Tendency Of Electrons To Enter Orbitals Of Lowest Energy First
Ever wondered why atoms are so predictable, or how tiny electrons manage to arrange themselves in such an orderly fashion? It's not magic, it's just a fundamental rule of the universe, and understanding it can be surprisingly fun and useful! Think of it like a game of musical chairs for electrons, where they always try to grab the most comfortable seat first. This tendency for electrons to fill the lowest energy orbitals first is a cornerstone of chemistry and helps us understand everything from how elements behave to why materials have the properties they do. It’s a concept that pops up in countless scientific fields, making it incredibly relevant.
For beginners just dipping their toes into the world of science, this idea is a fantastic starting point. It provides a simple, logical framework for understanding atomic structure, demystifying what goes on inside those tiny building blocks of everything. For families, it's a great opportunity to spark curiosity in children. You can use analogies like filling up a house with furniture, starting with the most important rooms, or a backpack where you pack your heaviest books first. Hobbyists, perhaps those interested in electronics or even gardening (as plant nutrient uptake is influenced by atomic structure!), can gain a deeper appreciation for the underlying principles that govern their interests. It’s about understanding the 'why' behind what you see and do.
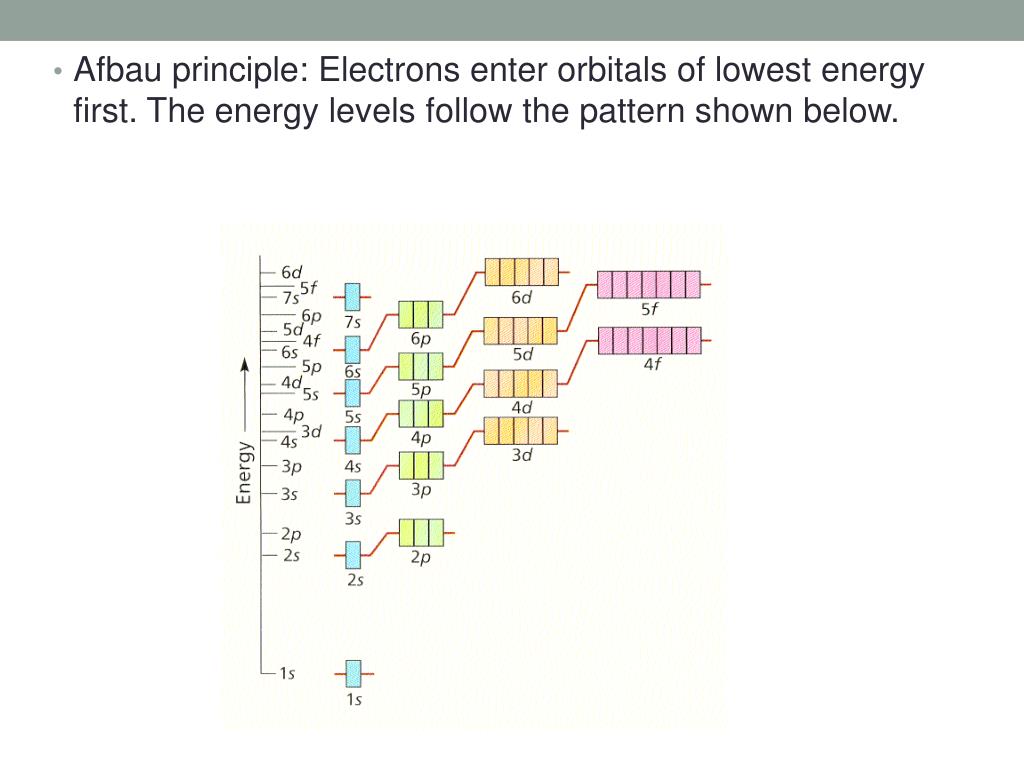
Let's consider an analogy: Imagine you have a multi-story building with rooms at different heights. The rooms on the lowest floors are the easiest to get to – they require the least energy to reach. Electrons are similar. They’re like tiny inhabitants that prefer the 'ground floor' orbitals. These are the orbitals with the lowest energy. Once those are full, they start moving up to the next available, slightly higher energy orbitals. This orderly filling is described by rules like the Aufbau principle. For example, the first two electrons in any atom will occupy the 1s orbital, the lowest energy orbital available. Then, as you add more electrons, they begin to fill the 2s orbital, and so on. It’s a pattern that repeats itself across the entire periodic table!
Getting started with this concept is easier than you might think! You don't need a fancy lab. Start with simple diagrams of atomic orbitals. You can find many visual aids online that show these energy levels and how electrons fill them. Think of it like a puzzle. Try to draw out the electron configurations for the first few elements, like Hydrogen, Helium, and Lithium, and see how the electrons follow this 'lowest energy first' rule. You can even use colored beads or small objects to represent electrons and containers to represent orbitals and physically move them around to simulate the filling process. It’s a hands-on way to solidify the concept.
Ultimately, understanding that electrons have a tendency to enter orbitals of lowest energy first is about appreciating the fundamental elegance of nature. It's a principle that elegantly explains so much about the world around us, from the stability of atoms to the formation of molecules. It’s a simple rule with profound implications, and exploring it can be an incredibly rewarding and enjoyable journey.
