Student Exploration Temperature And Particle Motion
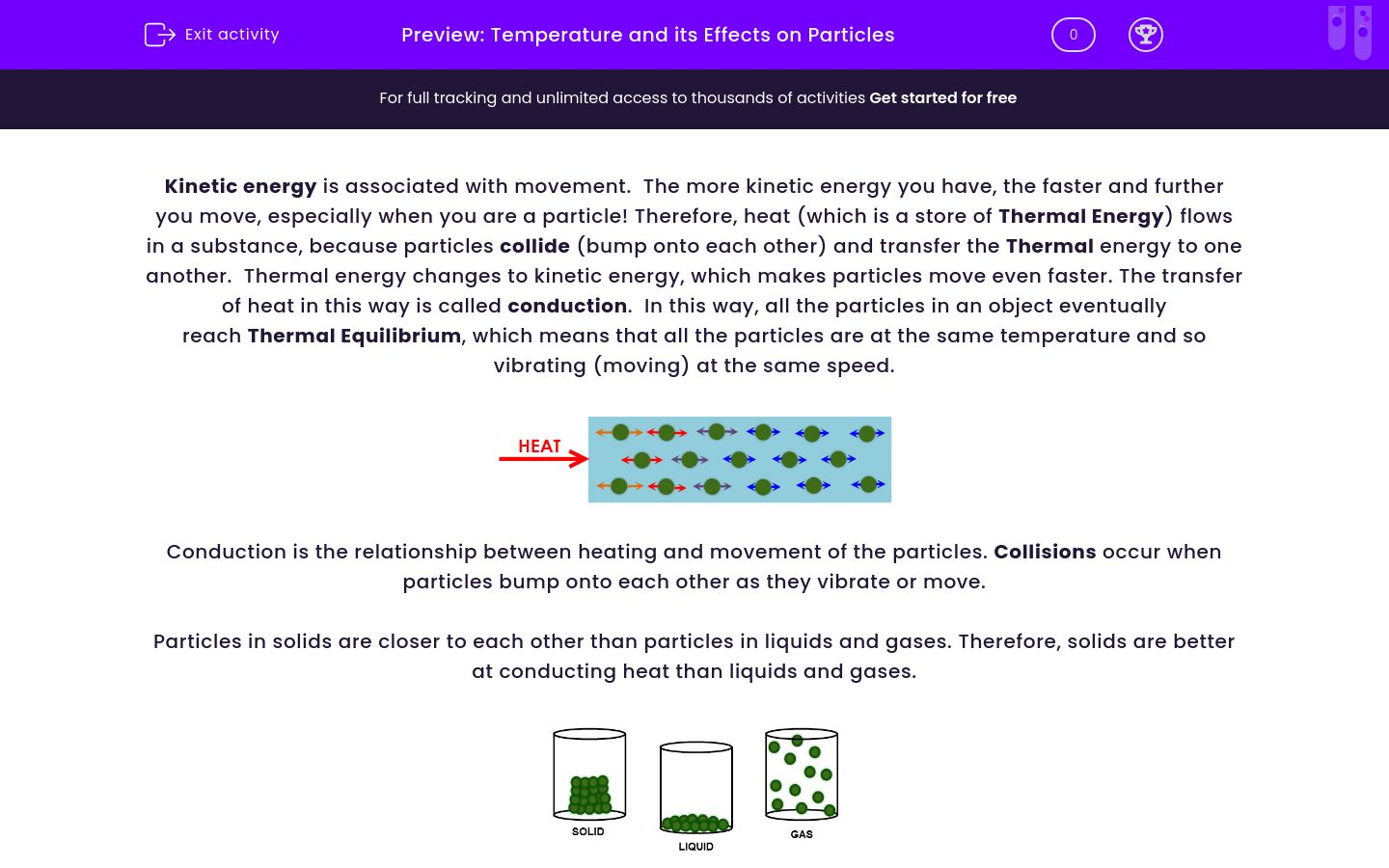
Hey there, science curious peeps! Ever wonder what's really going on when you heat up some water or stick your hand in a chilly breeze? It's way cooler than you think. We're talking about student exploration of temperature and particle motion. Yep, those tiny little bits that make up EVERYTHING. And guess what? It's actually super fun to dig into.
So, let's get real. Temperature isn't just a number on a thermometer. It's like a secret code for how much your little particle buddies are vibing. Think of it as their party energy. High temperature? They're going wild! Low temperature? They're chilling, maybe even snoozing.
Imagine you've got a bunch of hyperactive toddlers in a bouncy castle. That's like a hot gas. They're bouncing off the walls, bumping into each other constantly. Super energetic, right? They're moving super fast, all over the place. That's what happens to particles when things get hot. They get all excited and zoom around.
Now, picture those same toddlers after a long day, maybe after a nap. They're still moving, but much slower. Maybe they're just shuffling their feet or slowly bumping into each other. That's more like a cold liquid. Still got some energy, but not the full-blown rave.
And then, you have the ultimate chill. Think of a room full of sleeping puppies. They might twitch a little, but they're mostly settled. That's kind of like a solid. The particles are still there, doing their thing, but they're stuck in place, just wiggling a bit. They're holding hands, basically. Super close and not moving around much.
The More You Heat It, The More They Party!
This is where it gets really interesting. When you add heat, you're basically pumping up the volume on the particle party. You're giving them more energy to bounce, jiggle, and generally go nuts. Think of it like turning up the music at a concert. The crowd goes wild!

For example, ice. It's super cold. Those water particles are basically holding hands really tightly, just vibrating in their spots. They're in their solid, structured formation. It's like a super organized dance troupe. Very controlled.
But then, you start to melt that ice. You're giving those water particles a little more wiggle room. They start to break free from their tight embrace and slide past each other. They're still close, but they're not stuck. They're like a group of friends deciding to mingle at a party. They can move around, but they're still in the same room.
And when you boil that water? Oh boy, it's a full-on rave! Those water particles have SO much energy, they break free from each other completely. They zoom around, filling up the entire container, even escaping into the air as steam. They're like the dancers who've left the dance floor to mingle with everyone else in the entire building. They're spreading out everywhere!

Quirky Facts You'll Love
Did you know that even in the coldest place imaginable, like absolute zero (which is super cold, like, impossibly cold), particles aren't completely still? They still have a little bit of "quantum jiggle." It's like the universe's way of saying, "Nope, no total slacking allowed!" Kind of funny, right? Even when they're trying their hardest to chill, they can't be totally motionless.
And speaking of funny, think about popcorn. You heat up those little kernels, and BAM! All that stored energy in the water molecules inside bursts out, turning them into steam and making them explode into delicious fluffy clouds. It’s like the kernel had a secret party inside, and when the temperature got high enough, it just couldn't contain its excitement anymore.
Or consider that time you left a cold drink outside on a hot day. You know how it gets all foggy on the outside? That's water vapor from the air cooling down and turning back into tiny liquid water droplets on your cold cup. It’s like the air’s moisture decided to have a little condensation party on your drink because it was so much cooler.

It’s these little everyday phenomena that really highlight the connection. Temperature is the conductor, and particle motion is the orchestra. When the conductor raises their baton (or turns up the heat!), the orchestra plays louder and faster.
Why This Stuff Is Actually Kinda Cool
So, why should you care about particles partying and vibrating? Because it explains SO much! It's the why behind why things melt, boil, freeze, and even why your ice cream melts faster on a hot day. It’s the fundamental reason behind so many everyday experiences.
Understanding this stuff isn't just for fancy scientists in lab coats. It's for anyone who's ever wondered about the world around them. It’s about building a better picture of how everything works, from the smallest atom to the biggest planet. And honestly, thinking about tiny particles having tiny parties is just a fun mental image, isn't it?

When you see steam rising from a hot cup of tea, you’re not just seeing steam. You’re seeing water molecules that have had a serious energy boost. They’re so excited they’ve broken free and are zooming around like tiny, invisible race cars.
Or when you touch something cold, you’re feeling the absence of that high-energy party. The particles in the cold object aren’t vibrating as much, so they’re stealing a little bit of energy from your warmer hand. It’s like they’re saying, "Ooh, you're warm! Can I borrow some of that energy?"
It’s all about energy transfer, and temperature is our main clue. It’s a constant dance of energy, and particles are the dancers. Sometimes they’re doing a slow waltz, sometimes a frantic mosh pit. And all of it is dictated by how much energy they’ve got, which we call temperature.
So next time you’re feeling the heat or the chill, take a moment to appreciate the microscopic dance party happening all around you. It's a tiny universe of motion and energy, and it's pretty darn fascinating. Go forth and explore that particle motion! It’s a blast.
