allows for the immense diversity we see in nature. Each different arrangement, even with the same basic atoms, can lead to a molecule with a completely different job. It’s the ultimate form of molecular origami, constantly folding and unfolding into new and vital shapes.
Think about it: you can have a simple molecule like methane (CH4), which is just one carbon and four hydrogens – a basic building block. Then, you can have incredibly complex molecules like proteins or DNA, which have thousands, even millions, of atoms arranged in specific, intricate patterns. All of this complexity and variety arises from that one, simple fact: carbon is a molecular rockstar.
It’s this inherent flexibility and adaptability of carbon-based chemistry that has allowed life to evolve into so many different forms, filling every conceivable niche on our planet. The next time you look around at the amazing world of living things, remember that it’s all built upon the incredible versatility of these fundamental organic molecules. Pretty cool, right?
