The Carbon Tetrachloride Molecule Ccl4 Has The Shape Of A
Hey there, science curious folks! Ever found yourself staring at something and thinking, "What's its deal? What's its whole vibe?" Well, today we're diving into the shape of a molecule that might sound a bit fancy, but trust me, it's got a personality you'll totally get. We're talking about our pal, carbon tetrachloride. Yep, that's CCl4 if you're feeling jazzy.
Now, carbon tetrachloride. Sounds a bit like a villain's secret lair, doesn't it? Or maybe the name of a suspiciously powerful cleaning product from the good old days. And in a way, it kinda was. This stuff was the king of stain removers and degreasers for ages. Think of your grandma's attic, filled with old photo albums and maybe a slightly musty trunk. If there was a particularly stubborn smudge on that trunk, CCl4 was the go-to weapon. It was like the ultimate superpower against dirt, only it turns out superpowers can sometimes have unintended consequences. More on that later, but for now, let's focus on its good looks, or rather, its good shape.
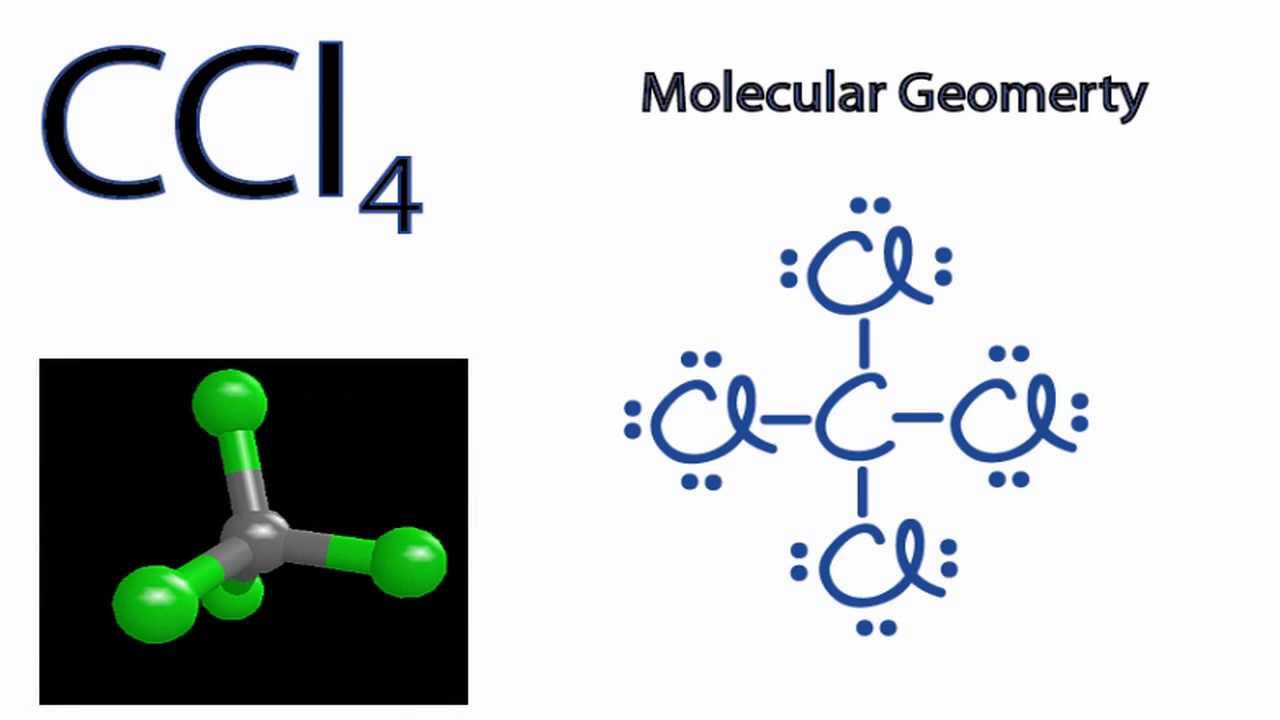
So, what does this CCl4 molecule look like? Imagine you've got a central character. This is our carbon atom. Think of it as the ringleader, the maestro, the person who always gets the last slice of pizza. This carbon atom is the nucleus of our molecular party. It's the one holding everything together.
And who are the attendees at this carbon party? They're the chlorine atoms. We've got four of them, all eager to hang out with our central carbon. Picture them as a group of really enthusiastic friends, all wanting to get as much face time as possible with the star of the show.
Now, here's where things get interesting. Molecules, just like us humans, don't like to be crowded. They want their personal space. And these chlorine atoms, they're no different. They're all negatively charged, so they're naturally going to push each other away. It’s like when you’re at a crowded concert, and everyone’s doing their best to get a little breathing room without actually leaving the mosh pit. They’re jostling, they’re shifting, they’re trying to find that sweet spot.
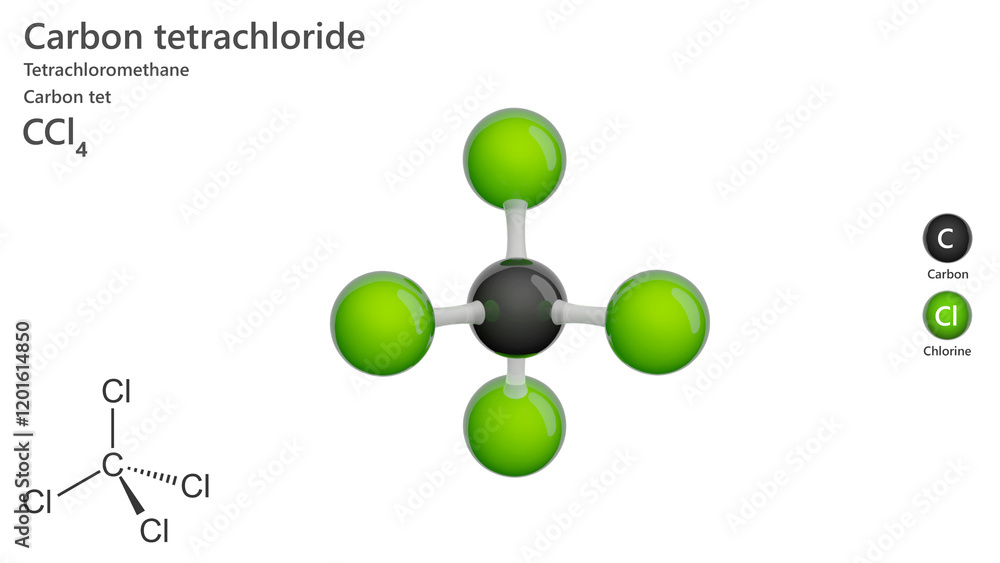
So, what's the best way for our four chlorine atoms to spread out around the carbon atom and keep their distance? They can't just line up. That would be like trying to fit four people into two seats on the bus – not ideal. They need to spread out in three dimensions. And the absolute best way to do that, to achieve maximum distance between each of them, is to form a shape called a tetrahedron.
Have you ever seen one of those geometric shapes? It looks like a pyramid, but it’s got a triangular base and then three more triangles meeting at a point. Think of a fancy diamond cut, or a perfectly sculpted gemstone. Or, even better, imagine a really awesome four-sided die – you know, the kind that looks like a pyramid with its top chopped off and another face added? That’s pretty much the vibe.

So, picture our carbon atom sitting right in the dead center of this tetrahedron. And then, each of our four chlorine atoms is perched on one of the four corners. They are spread out as far as they possibly can be from each other, all thanks to the carbon atom's willingness to share its space.
It’s like a game of molecular musical chairs, but instead of chairs, they’re trying to find the positions that are furthest apart. And the tetrahedron shape is the ultimate solution to that puzzle. It’s the most stable and energetically favorable arrangement for these atoms to be in. They're not squashed, they're not bumping into each other awkwardly. They're perfectly spaced, like well-behaved guests at a very organized party.
Let’s break it down a bit more. A tetrahedron has four identical faces, and in the case of CCl4, each of those faces is an equilateral triangle. Our carbon atom is at the heart of it all, and the four chlorine atoms are at the vertices, the pointy bits. If you were to draw lines connecting the carbon to each chlorine, you'd form four identical triangular pyramids, all meeting at that central carbon. It’s a symmetrical beauty, really.
Think about it in a less scientific way. Imagine you’ve got a bunch of balloons, and you want to tie them all to a central point. If you tie four balloons, and you want them to be as far apart as possible, you wouldn’t bunch them up. You’d let them spread out, and they’d naturally want to go to the corners of a tetrahedron. It’s like they know that’s the best way to chill.
Or, consider those old-school fireworks that explode into a shower of sparks. Some of them form beautiful, symmetrical patterns. A good tetrahedral shape is like one of those perfectly formed bursts. Each spark (the chlorine atom) is at an equal distance from the center (the carbon atom), creating a balanced and pleasing aesthetic. Well, as pleasing as a molecule can be, anyway.
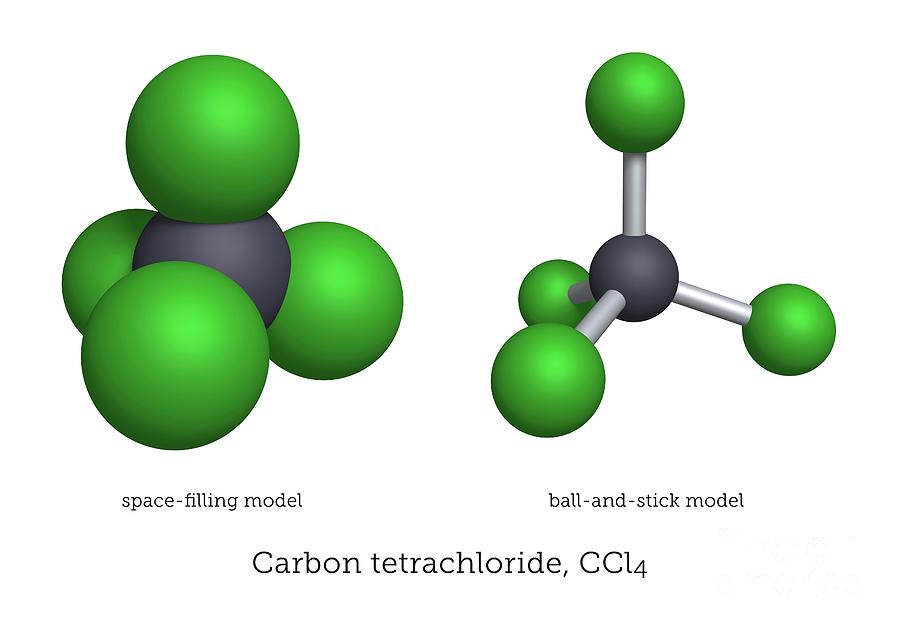
So, the key takeaway here is that the carbon atom in CCl4 is in the middle, and the four chlorine atoms are all around it, spread out equally in a shape that looks like a pyramid with a triangular base. This arrangement is called a tetrahedral geometry. It’s a super stable arrangement, which is why molecules often naturally adopt these shapes. Nature, it seems, likes things neat and tidy.
This tetrahedral shape is incredibly important. It dictates how the molecule interacts with other molecules. It influences its physical properties, like its boiling point and its solubility. It's the foundation for everything this molecule does. It’s like the foundation of a house – if it’s strong and well-built, the whole structure can stand tall.
Now, you might be wondering, "Why all this fuss about shape?" Well, think about how we humans are. The shape of our hands allows us to grab things. The shape of our lungs allows us to breathe. Our entire existence is built on shapes and how they function. Molecules are no different, just on a much, much smaller scale.
And CCl4’s tetrahedral shape made it a fantastic solvent. Because it’s so symmetrical, it’s what we call nonpolar. This means it doesn't have a significant positive or negative end. Think of it like a perfectly balanced seesaw – nothing's dipping lower than the other. This nonpolar nature makes it excellent at dissolving other nonpolar substances, like oils and greases. That's why it was so good at cleaning.
Imagine trying to wash a greasy frying pan with just water. Doesn't work too well, right? Water is polar, and oil is nonpolar. They don't mix. But if you add a little bit of a nonpolar solvent like CCl4, bam, the grease dissolves right up. It’s like inviting two very different types of people to a party, and one of them (the water) just stands awkwardly in the corner while the other (the oil) mingles freely with the nonpolar CCl4.
However, as we alluded to earlier, this powerful cleaning agent had a darker side. Its very effectiveness and stability meant it didn't break down easily in the environment. And, it turns out, it's not exactly a friend to our ozone layer. Yep, that protective shield in the atmosphere that keeps the sun’s harsh rays from frying us all. CCl4, when it got up there, was a real party pooper for ozone.
So, while its tetrahedral shape made it a chemical marvel for cleaning and other industrial uses, that same stability made it a bit of an environmental menace. It’s like that friend who’s incredibly useful and always there for you, but then they accidentally break your favorite vase. You appreciate their help, but you’re also a little wary.
Because of its ozone-depleting properties, the use of carbon tetrachloride has been significantly restricted and phased out in many parts of the world. We’ve moved on to greener, less harmful alternatives. It's a classic case of how understanding the molecular world, including its shapes, can lead to both amazing innovations and the realization that sometimes, even the most useful things need to be rethought.
So, next time you hear about carbon tetrachloride, remember its perfectly symmetrical, tetrahedral shape. It’s a shape that’s not just an abstract concept in a chemistry textbook, but a fundamental aspect that defined its properties and its role in our world. It’s a little bit of molecular architecture that has had a very real impact on our lives, both good and, well, not so good. And that, my friends, is the simple, yet fascinating, story of the CCl4 molecule and its oh-so-tidy, tetrahedron-tastic shape.
It’s a reminder that even the tiniest things, the building blocks of everything around us, have intricate structures and behaviors that shape the larger world in ways we might not always immediately realize. And frankly, a molecule that arranges itself into a perfect, balanced tetrahedron? That’s pretty cool in my book. It’s like nature’s way of showing off its geometric skills, one molecule at a time.
So, there you have it. Carbon tetrachloride, the molecule with the shape of a tetrahedron. Not so scary, right? Just a central carbon atom, four chlorine atoms, and a whole lot of personal space leading to a beautifully balanced, three-dimensional arrangement. It’s a simple shape, but it’s the key to its story, from its days as a cleaning superstar to its current status as a chemical that’s best admired from a distance.
It’s a bit like admiring a really well-made piece of furniture. You appreciate the craftsmanship, the symmetry, how all the parts fit together perfectly. The tetrahedral shape of CCl4 is its own kind of molecular furniture – functional, elegant in its own way, and with a history that teaches us a lot about the delicate balance between utility and environmental responsibility. And who knew chemistry could be so… geometrically satisfying?
So, the next time you’re thinking about molecules, give a nod to the humble tetrahedron. It's out there, in countless compounds, quietly dictating how the universe at its smallest level behaves. And for CCl4, it was its defining characteristic, a perfectly balanced shape that led to both its triumphs and its eventual retirement from the front lines of cleaning duty. A truly interesting character in the grand play of chemistry.
The beauty of science is that even the most complex ideas can be broken down into relatable concepts. And the tetrahedral shape of CCl4, while sounding technical, is really just about atoms finding their happy, spaced-out equilibrium. It's a fundamental principle of how matter organizes itself, a tiny dance of attraction and repulsion that results in elegant, stable structures. So, feel a little smarter today, because you now know that CCl4 is shaped like a geometrically perfect, four-sided pyramid. Pretty neat, huh?
