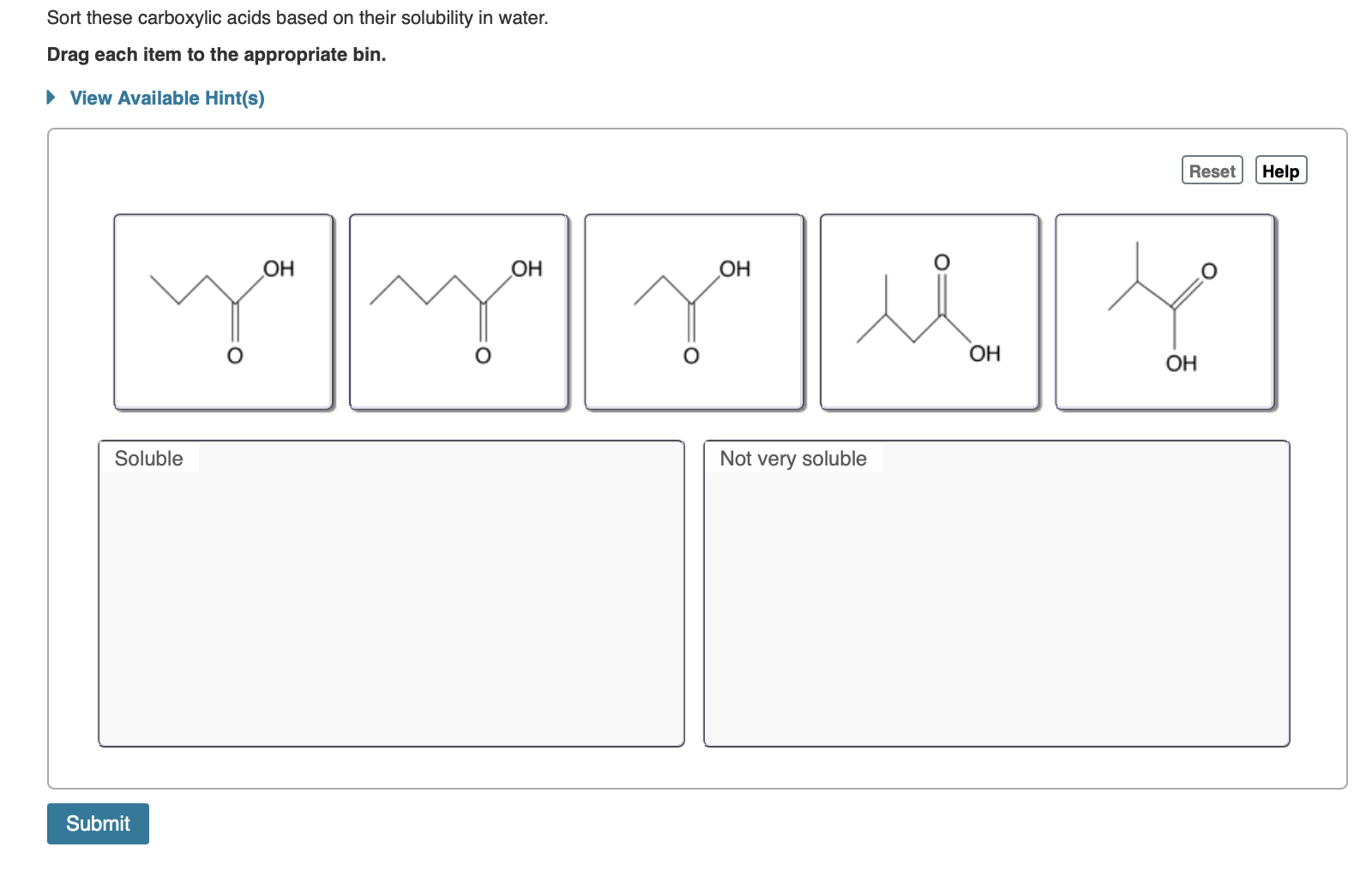
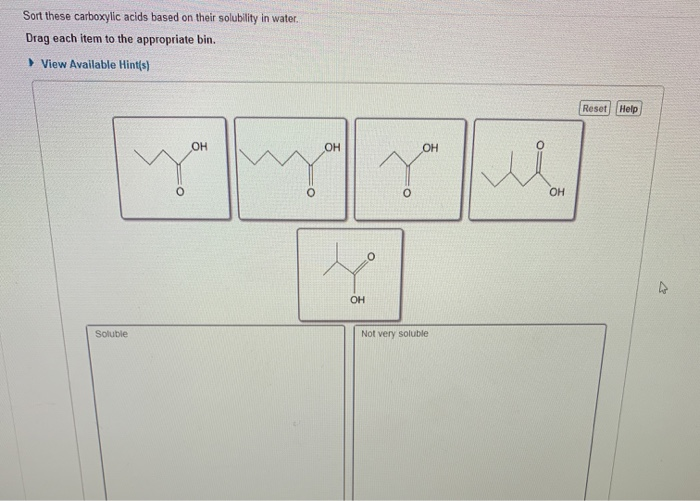
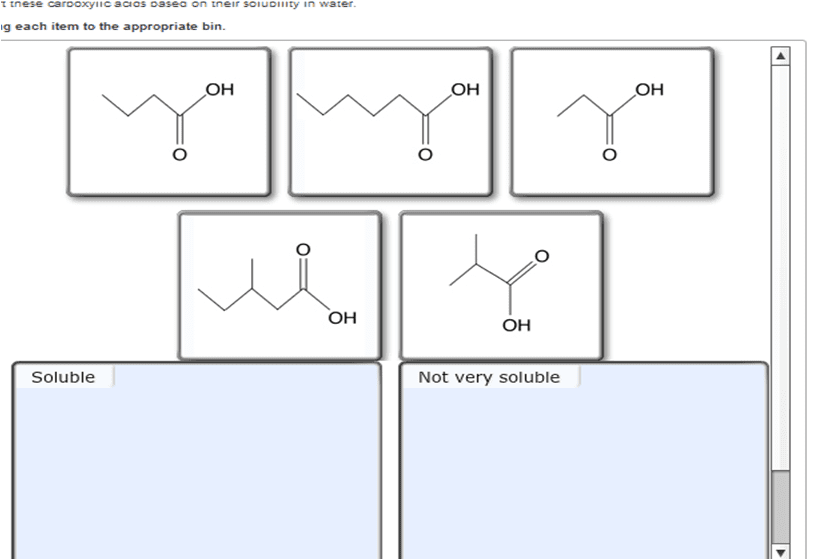
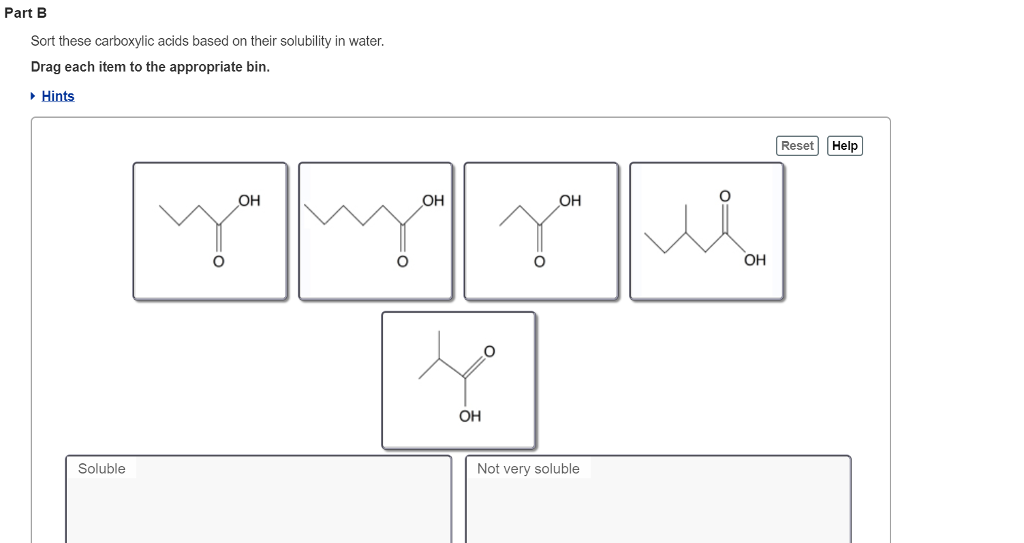
Sort These Carboxylic Acids Based On Their Solubility In Water
Ever looked at a bunch of scribbled-out chemical names and thought, "Wow, that looks like a puzzle!"? Well, guess what? We've got a fun little challenge for you today. It’s all about sorting some carboxylic acids based on how well they play with water. Think of it like a splashy sorting game for tiny molecules!
This isn't your average homework assignment, oh no. This is where the magic happens! We're talking about a little adventure into the world of chemistry, but don't worry, it's more like a treasure hunt than a pop quiz. You get to be the detective, figuring out which of these interesting compounds are total water lovers and which are a bit more standoffish.
Why is this so cool? Because it’s all about predicting behavior! You see, these carboxylic acids have personalities, and their personality determines how they interact with everyone else, especially good old H2O. It’s like knowing which friends will jump into the pool with you and which will politely watch from the poolside with a lemonade.
So, what exactly are these mysterious carboxylic acids? Imagine a little molecule with a special "head." This head has a carbon atom, an oxygen atom with a double bond, and another oxygen atom with a single bond, which is also happily attached to a hydrogen atom. That’s the secret sauce, the part that makes them acidic and gives them their unique water-loving (or not-so-water-loving) tendencies. It's like a molecular handshake with water!
Now, the fun really begins when we start looking at the "tail" of these molecules. This tail can be a tiny little thing, like just a few carbon atoms, or it can be a long, sprawling chain. And guess what? That tail is the key to our sorting game. It’s the difference between a molecule that dissolves easily and one that prefers to hang out with other oily molecules.
Think about it like this: water is like a big, friendly party. Some molecules are great dancers and easily join the crowd. Others are a bit shy, or maybe they’re more interested in talking to people who have similar interests. Our carboxylic acids, with their different tails, are those party guests.
The smaller the tail, the more likely our carboxylic acid is to be best buddies with water. Why? Because the "head" part of the molecule is a real water magnet. It’s polar, meaning it has a positive and a negative end, just like tiny magnets. Water molecules are also polar and love to stick to these charged ends. It’s a perfect match!
When the tail is short, the water-loving "head" can easily take over. It's like having a very enthusiastic host at a party who can draw everyone in. The whole molecule gets a warm welcome into the water. This is why small carboxylic acids are super soluble, meaning they dissolve almost completely.
But what happens when that tail gets long and fatty? Imagine a very long, non-polar chain attached to our polar head. This long tail is like someone who's more interested in chatting with other long-chain molecules. It’s not attracted to water at all. It's hydrophobic, which is a fancy word for "water-fearing."
When this hydrophobic tail gets too big, it starts to outweigh the water-loving power of the head. The water molecules can't quite pull the whole thing into their embrace. Instead, the long tail tries to avoid the water, and the molecule doesn't dissolve very well. It’s like trying to mix oil and water – they just don't want to be friends.
So, our sorting challenge is all about recognizing the size of this tail. We're going to present you with a list of carboxylic acids. Some will have very short tails, and these will be our top-tier water-lovers. Others will have progressively longer tails, and they will become less and less soluble.
Let's talk about some of the players. You might see something like formic acid. This little guy has no carbon tail at all! It's just a hydrogen atom attached to our acid group. This makes it incredibly soluble. It’s like the life of the party, diving right in!
Then there’s acetic acid, the star of vinegar. Its tail is just one carbon atom. Still pretty small, so it's very happy in water. It’s like the friendly neighbor who’s always up for a chat and a dip in the pool.
As we go up the chain, we encounter propanoic acid (three carbons), butanoic acid (four carbons), and so on. Each extra carbon in the tail makes the molecule a little less keen on water. It’s a gradual shift, a slow goodbye to full solubility.
You'll see names like hexanoic acid, octanoic acid, and then we get into the really long ones, like lauric acid or palmitic acid. These have many, many carbon atoms in their tails. By the time you get to these longer chains, the molecules are practically swimming in their own little oil slicks, barely mixing with water.
The beauty of this exercise is that you don't need a chemistry lab. All you need is a keen eye and a bit of logic. You're using your brain to predict molecular behavior. It’s like a brain teaser with real-world implications!
Think about where these carboxylic acids pop up. Formic acid is found in ant stings. Acetic acid is in your kitchen. Longer chain fatty acids are what make up fats and oils in your food and in your body. Understanding their solubility helps us understand how they behave in biological systems and in industrial processes.
So, the task is simple: take the list of carboxylic acids provided. Identify which ones have the shortest carbon chains (meaning the most water-soluble) and which have the longest (meaning the least water-soluble). Arrange them from most soluble to least soluble.
Ready for the challenge?
We’re talking about a spectrum here. On one end, you have molecules that disappear into water like magic. On the other, you have molecules that prefer to form a separate layer, like an oily sheen on a puddle. It’s a fascinating gradient of solubility.
Why is this so engaging? Because it’s a puzzle you can solve with a little bit of thinking. It’s about recognizing patterns. The pattern here is: short tail = water lover, long tail = water shy. It’s elegant in its simplicity, yet it explains a lot about chemical interactions.
It’s special because it highlights how the size of a molecule can dramatically change its properties. Even a few extra atoms can make a big difference in how something behaves. It’s a tiny detail with a huge impact!
Imagine you're a chef, and you need to mix ingredients. Knowing which acids will blend smoothly into your water-based sauce and which will float on top is crucial, right? This kind of knowledge is fundamental.
So, dive in! Look at the names. Count those carbons in the tails. Mentally picture those molecules. Are they tiny and eager to mingle with water, or are they long and perhaps a bit more exclusive? This sorting task is a playful introduction to the fundamental concept of solubility, driven by molecular structure.
It’s a chance to feel smart, to unravel a small mystery of the chemical world. You’re not just memorizing facts; you’re understanding why things happen. And that, my friends, is the most rewarding part of any learning adventure.
So, get ready to sort! Let the water-loving molecules shine through, and let the hydrophobic ones demonstrate their unique charm. It’s a splashy, insightful, and surprisingly fun journey into the heart of carboxylic acids and their dance with water!
