Substances Whose Water Solutions Conduct Electricity Easily

Hey there, science curious friend! Ever wondered why some liquids spark and others just… well, sit there looking wet? Today, we're diving into the awesome world of things that make water a little more electric. Think of it like adding some extra zest to your plain ol' water. It’s not rocket science, but it’s definitely cool science! And don't worry, we're keeping this super chill, like sipping lemonade on a porch. No intimidating textbooks here, just good ol' chatting about stuff that conducts electricity.
So, what’s the big deal about conducting electricity? Basically, it means that these special substances, when dissolved in water, let electricity flow through them. It's like they open up little highways for those tiny electrical charges to zip and zoom around. Imagine a traffic jam versus an open freeway – these guys create the freeway!
The main culprits behind this electric magic are usually things that break apart into charged particles when they dissolve. We call these charged particles ions. Think of them as tiny, energetic little bits with a positive or negative attitude. When they're floating around in water, they’re free to move, and that’s where the electricity comes in. Pretty neat, right?
The Usual Suspects: Acids and Bases, Oh My!
Let's start with some of the most famous conductors. You've probably heard of acids and bases. They're like the dynamic duo of the chemical world, and when they meet water, things get electrifying!
Think about vinegar, that stuff you use on salads. It's an acid, specifically acetic acid. When you put vinegar in water, those acetic acid molecules get a bit… jumpy. They break apart into hydrogen ions (H+) and acetate ions (CH3COO-). Those little guys are ready to party with electricity!
So, if you were to stick a couple of electrodes into a saltwater solution (which is basically just a solution of sodium chloride, or table salt, in water – we'll get to that one!), you'd see a nice, steady flow of electricity. It's because the salt breaks down into positively charged sodium ions (Na+) and negatively charged chloride ions (Cl-).
Now, bases are like the opposite of acids, but they also love to create ions. Think about baking soda. It's a base. When you dissolve baking soda in water, it forms sodium ions (Na+) and bicarbonate ions (HCO3-). Again, bingo! More ions, more conductivity. It’s like a conductivity party, and everyone’s invited!
This is why laboratories and even some household cleaners can be a bit zappy if you're not careful. They’ve got these ions just hanging out, waiting for a chance to conduct. So, always be mindful, folks!

Acids: The Zesty Conductors
Acids are pretty cool because they tend to break apart quite easily in water, releasing a lot of those positively charged hydrogen ions. These hydrogen ions are super mobile and love to get things moving electrically. It's like they’re the sprinters of the ion world!
The stronger the acid, the more ions it tends to produce, and therefore, the better it conducts electricity. Think of hydrochloric acid (HCl) – that’s the stuff in your stomach to digest food! It's a strong acid and makes water a fantastic conductor. Of course, you don't want to go around drinking battery acid, that would be a very bad idea and would not end well. Stick to vinegar for your experiments, please!
Even weak acids, like the citric acid in lemon juice, can make water conduct electricity to some extent. So, that fizzy lemonade you enjoy? It's got a little bit of electrical conductivity happening in there, thanks to those citrusy acids!
Bases: The Slippery Conductors
Bases are the flip side of the coin, but just as effective at creating conductive solutions. They often release negatively charged ions and sometimes positively charged metal ions.
Sodium hydroxide (NaOH) is a common strong base. Dissolve it in water, and it breaks into sodium ions (Na+) and hydroxide ions (OH-). These guys are great at letting electricity flow. It’s like they’re setting up a whole system of electrical highways.

Ammonia (NH3) is another base, though it's a bit weaker. When you add ammonia to water, it doesn’t break apart as much as sodium hydroxide, but it still produces enough ions to make the water conduct electricity. That’s why ammonia-based cleaners can sometimes be a bit… energetic. Just a friendly reminder to handle them with care!
The All-Star: Salt Water!
Ah, salt water. The classic example. You know, the kind you find in the ocean or when you’re making pretzels. It’s a champion conductor, and it’s incredibly simple.
Table salt, or sodium chloride (NaCl), is an ionic compound. This means it’s made up of positively charged sodium ions and negatively charged chloride ions that are held together in a crystal structure. When you toss that salt into water, the water molecules are super friendly and pull those ions apart, scattering them throughout the solution.
Each of those little Na+ and Cl- ions is now free to roam, and when you apply an electrical voltage, they all start moving. The positive ions move towards the negative electrode, and the negative ions move towards the positive electrode. It's like a tiny, orderly migration!
This is why swimming in the ocean is generally safe from electrical shock (though swimming during a thunderstorm is a terrible idea for other reasons!). The salt in the water makes it conductive, but it’s not like it’s carrying a huge electrical current on its own. It just provides the pathway. Think of it as providing the infrastructure for electricity, not generating the power itself.

Other salts, like potassium chloride (KCl) or magnesium sulfate (MgSO4 – Epsom salt!), also dissolve in water to form ions and make the solution conductive. They’re all part of the same conductive club!
What About Pure Water? The Shy One
Now, you might be thinking, "What about plain old, super-pure water? Is it a conductor?" And the answer is… not really. Pure water is like that quiet kid at the party who doesn't do much. It has a few water molecules that naturally break apart into H+ and OH- ions, but there are so few of them that it barely conducts electricity at all.
It’s like trying to have a conversation in a library – there’s just not enough chatter. For water to be a good conductor, you need those ions to be plentiful. That’s why even tap water, which has small amounts of dissolved minerals (which are basically salts!), conducts electricity better than pure water. It’s got a little bit of that ion action going on!
The Not-So-Conductive Pals: Sugar and Oil
Okay, so we’ve talked about the stars of the show. But what about the ones that don't conduct electricity well? These are often substances that don't break apart into ions when they dissolve.
Take sugar, for example. When you stir sugar into your tea or coffee, those sugar molecules stay whole. They dissolve, sure, but they don't split into charged particles. They’re like a bunch of buddies going for a swim together, all sticking close and not breaking up. So, sugary water is a pretty poor conductor of electricity. It’s like the shy kid from the library who’s now at a party but still doesn’t want to mingle much.
Oil is another one. Oil and water don't even mix properly, do they? And if they did, the oil molecules are large and non-polar. They don't have those charged ends that can interact with water to form ions. So, oil in water is a terrible conductor. It’s like trying to get oil and water to conduct electricity – a messy, non-conductive situation!
Why Does This Matter Anyway?
You might be asking, "Why should I care if water conducts electricity?" Well, this stuff is actually super important in real life! It's the principle behind many things:
- Batteries: The liquid inside batteries, called electrolyte, is a conductive solution that allows chemical reactions to happen and generate electricity.
- Electroplating: This is how we put a thin layer of one metal onto another, like making jewelry shiny. It relies on conductive solutions.
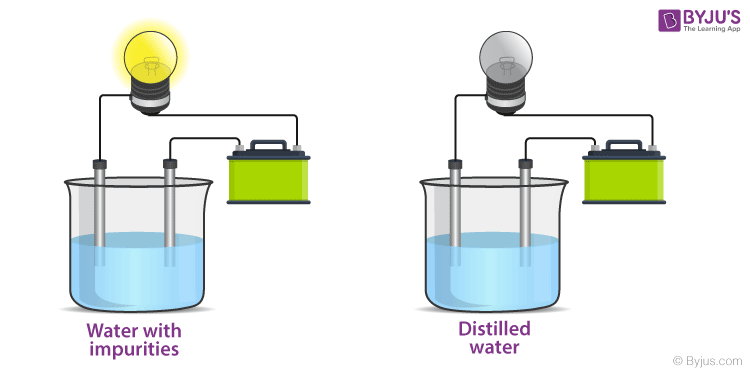
- Water Purification: Understanding conductivity helps us know how pure our water is. High conductivity in supposedly pure water often means there are dissolved impurities.
- Biological Systems: Our own bodies rely on ions in our blood and cells to conduct electrical signals. Your nerves firing? Yep, that's ion-based conductivity at work! Pretty mind-blowing to think about, right?
So, next time you're sipping on something that's not just water, or you're handling common household items, remember that there's a whole world of invisible electrical activity going on!
The Electrifying Takeaway
So there you have it! We've chatted about how acids, bases, and especially salts can turn ordinary water into an electrical highway. It's all thanks to those amazing little charged particles called ions that love to roam free when dissolved.
It’s a reminder that even seemingly simple things, like a glass of water, can have hidden depths and fascinating properties. The world around us is full of these little wonders, just waiting to be noticed and appreciated. So, keep that curious spirit alive, and remember that science isn't just for labs – it's for everywhere, making everyday life just a little bit more awesome and, dare I say, electrifying!
Isn't that cool? Now go forth and impress your friends with your newfound knowledge of conductive waters. Or, you know, just enjoy your salty snacks with a little more scientific appreciation. Either way, happy exploring!
