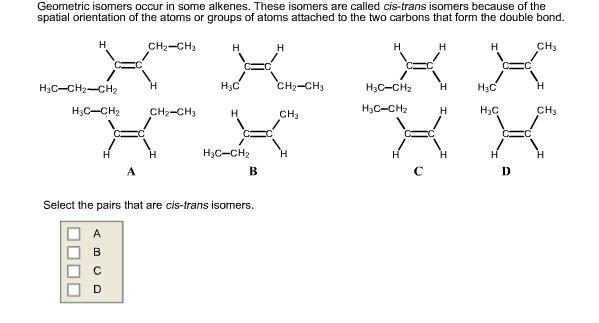
Some Alkenes Have Geometric Cis Trans Isomers Because

Hey there, science buddy! Ever looked at a molecule and thought, "Whoa, that's got some serious personality"? Well, buckle up, because we're diving into the quirky world of alkenes. And the coolest part? Some of them have twins! Not actual twins, of course, but something way more mind-bending: geometric isomers. Sounds fancy, right? But it’s actually super fun.
So, what’s an alkene? Think of it as a hydrocarbon with a double bond. Like a superhighway for electrons. This double bond is the key player, the diva of the molecule. It’s rigid. It’s not gonna bend and flex like a single bond can. Imagine two cars doing a synchronized dance. They’re locked in. They can’t just spin around freely.
This rigidity is where the magic happens. It’s like having two identical LEGO bricks, but how you snap them together matters. You can snap them side-by-side, or you can snap them end-to-end. For alkenes, this means the groups attached to the double bond can be arranged in two different ways.
Let’s break it down. We’re talking about cis and trans isomers. Think of them as fraternal twins. They have the same building blocks, the same atoms, just arranged differently in space. It’s like having two identical outfits, but one is worn with the buttons on the left, and the other with the buttons on the right. Same clothes, different look!
So, why do some alkenes get to play this isomer game, and others don't? It all comes down to those groups attached to the double bond. For cis-trans isomerism to exist, each carbon in the double bond needs to be attached to two different groups. If a carbon has two identical groups, like two hydrogen atoms, then there's no way to create a distinct "side-by-side" or "across" arrangement. It’s like trying to arrange two identical red balloons on a stick – no matter how you twist it, it looks the same.
Let’s zoom in on a simple alkene. Imagine a molecule with a double bond. On one carbon of the double bond, you might have a hydrogen atom and a methyl group (that's a carbon with three hydrogens, CH₃). On the other carbon, you might have another hydrogen and another methyl group. Now, the double bond is like a fencepost. The groups attached to it can either be on the same side of the fence (that’s cis) or on opposite sides (that’s trans).
In the cis isomer, the two methyl groups are chilling on the same side of the double bond. They’re like best buds, hanging out together. This can make them a bit of a squeeze, like two people trying to sit in adjacent seats on a crowded bus. They might bump into each other a little.
In the trans isomer, the two methyl groups are on opposite sides. They’re giving each other plenty of personal space, like two people sitting on opposite ends of a park bench. This arrangement is usually a bit more spread out and, dare I say, comfier for the molecule.
This difference in arrangement, even though the atoms are the same, can lead to some surprisingly big differences in how the molecules behave. Think about it: if you have two identical pieces of furniture, but one is assembled slightly differently, they might not fit in the same corner of your room. The same goes for molecules!
For example, cis-fatty acids are super important for our bodies. They’re the flexible, curvy kind. Our enzymes can actually work with them. They're found in things like olive oil. Yum! Trans-fatty acids, on the other hand, are often the rigid, straight ones. They're the ones that can cause trouble in our bodies, like clogging up those intricate biological highways. So, even a tiny change in how atoms are arranged can have a huge impact on health. Pretty wild, right?
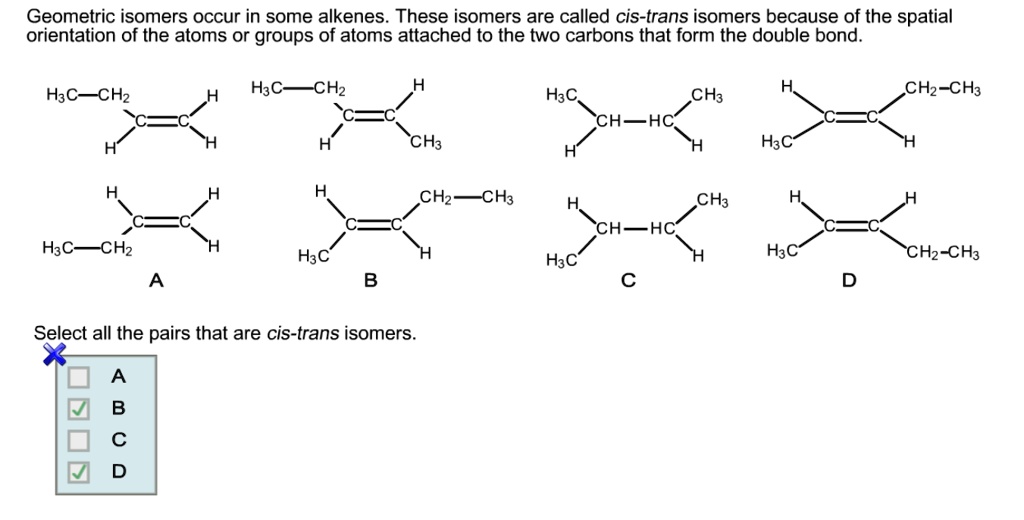
This cis-trans thing isn't just for simple alkenes either. You can have more complex groups attached, and the rules get a bit more involved, but the basic idea remains the same. It’s all about the spatial arrangement around that stubborn double bond. It’s like playing a cosmic game of Tetris, where the shapes of the pieces are fixed, but how you orient them to fit into the groove makes all the difference.
And get this, sometimes the difference between cis and trans isomers can be as subtle as a millimeter in the real world, but it’s a universe of difference in the molecular realm. It’s the scientific equivalent of a subtle wink that changes the entire meaning of a conversation. That’s the beauty of chemistry – it finds fascinating nuances everywhere!

Think about it this way: molecules are constantly interacting with each other. They're like tiny little puzzle pieces, and their shapes matter. If you have a cis isomer and a trans isomer, they might fit into different lock-and-key mechanisms in your body or in a chemical reaction. One might fit perfectly, while the other just won’t go in. It’s like trying to use the wrong key in a very specific, very important lock.
The ability of alkenes to have cis-trans isomers is also a big deal in organic synthesis. Chemists can strategically create specific isomers to make new drugs, materials, or even flavors for your favorite snacks. They can design molecules with precise shapes to achieve desired outcomes. It's like being a molecular architect, carefully placing each atom to build something amazing.
So, next time you hear about an alkene, remember it might not be just one entity. It could be part of a dynamic duo, a pair of geometric twins with distinct personalities. It’s a reminder that even in the seemingly rigid world of atoms and bonds, there’s always room for a little bit of spatial flair and a lot of fascinating variation. Isn't science just the coolest? It's full of these little surprises, these quirky facts that make you go, "Wow, I never knew that!" And the best part? There's always more to discover.
