The Complete Electron Configuration Of Gallium Element 31 Is

Imagine a universe where tiny little things zip and zoom around like hyperactive toddlers at a birthday party. That's kind of what's happening inside every atom, and today, we're going to peek into the energetic playground of a fascinating element called Gallium. It's element number 31, which means it has a certain number of these speedy little charges, called electrons, all tucked away in their own special rooms.
Think of an atom like a cozy little apartment building. The electrons are the tenants, and they don't just cram into any old room. Nope, they have very specific preferences and follow a strict seating chart, almost like a VIP list at a popular club. This chart, or what scientists call its electron configuration, tells us exactly where each electron likes to hang out.
For Gallium, with its 31 electrons, it's like organizing a bustling dorm with 31 students. They have different types of rooms, too. There are the cozy single rooms, the slightly larger double rooms, and even some common areas for group activities. It's all about finding the most comfortable and energetic spot for each little electron.
The Grand Tour of Gallium's Electron Apartments
Let's start with the ground floor, the most basic and energy-efficient rooms. These are called the 's' orbitals, and they're like the studio apartments of the atom. They can only hold a maximum of two electrons. Gallium fills up its first 's' orbital, which we can write as 1s2. That's two electrons happily settled in the very first room, right by the entrance.
Then we move up to the next floor, where we find another 's' orbital. This one is a bit further from the nucleus (the central hub of the atom), so it has a little more energy. This is the 2s2, another two electrons finding their comfortable space. They're like the next set of tenants moving into the slightly bigger studio on the second floor.

But wait, there's more! On this second floor, there are also some 'p' orbitals. Think of these as the slightly larger apartments that can hold up to six tenants each. They're arranged in a way that's a bit more spread out. Gallium fills up all three of these 'p' orbitals, so we get 2p6. That's a total of six energetic electrons finding their place in these 'p' rooms.
Now we're venturing onto the third floor, and things are getting even more interesting. We've got another 's' orbital here, the 3s2, once again housing two electrons. These are like the main tenants on the third floor, nicely settled in their own space.
And just like the second floor, the third floor also has those 'p' orbitals, ready to welcome more residents. So, Gallium adds another 3p6 to its configuration. That's another six electrons joining the party, filling up those 'p' rooms on the third floor. It’s starting to feel like a full building!
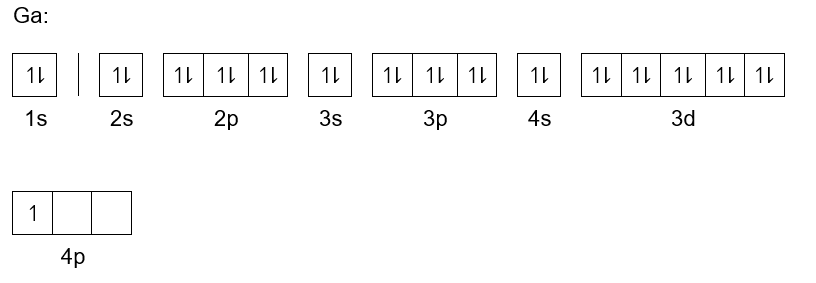
But here’s where things get a little more complex, and a tad bit funny. Before we get to the really spacious apartments, there's a set of rooms called the 'd' orbitals. These are like the fancy suites with extra space and a bit more privacy. They can hold up to 10 electrons. Gallium fills up all of these 'd' orbitals completely, giving us 3d10. Imagine ten students comfortably sharing a suite – they’ve probably got their own study groups and gaming consoles set up.
We’ve accounted for quite a few electrons now, haven’t we? We’re building up to our total of 31. The building is getting full, but there are still a few more rooms to consider. These are the highest energy levels, the penthouse suites, if you will.
On the fourth floor, we have another 's' orbital, and it gets two more electrons: 4s2. These are the last tenants to grab the most energy-efficient spots on this floor. They're probably enjoying the views from their higher vantage point.

Now, here's the grand finale for Gallium! After all those filled rooms and suites, we have the 'p' orbitals on the fourth floor. These are the most energetic of the 'p' rooms. And guess what? Gallium only needs to fill some of these, not all of them. It places three more electrons here, making it 4p3.
Putting It All Together: Gallium's Address Book
So, when we put all these cozy homes and bustling suites together, the complete electron configuration of Gallium looks like this: 1s22s22p63s23p63d104s24p3. It’s like the ultimate address book for every single electron in a Gallium atom, telling them exactly where to be.
Isn’t it amazing how these tiny particles have such a structured life? They’re not just randomly bouncing around; they’re meticulously organized. It’s like a perfectly choreographed dance that happens on an unimaginably small scale.

And the fun part? This configuration dictates how Gallium behaves! The electrons in the outermost shells, especially those in the 4p3, are the ones that get to interact with other atoms. They’re the ones who decide if Gallium wants to make friends and form new substances.
Think of it as the electrons in the penthouse having the most exciting social life, being the first to meet new neighbors. They’re the ones who get to decide who Gallium dances with in the grand ballroom of chemistry.
So, the next time you hear about Gallium, remember its well-organized electron family. It’s not just a bunch of numbers and letters; it’s a story of tiny energetic beings finding their perfect spots in the atomic universe, all thanks to its complete electron configuration. It's a little bit of order in a world that can seem chaotic, a heartwarming tale of structure and belonging for element number 31.
