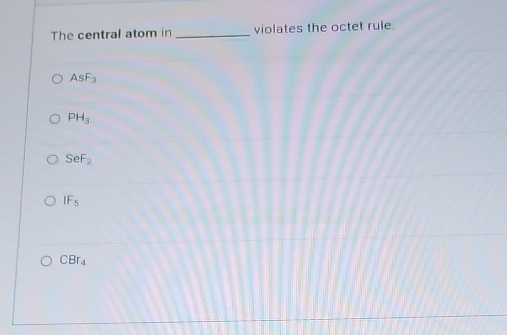
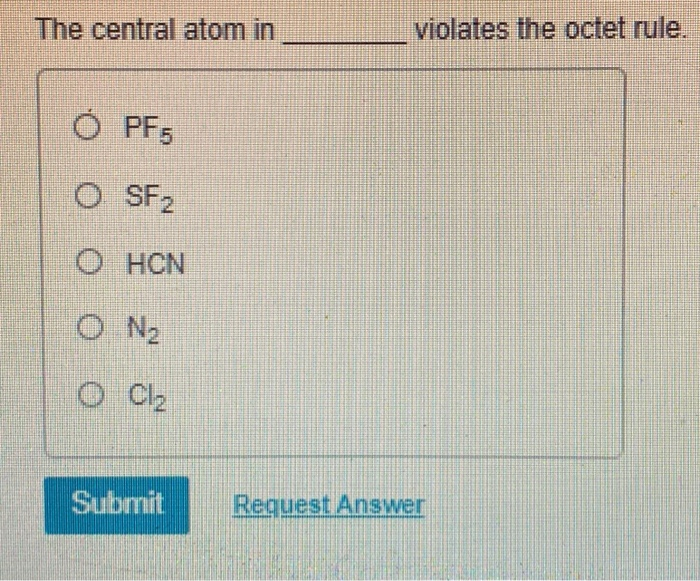
The Central Atom In ________ Violates The Octet Rule.

Okay, picture this. You're at a party, right? Everyone's trying to be cool, trying to fit in. They're all doing the same dance moves, wearing the same trendy outfits. It's all very predictable, very… eight-y.
But then, there's always one. One person who just… isn't. They're not trying to be different, they just are. They're rocking a neon green boa with glittery moon boots. They're singing off-key with gusto. They're the life of the party, but also the most baffling.
Well, in the world of atoms, there's a similar vibe happening. Some atoms are total party animals. They're all about sharing and fitting neatly into their little eight-electron circles. They're the backbone of so many things we know and love. They're the reliable friends, the ones who always have your back.
But then, you have those other atoms. The rebels. The ones who just can't seem to stick to the script. They're out there, in the grand cosmic ballroom, doing their own thing. And it turns out, they’re pretty darn important too.
Let’s talk about the Central Atom In Boron Trifluoride, for instance. Boron. Bless its little electron-deficient heart. It’s supposed to be all about the octet rule, that magical number eight that keeps everything stable and tidy. It’s the atomic equivalent of a perfectly made bed.
But Boron? It’s like, “Nah, I’m good.” It’s happy with just six electrons. Six! Can you imagine? That’s like going to the party with only half a slice of pizza. It’s just… incomplete. But somehow, it works.
And honestly, I kind of respect it. It’s a bold move. It’s saying, “I don’t need to be like everyone else to be valid.” It’s a tiny, powerful declaration of individuality. The Central Atom In Boron Trifluoride is the anti-octet rebel we didn't know we needed.

Then we have the big guys. The ones who just can't help but accumulate. Think about Phosphorus Pentachloride. Phosphorus is right there, supposed to be playing by the rules. But no. It’s got five chlorines clinging to it. Five!
That means Phosphorus is juggling a whopping ten electrons around itself. Ten! It’s like that friend who brings their entire extended family to a small gathering. It’s a lot. It’s chaotic. It’s undeniably impressive in its sheer audacity.
The Central Atom In Phosphorus Pentachloride is the overachiever. The one who volunteers for *every committee. The one who brings three dishes to a potluck. It’s not malicious; it’s just… a lot. And frankly, it’s fascinating to watch.
And what about Sulfur Hexafluoride? Oh, Sulfur. You darling. You just love to be surrounded. Six fluorines, all cozy around the sulfur. That's twelve electrons, people! Twelve!

Sulfur is practically drowning in electrons. It’s the atom equivalent of a giant hug, but with more electronegativity. The Central Atom In Sulfur Hexafluoride is the person who always has an extra pillow on the couch for you. It’s generous to a fault. It’s expansive.
It just makes you wonder, doesn't it? This whole octet rule thing. It’s like the ultimate trend. Everyone’s trying to get in on it. But what if some atoms are just… fashion-forward? What if they’re seeing beyond the immediate, the popular, the eight?
Maybe the octet rule is less of a strict law and more of a suggestion. A guideline. The atomic equivalent of “dress code: smart casual.” And some atoms are just born to break it. They’re the ones who show up in sequined overalls to a black-tie event and somehow pull it off.
It's easy to look at these exceptions and think, "Oh, that's weird." But isn't that where the real magic happens? In the unexpected, the unconventional? The Central Atom In Xenon Tetrafluoride, for example. Xenon, a noble gas! It's supposed to be aloof, unbothered by the electron-sharing drama.
But here it is, shackled to four fluorines. It's got twelve electrons! Xenon, you sly dog. You thought you were so above it all, didn't you? The Central Atom In Xenon Tetrafluoride is the quiet kid who turns out to be a secret rock star.
And what about Iodine Heptafluoride? Iodine is like, "You know what? Let's go big or go home." Seven fluorines. Fourteen electrons. Fourteen! Iodine is practically a black hole of electron density. It’s the atom that ate the party.
The Central Atom In Iodine Heptafluoride is the ultimate host. It’s so full of energy, it’s radiating it outwards. It's not just participating; it's dominating the electron space. And we love it for it.
It’s easy to get caught up in the idea that there’s only one way to be. One way to be stable, one way to be complete. But these central atoms, these rule-breakers, they’re here to remind us that there are many paths to success. Many ways to be. Many ways to… hold electrons.
They’re the proof that sometimes, the most interesting things happen when you dare to be different. When you’re not afraid to have too many friends, or too many electron partners. When you’re the Central Atom In Sulfur Tetrafluoride, or the Central Atom In Chlorine Pentafluoride, just doing your thing.
So, next time you’re looking at a molecule and you see a central atom rocking more or less than eight electrons, don't just dismiss it. Give it a little nod. A knowing smile. Because that atom is a little bit of a rebel, a little bit of an overachiever, and a whole lot of fascinating.
They’re the unexpected stars of the molecular show. The ones who prove that the octet rule, while a good starting point, isn't the be-all and end-all. The universe, and chemistry, is far more interesting than that. It's got its share of outliers, and thank goodness for them.
They're the ones that make us scratch our heads, then lean in closer. They’re the sparks of curiosity. They’re the reminders that even in the most rigid of systems, there’s room for individuality and, dare I say, a little bit of chaos. And that’s perfectly okay. In fact, it's more than okay. It’s wonderful.
