Strontium Hydroxide And Lithium Phosphate Complete Ionic Equation
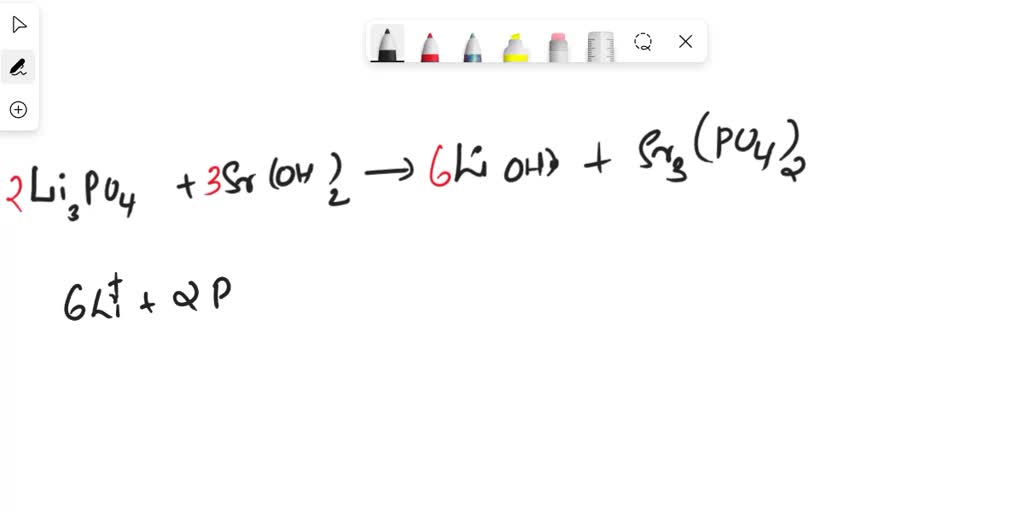
Ever wondered what happens when different chemical compounds mix? Sometimes, the results are straightforward, and other times, they reveal a hidden world of interactions. Today, we're diving into the fascinating world of strontium hydroxide and lithium phosphate, specifically looking at their complete ionic equation. Now, "complete ionic equation" might sound a bit intimidating, but think of it as a chemical detective story, revealing exactly which charged particles are present and how they rearrange. It’s a fun way to peek behind the curtain of chemical reactions and understand the fundamental building blocks of matter!
So, what’s the big deal about strontium hydroxide and lithium phosphate? Understanding their interaction, and specifically their complete ionic equation, helps us grasp stoichiometry (the quantitative relationships between reactants and products in chemical reactions) and the behavior of ions in solution. It's a core concept in chemistry that unlocks further understanding of more complex reactions. The benefit is a clearer picture of chemical transformations, which is crucial for anyone pursuing science, engineering, or even just curious about how things work at a molecular level.
While you might not be mixing these exact chemicals in your kitchen, the principles behind their ionic reactions are everywhere. Think about how detergents work – they rely on the interaction of ions to lift dirt. Or consider the processes involved in water treatment, where specific ions are added or removed to purify water. Even the batteries in our everyday devices operate on complex electrochemical reactions involving various ions. Learning about complete ionic equations, like the one for strontium hydroxide and lithium phosphate, builds a foundational understanding that connects to these practical applications.
In educational settings, this topic is often explored in high school and introductory college chemistry courses. Students learn to balance equations, identify electrolytes, and predict precipitation reactions. It’s a stepping stone to understanding more advanced chemical concepts and laboratory techniques.
Curious to explore this further? You don't need a full lab coat! You can start by looking up dissolubility rules for different ionic compounds. These rules tell you whether a compound will dissolve in water, which is key to forming ions. Then, you can find examples of chemical reactions online that involve hydroxides and phosphates. Many educational websites offer interactive simulations where you can "mix" virtual chemicals and see the results. It’s a safe and engaging way to experiment and build your intuition about chemical reactions. The journey into the world of ions might just spark a lifelong curiosity about the incredible science all around us!
