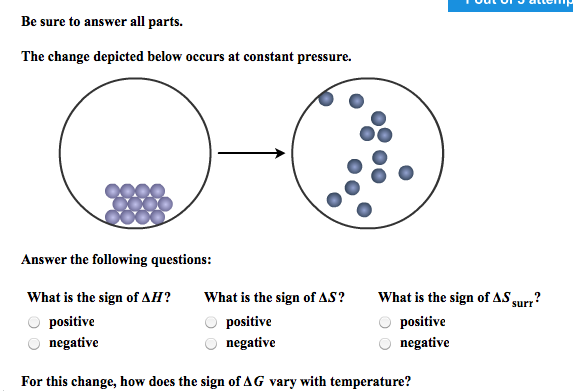
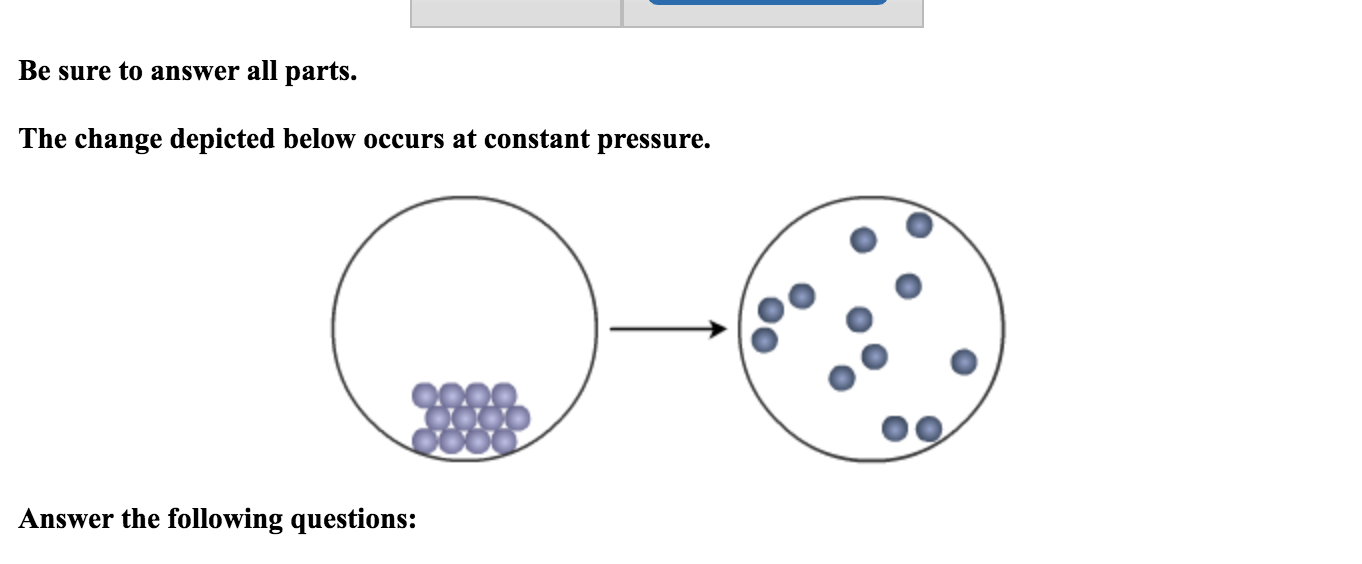
The Change Depicted Below Occurs At Constant Pressure.

Okay, so imagine this. You're chilling, right? Just vibing. And then, BAM! Something changes. But here's the super cool part: the pressure is staying exactly the same. Like, zero drama on the pressure front. This is what we're diving into today!
Think of it like this: you're at a party. The music's loud, people are chatting, the energy is chefs kiss. But the DJ is keeping the volume knob glued in one spot. No sudden blasts, no awkward silences. Just good, consistent vibes. That's our constant pressure.
So, what kind of magical changes can happen when pressure is just chilling out? Let's spill the tea!
Water Wonders!
You know water, right? Our favorite liquid. Well, when you keep the pressure steady, you can do some seriously neat stuff with it. Like, heating it up.
When you heat water at constant pressure, things get lively. First, it gets hotter. Duh. But then, it starts to do this cool little dance. You might see little bubbles forming. These aren't just any old bubbles, oh no. These are steam bubbles making their grand entrance.
And then... booooom! It boils! The whole thing turns into steam, this invisible gas. It's like the water decides it's had enough of being liquid and wants to explore the gaseous dimension. All while the pressure is being a total chill dude, just observing the transformation.
Why is this fun? Because boiling is a show! It's a phase change, a transformation. It's the water throwing a little party and inviting everyone to become vapor. And the best part? You can see it happen in your own kitchen! That kettle whistling? That's constant pressure magic in action.
Quirky fact: Did you know water boils at different temperatures at different pressures? Like, if you're on top of a mountain, where the pressure is lower, your water boils sooner. It's like the water is saying, "Phew, less pressure, let's get this boiling party started!" But for us regular folks, our kitchen kettles are usually playing the constant pressure game.

Balloon Bonanza!
Okay, picture a balloon. It's filled with air, right? Now, imagine you're holding that balloon, and you're not squeezing it any harder or looser. That's our constant pressure scenario for the air inside.
Now, let's say you decide to warm up that balloon. What happens? The air inside gets all excited. The little air molecules start zooming around super fast. They start bumping into the inside of the balloon walls with more oomph.
What's the result? The balloon starts to get bigger! It expands! It inflates itself even more, all by itself! It’s like the air inside is celebrating the warmth by stretching out and taking up more space. It's a glorious expansion!
Why is this fun? Because it's like the air is a tiny, enthusiastic cheerleader. When it gets warmer, it just wants to go big or go home. And it pushes outwards, making the balloon grow. It’s a visual representation of energy making things expand.
Funny detail: If you've ever left a balloon in a hot car, you've seen this in action. The sun heats the air inside, and poof, it gets bigger. Sometimes, if it gets too hot, it might even do a dramatic exit. Whoops!
Metal Meltdown (Sort Of!)
Let's talk about metals. Tough stuff, right? But even metals can be persuaded to change their tune under constant pressure.
Think about heating up a metal rod. If you keep the pressure the same, and just crank up the heat, something interesting happens. At a certain temperature, the metal starts to soften. It gets a bit… bendy.
And if you heat it even more? It can actually melt! It goes from being solid and rigid to a flowing, liquid metal. Like mercury, but for, you know, stronger stuff. All this happening while the pressure is just being polite and not getting in the way.
Why is this fun? Because it shows that even seemingly unchangeable things have a breaking point. And when that point is reached at constant pressure, you get a dramatic transformation from solid to liquid. It's the metal's way of saying, "Okay, fine, I'll flow!"
Quirky fact: Many industrial processes rely on melting metals at constant pressure. Imagine making intricate metal parts! You need to control the heat precisely, but the pressure is usually just… there. Doing its thing. Like background music at a concert.
Gases Galore!
Gases are the ultimate chillers. They just love to spread out. And when you heat them up at constant pressure, they really show their true colors.
Remember our balloon example? It's similar for any gas. When you heat a gas at constant pressure, its volume increases. It wants more space. It's like it's saying, "Hey, thanks for the energy boost! I'm gonna take this moment to explore new horizons… in terms of volume, of course."
This is super important in lots of sciencey things. Like weather! Hot air rises because it expands and becomes less dense at constant atmospheric pressure. It's a fundamental principle.
Why is this fun? Because gases are the masters of expansion. They’re the rebels of the states of matter. And watching them puff up at constant pressure is like observing a quiet revolution. A revolution of volume!
Funny detail: Ever seen one of those hot air balloons? That’s literal hot air expanding at essentially constant atmospheric pressure, lifting the whole basket. It's science that lets you float through the sky! How cool is that?
So, Why Bother?
You might be thinking, "Okay, this is neat, but why is it fun to talk about?"
Because it's about transformation! It's about how things change, how they react to their environment, even when one crucial factor is just… being chill. It’s the science of everyday marvels. The science of your kettle, your balloons, and even the weather.
It’s a reminder that even when things seem stable (like constant pressure), there’s always potential for change, for growth, for a little bit of drama. And that, my friends, is pretty fun to ponder.
It’s the simplicity that makes it fascinating. No need for complex pressure adjustments. Just add a little heat, and watch the magic unfold. It’s accessible science. It’s the universe winking at you, showing you its cool tricks.
So next time you're watching water boil, or a balloon inflate, or even just feeling the warmth of the sun, remember the quiet hero of this story: constant pressure. It's the steady hand that allows for all sorts of fascinating changes to happen. And that's a pretty fun thing to have in your back pocket of knowledge!
