Student Exploration Dehydration Synthesis Answer Key

Ever feel like your brain is running on fumes? Like you've been pouring out all your good ideas and now there's just… nothing left? Well, turns out, us humans (and all living things, really!) have a surprisingly simple, yet utterly vital, way of dealing with this. It’s called dehydration synthesis, and it’s basically how we build all the cool stuff our bodies need to keep going. Think of it like a tiny, biological construction site happening inside you every second of every day.
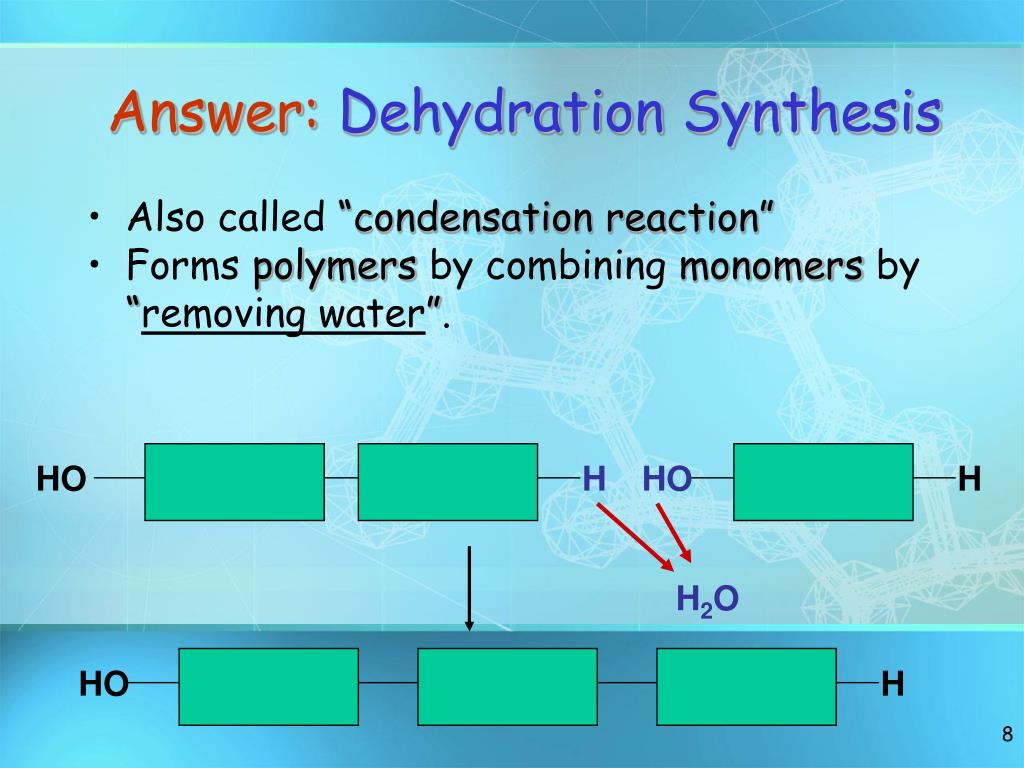
Now, before you picture a bunch of sweaty construction workers with tiny hammers, let’s break it down. Imagine you have a bunch of LEGO bricks. These bricks are like small molecules. Dehydration synthesis is the fancy name for when your body takes two of these small LEGO bricks and sticks them together to make a bigger, more useful LEGO creation. What’s the catch? Well, to make them stick, you have to squeeze out a tiny bit of water. Hence, dehydration (losing water) and synthesis (making something new). It's like saying, "Okay, you two little guys, hold hands, but you gotta give up a tiny tear to seal the deal!"
This process is everywhere. Remember that energy bar you had earlier? Dehydration synthesis was busy at work, taking smaller sugar molecules and linking them up to create bigger, more complex carbohydrates that your body could slowly use for fuel. Or that protein you ate? Your body used dehydration synthesis to link together amino acids, those little building blocks of protein, to create muscles, enzymes, and all sorts of other amazing things. It's like your body is a master chef, constantly combining simple ingredients (small molecules) into delicious and essential meals (big molecules), and the "secret ingredient" to get them to bind? A little bit of water, given up graciously.
Think of it as nature's way of saying, "Let's build something awesome, and all it costs is a tiny bit of H2O!"
And what about the "answer key" part of this whole saga? Well, in the grand, chaotic classroom of life, sometimes we need a little help figuring out how these complex processes work. When students are learning about biology, they often get exercises or problems that ask them to illustrate or explain dehydration synthesis. The answer key is simply the guide that shows them the right way these little molecules link up, the correct places where the water is "squeezed out," and the resulting larger molecule. It's like the teacher's cheat sheet, but for the amazing machinery inside us!

It's quite humbling, really, to think about the intricate dance of molecules happening constantly within us. This process isn't just about building; it's about efficiency. It’s about taking readily available, smaller components and turning them into exactly what’s needed. Imagine a tiny city being built, brick by brick, molecule by molecule, all powered by a surprisingly simple chemical reaction. The answer key for these students isn't just about memorizing facts; it's about understanding the elegant, fundamental ways life builds itself. It’s the blueprint for the amazing structures that make us, well, us.
Sometimes, when you're feeling a bit sluggish, a little worn out, it might be a sign that your internal construction crew needs more raw materials. More food, more water to keep the process going smoothly. It’s not a failure of the system; it’s a call for more building blocks and a bit of energy to fuel the dehydration synthesis machine. So next time you feel a bit drained, or marvel at the complexity of your own body, remember the humble LEGO bricks and the tiny tear of water that makes it all possible. It’s a beautiful, ongoing construction project, and the answer key is written in the very fabric of life itself.
This process is also crucial for how our bodies store energy. When we eat more sugar than we need immediately, our bodies link these simple sugar molecules together to form glycogen or fat. This is dehydration synthesis in action, creating larger, more stable molecules for later use. Think of it as packing away your LEGO creations for a rainy day. When you need that energy back, your body uses a different, but related, process to break those big molecules down again, but that’s a story for another day!
The beauty of dehydration synthesis lies in its universality. From the smallest bacterium to the largest whale, this fundamental reaction is at play. It’s a testament to the elegant simplicity that underlies the incredible diversity of life on Earth. So, while the term might sound a bit technical, the concept is as down-to-earth as it gets: building big things by joining small things and giving up a little water in the process. And for students grappling with this concept, the answer key isn't just about passing a test; it’s about unlocking a fundamental secret of life itself.
