Structural Isomers For The Molecular Formula C4h9br
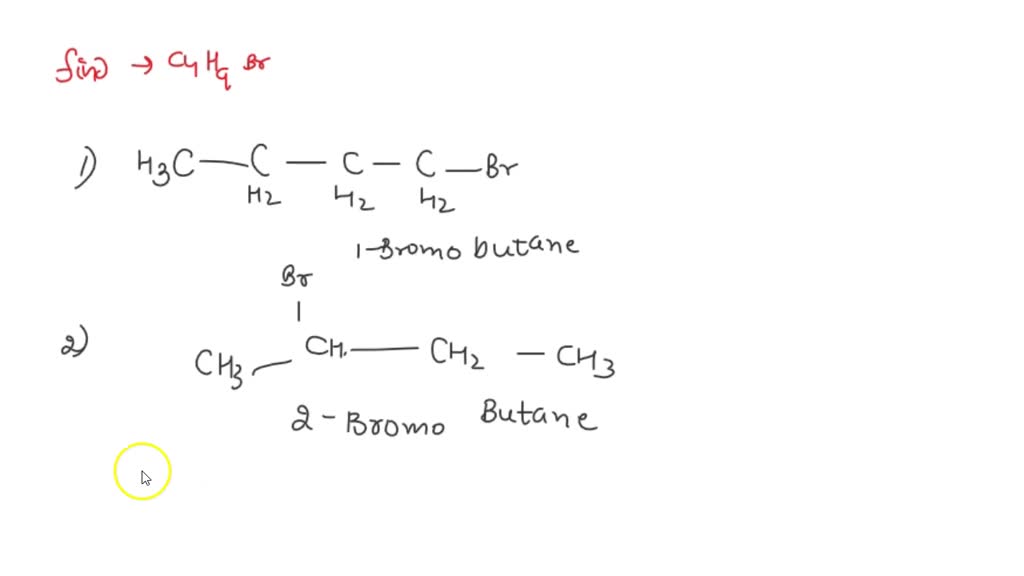
So, we're going to talk about molecules. Specifically, molecules with the formula C4H9Br. Sounds a bit like a secret agent code, doesn't it? But trust me, these guys are way less glamorous and a lot more… structurally complex.
Imagine you have four carbon atoms, nine hydrogen atoms, and one bromine atom. You can snap them together in different ways. It's like building with LEGOs, but the final shape matters a lot. Even though you have the same number of bricks, you can build a castle or a very wobbly tower.
These different shapes are what we call structural isomers. Think of them as molecular twins who look a bit different, or maybe even cousins who are only distantly related in their arrangement. They share the same ingredients but have distinctly different personalities. It's a bit like having the same amount of flour, sugar, and eggs, but making a cake versus a cookie. The outcome is pretty different.
The Straight-Laced One
n-Butyl Bromide
First up, we have the most straightforward one, the rule-follower of the bunch. This is n-butyl bromide. It's like the dependable friend who always takes the direct route.
Here, all four carbon atoms are lined up in a neat, unbroken chain. Think of it as a perfectly straight LEGO brick wall. The bromine atom is happily attached to one of the ends. It's chill, it's orderly, and it gets the job done without much fuss.
It’s the one you might think of first, the one that seems most "obvious." And honestly, there’s a certain comfort in its simplicity. No fancy twists or turns, just a good old-fashioned linear arrangement. It’s the vanilla ice cream of the C4H9Br family.
Its name, n-butyl bromide, tells you exactly what’s up. The "n" stands for "normal," which is basically code for "straight as an arrow." It’s the molecule that probably gets picked first for the most sensible tasks.

It might not be the most exciting, but it’s the foundation. It’s the baseline. It’s the molecule that says, "I’m here, and I’m perfectly fine being this way." And you know what? There’s nothing wrong with that. We all have our dependable moments.
The Slightly Branchy One
sec-Butyl Bromide
Now, let’s shake things up a little. Meet sec-butyl bromide. This one is a bit more of a planner, a strategist. It likes to have a central hub.
In this case, the carbon chain isn’t perfectly straight. It has a slight bend, a little detour. Specifically, the bromine atom is attached to a carbon atom that’s connected to two other carbon atoms. It’s like building a LEGO structure where a few bricks stick out from the main wall.
Think of it as the molecule that’s always looking for a shortcut, but in a slightly more complex way. It’s not a full-on detour, just a little zig-zag. The bromine atom is sitting on a carbon that’s part of the main chain, but it's not at the very end.
The "sec" in sec-butyl bromide is short for "secondary." This refers to the fact that the carbon atom holding the bromine is a secondary carbon – meaning it’s bonded to two other carbon atoms. It’s a subtle difference, but it makes all the difference in how this molecule behaves.
This isomer is a bit more reactive than its straight-laced cousin. It's like it's eager to get involved in things. It's the molecule that might be willing to try a new recipe, even if it's a little more complicated. It adds a bit of spice to the C4H9Br party.
It’s a good example of how a tiny change in structure can lead to different properties. This slight branching gives it a unique character, making it useful in different chemical reactions. It’s proof that sometimes, a little complexity can be a good thing.
The Truly Branchy One
isobutyl Bromide
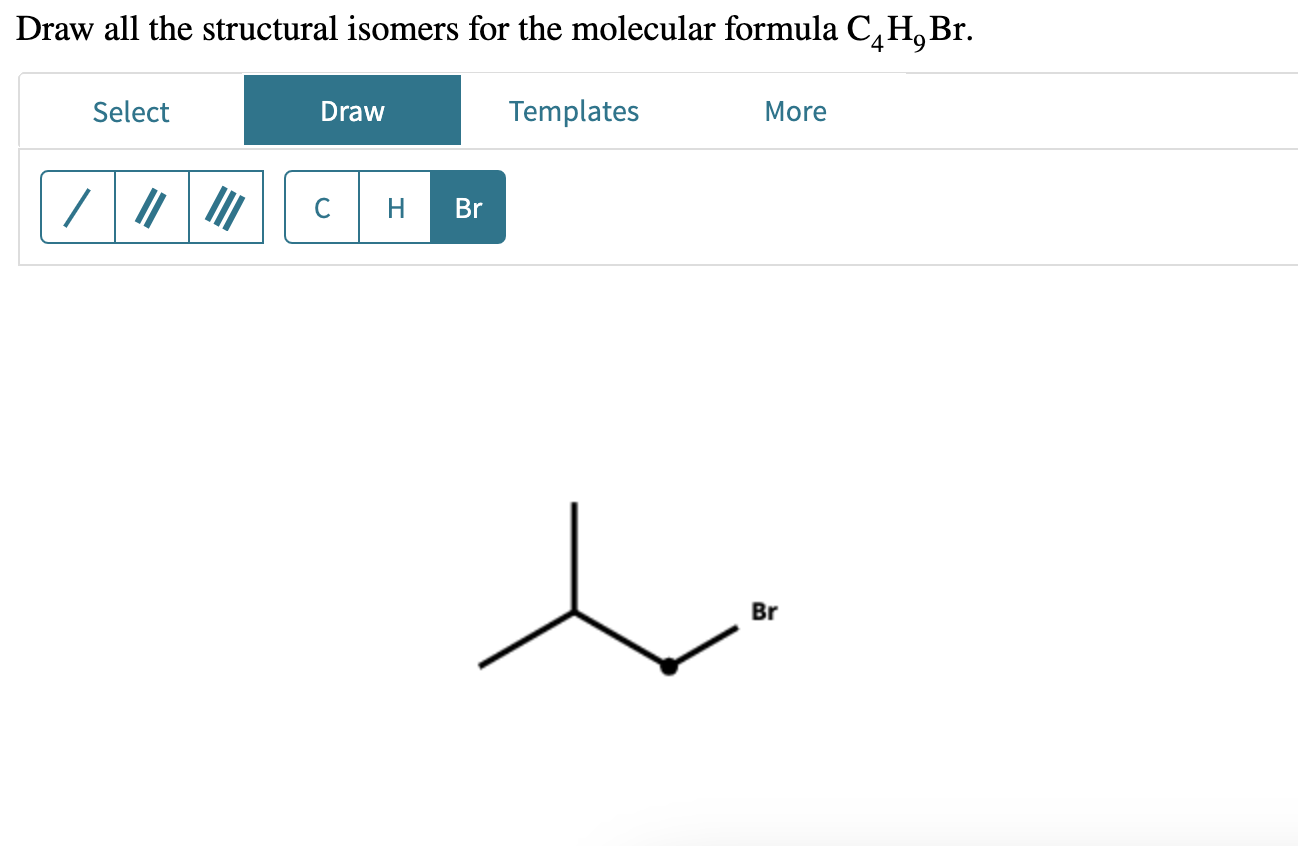
Next up, we have isobutyl bromide. This molecule is the free spirit, the one who likes to get creative with its structure. It’s definitely not afraid to branch out.
Here, one of the carbon atoms isn't in the main line at all. It's sticking off to the side, like a little bonus appendage. Imagine a LEGO structure with a few extra bricks attached to the side of the main wall, creating a more "bushy" look. The bromine is still attached to an end carbon, but the overall shape is much less linear.
It's like the molecule that decides to take the scenic route. It’s got a main chain, but then it has a side chain that makes it all a bit more compact and… well, branched. The bromine atom is attached to a carbon that’s at the end of a chain, but that chain is itself attached to another carbon that’s part of the main structure. It’s getting a little complex to visualize, right?

The "iso" prefix is a classic indicator of this type of branching. It signals that there’s a central carbon atom with two methyl groups (CH3) attached, and then the rest of the molecule extends from there. It's a specific pattern of branching.
Isobutyl bromide is another interesting character. Its slightly different arrangement gives it its own set of chemical properties. It’s the molecule that might be willing to try a more adventurous hobby. It’s not as straightforward as n-butyl bromide, and it’s definitely not the same as sec-butyl bromide.
This isomer highlights how dramatically the arrangement of atoms can change the molecule's identity. Even though it has the same atomic count, its unique shape makes it distinct. It’s like having the same set of ingredients but baking a muffin instead of a cake.
The Most Branched One
tert-Butyl Bromide
Finally, we arrive at the most heavily branched molecule of the bunch: tert-butyl bromide. This one is like the social butterfly of the C4H9Br family, with carbon atoms all around.
Here, the carbon atom directly attached to the bromine is connected to three other carbon atoms. That’s a lot of connections! Think of a central LEGO brick with three other LEGO bricks firmly attached to its sides. The bromine is dangling off that central, highly connected carbon.
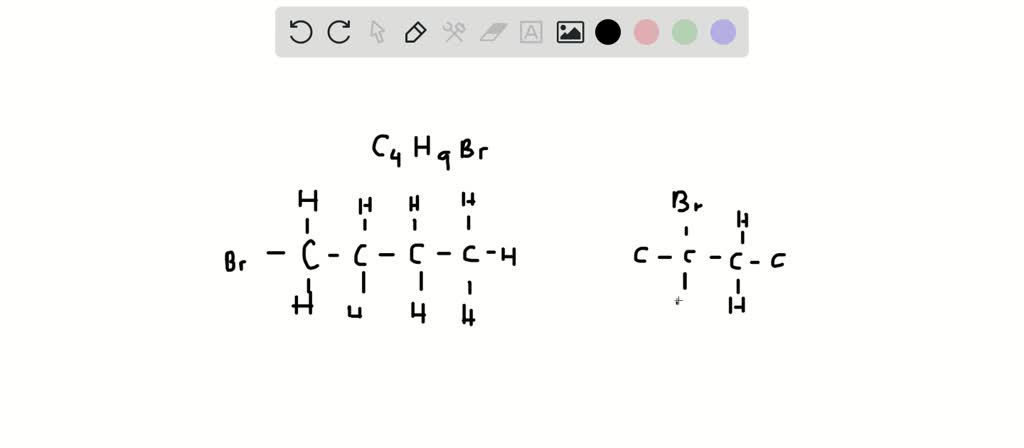
This is the ultimate in branching for our little C4H9Br group. The bromine atom is sitting on a carbon that is absolutely surrounded by other carbon atoms. It’s the most compact and branched arrangement possible for this formula.
The "tert" in tert-butyl bromide stands for "tertiary." This tells us that the carbon atom bonded to the bromine is a tertiary carbon – it's bonded to three other carbon atoms. This makes it quite special, and also quite reactive in certain ways.
This isomer is known for its unique reactivity due to this very crowded central carbon. It’s the molecule that might have a very specific set of friends it hangs out with. It’s the one that’s a bit more isolated from direct interaction due to its steric hindrance.
It’s fascinating how these four molecules, all with the same C4H9Br formula, can exist as completely different entities. They're like siblings who share the same parents but have vastly different life paths and personalities. And honestly, who can't relate to that? The world would be a lot less interesting if everything was just one single, identical shape.
And that, my friends, is the wonderful, sometimes bewildering, world of structural isomers. They remind us that sometimes, it’s not just what you have, but how you arrange it that truly defines you. So next time you see a molecule, remember: it might have hidden cousins.
