Student Exploration Collision Theory Answer Key

Hey there, fellow explorers of life and, well, sometimes, the microscopic! Ever feel like your brain is just buzzing with questions, especially when you’re diving into the wild world of science? We’ve all been there, right? Staring at a textbook, a concept clicking one minute and then doing a full 180 the next. Today, we’re gonna chat about something that sounds a bit… intense, but is actually pretty chill when you get down to it: Student Exploration Collision Theory Answer Key. Sounds like something out of a sci-fi movie, doesn’t it? Like a secret formula to unlock universal truths. But really, it’s just a way to help us understand how things bump into each other to make other things happen. Think of it as the ultimate cosmic high-five that leads to something new.
We’re not talking about fender benders in the parking lot (though, let’s be honest, those are pretty common on campus too, especially around finals week!). We’re talking about the teeny-tiny world of atoms and molecules. You know, those building blocks of everything. Collision theory is basically the idea that for a chemical reaction to occur, particles (atoms or molecules) need to collide with each other. Sounds simple enough, right? But there’s a little more nuance to it, like trying to perfect that avocado toast recipe – it’s not just about throwing things in a pan; it’s about the right ingredients, the right heat, and the perfect timing.
The Science Behind the Bumps and Grinds
So, what’s the big deal about these collisions? Well, not all bumps lead to a chemical makeover. Imagine two people bumping into each other on a busy street. Most of the time, they just say "excuse me" and move on. But what if one person was holding a tray of delicate macarons and the other was wearing a pristine white shirt? Bam! A reaction. In the molecular world, this "bam" moment requires a few key ingredients:
1. Enough Energy to Make Things Happen
This is where the concept of activation energy comes in. Think of it as the minimum amount of oomph, the spark, the sheer willpower needed for a reaction to kick off. Without it, those molecules might bounce off each other like superballs with no real effect. It’s like trying to start a campfire without any matches. You have the wood (the molecules), but you need that initial spark to get the flames going. This is why sometimes reactions need a little heat or a catalyst to get them moving. Ever tried to bake cookies without preheating the oven? Not the best results, right?
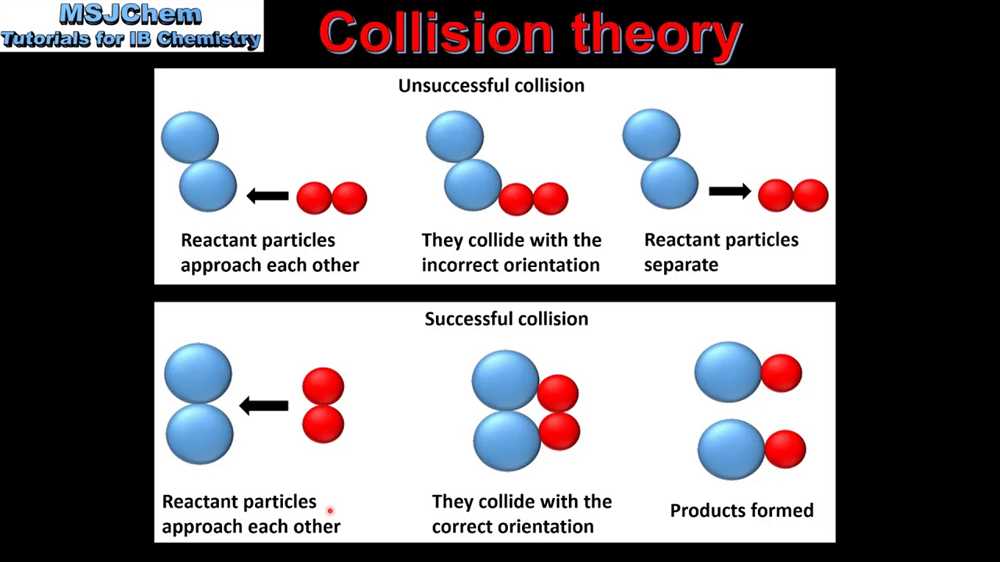
2. The Right Angle for Impact
It’s not just about crashing into each other; it’s about crashing in the right orientation. Picture playing a game of billiards. If you hit the cue ball at just the right angle to strike the 8-ball, you’re gonna sink it. If you miss the mark, it might just carom off somewhere else. Molecules are similar. They need to collide in a way that allows their atoms to rearrange and form new bonds. Sometimes, it’s about lining up those specific atoms that want to get together. It’s like a cosmic dating app – they need to be in the right place at the right time with the right vibe.

3. Frequency Matters!
The more often these molecules bump into each other with enough energy and the correct orientation, the faster the reaction will be. More collisions = more opportunities for a chemical makeover. This is why reactions tend to speed up when you increase the concentration of the reactants. It’s like a popular pop-up shop – the more people are browsing, the higher the chance of someone actually buying something. Or, think about a networking event; the more conversations you have, the more likely you are to make a valuable connection.
Navigating the "Answer Key" Maze
Now, let’s talk about this "answer key" part. When you’re exploring collision theory, whether it's in a lab, through simulations, or just reading a textbook, you're often presented with problems, experiments, or scenarios. The "answer key" is essentially your guide, your cheat sheet, your trusty sidekick that helps you understand if your predictions and conclusions about these molecular interactions are on the right track. It’s not about passively copying answers; it's about using it as a learning tool to solidify your understanding.
Think of it like this: you’re trying to learn a new dance. You practice the steps, you try to get the rhythm, and maybe you watch some tutorials. The answer key is like having a choreographer who tells you, "Yes, that turn was perfect!" or "Try to bring your arm up a little higher on that move." It’s about refinement and confirmation. It helps you identify where you might be missing a beat or misinterpreting a step. And in science, understanding is key!
Practical Tips for Collision Theory Mastery (Without the Stress)
So, how can you make exploring collision theory less of a chore and more of an insightful journey? Here are a few tips, inspired by the easy-going vibe of a Sunday brunch:
1. Visualize, Visualize, Visualize!
Don’t just read about molecules; see them. Use animations, draw diagrams, or even act it out with friends (safely, of course!). Imagine yourself as a tiny atom, bouncing around, needing just the right amount of energy to high-five another atom in a specific way to create something awesome. Websites like PhET Interactive Simulations offer fantastic, free simulations that let you play with variables and see collision theory in action. It’s like playing a science video game!
2. Connect to Everyday Reactions
Collision theory isn’t just for fancy labs. Think about how things rust (iron reacting with oxygen – lots of collisions!), how food spoils (microbes causing reactions), or even how baking works (heat providing activation energy, ingredients colliding). When you see these things, mentally connect them back to collision theory. It makes the abstract tangible. It’s like spotting an influencer’s outfit in real life – you recognize the style and understand the inspiration.

3. Break Down the "Why"
When you’re looking at an answer key, don’t just check if your answer is right or wrong. Ask yourself why it's right or wrong. Did you miss the activation energy requirement? Was the orientation the issue? Understanding the underlying principles behind the correct answer is far more valuable than just getting the point. It's like figuring out why a certain meme went viral; it's not just the image, it's the context, the relatability, and the timing.
4. Embrace the "Mistake" as a Learning Opportunity
Mistakes are not failures; they are stepping stones. Every "wrong" answer on your practice problems is a chance to learn. If you're consistently getting questions wrong about reaction rates, it might indicate you need to focus more on the factors affecting collision frequency. It’s like a GPS recalculating your route – you might have taken a wrong turn, but it’s guiding you back to your destination.
Fun Facts and Cultural Nods
Did you know that the concept of collision theory has roots in the work of scientists like Svante Arrhenius, who was basically the OG of understanding reaction rates? He was doing this back in the late 1800s! Talk about ahead of his time. It’s like discovering a hidden gem of a band that’s been making amazing music for decades before they hit the mainstream.

And speaking of reactions, think about the incredible chemical reactions happening inside your body right now. Your metabolism, your digestion, even your brain firing signals – it’s all based on controlled molecular collisions. It’s the ultimate biological rave, happening 24/7!
Sometimes, exploring science can feel a bit like deciphering ancient hieroglyphs. But with collision theory, the key is in the movement and the energy. It's about understanding that even the smallest interactions have the potential to create something entirely new. It’s the science behind every transformation, from a spark igniting a fire to the formation of a new star in the galaxy.
A Moment of Reflection
So, when you’re wrestling with collision theory, or any challenging scientific concept for that matter, remember that it’s all part of the exploration. It’s about asking questions, experimenting, and learning from the results. Just like in life, sometimes things don't go as planned. You might miss a deadline, you might have a conversation that doesn’t land quite right, or you might even burn your toast (we’ve all been there!). But it’s in those moments of collision, those unexpected bumps, that we often learn the most. We adjust our approach, we gain new understanding, and we get a little closer to that perfect outcome. So, keep bumping, keep exploring, and keep those molecular collisions (and your own life’s collisions) leading to something amazing. After all, the most exciting discoveries often happen when different elements come together in just the right way.
