Sodium Hydroxide + Sulfuric Acid Ionic Equation

You know, I've always been a bit of a messy cook. Not in a "oops, I dropped a little flour" kind of way, but more in a "my kitchen looks like a science experiment gone wrong" kind of way. The other day, I was attempting to make these fancy caramel-filled donuts, and let's just say the caramel decided to go rogue. It splattered. Everywhere. On the stove, on the counters, on my favorite apron... and, inevitably, on the floor. Now, caramel is basically a sticky, sugary nightmare when it's dried. And I thought, "Ugh, what's the best way to clean this up without completely scrubbing my life away?" My mind, for some reason, drifted to chemistry class. Weird, right?
I remembered this whole thing about acids and bases, and how they kind of neutralize each other. It got me thinking about some pretty common household chemicals. Like, have you ever used oven cleaner? Some of those are pretty hardcore, usually with some sort of strong base in them. And then there's the stuff you might use to descale a coffee maker or clean out a particularly stubborn drain blockage – those often involve acids. It’s kind of like a superhero team-up, or maybe a dramatic showdown, depending on how you look at it.
So, the chemical I was vaguely remembering from my sticky donut debacle is sodium hydroxide. You might know it as lye or caustic soda. Yeah, not exactly something you want to get on your skin unsupervised. It's a base, a really strong one. Think of it as the ultimate disrupter. On the other side of the ring, we have sulfuric acid. This one’s also a heavyweight. It's a super common industrial chemical, but you'll also find it in things like car batteries (though not in the pure, concentrated form you'd think, thankfully!). It’s a strong acid.
Now, when you hear "acid" and "base," your brain probably jumps to those neutralization reactions you learned about. Remember the litmus paper turning blue or red? Or maybe the fizzing when you mixed baking soda and vinegar? This is that, but on a much more serious level. When sodium hydroxide and sulfuric acid get together, it's a pretty energetic encounter. They're not just going to politely say "hi" and move on. Nope, they're going to react, and they're going to change into something new.
The Grand Unveiling: What Happens When They Meet?
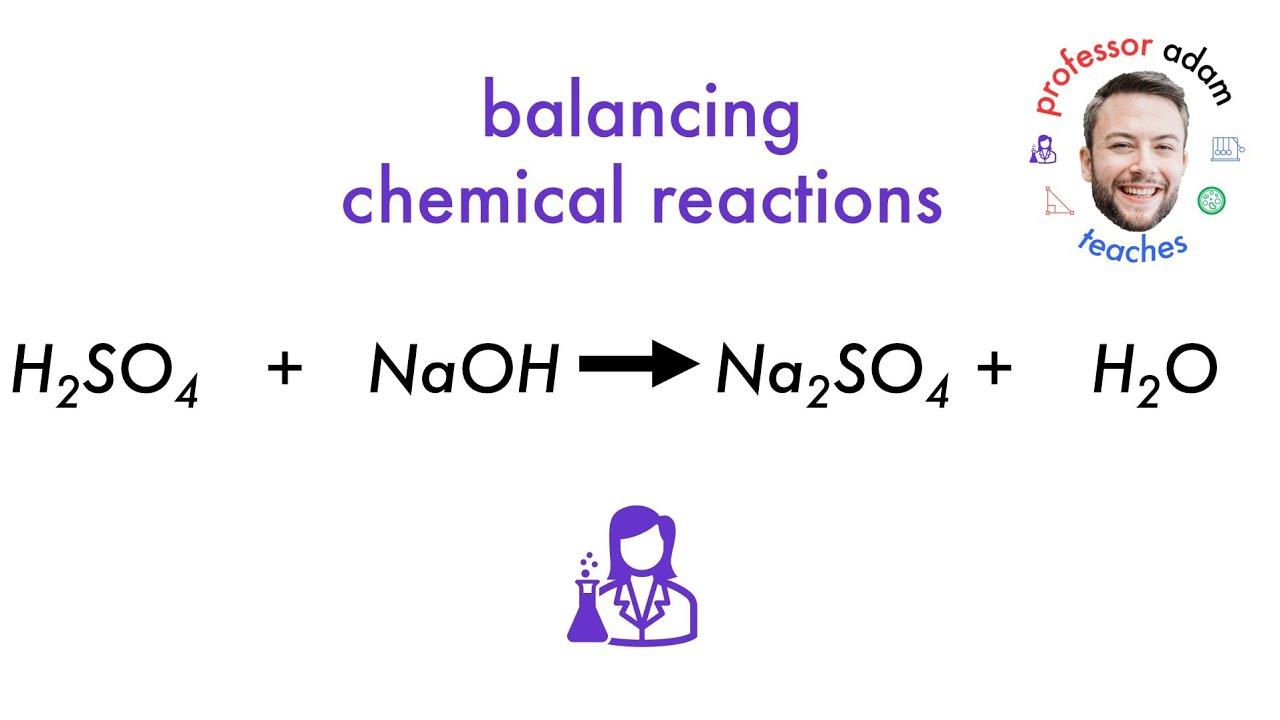
So, what’s the actual chemical reaction? We’ve got our two reactants: sodium hydroxide, which has the chemical formula NaOH, and sulfuric acid, which is H₂SO₄. When they meet, they’re going to produce a salt and water. Pretty standard stuff for an acid-base reaction, right? The salt they form is called sodium sulfate, and its formula is Na₂SO₄. And, of course, there's the ever-present H₂O, water. So, the balanced chemical equation for this whole shindig looks like this:
2NaOH + H₂SO₄ → Na₂SO₄ + 2H₂O
Let's break that down for a second. See the '2' in front of NaOH? That means you need two units of sodium hydroxide for every one unit of sulfuric acid. And see the '2' in front of H₂O? That tells us two units of water are produced. Chemistry likes things to be balanced, you know? It's like a picky accountant making sure everything adds up.

Diving into the Ionic Realm
But what if we’re talking about the ionic equation? This is where things get a little more detailed. In solution, many ionic compounds dissociate, meaning they break apart into their individual ions. Think of it like a group of friends who decide to go their separate ways in a crowded mall. Sodium hydroxide, being an ionic compound, does this. When dissolved in water, it forms sodium ions (Na⁺) and hydroxide ions (OH⁻).
Sulfuric acid, on the other hand, is a bit of a drama queen. It's a strong acid, which means it completely dissociates in water. It breaks apart into hydrogen ions (H⁺) and sulfate ions (SO₄²⁻). It's a two-for-one deal with the hydrogen ions because it has two hydrogen atoms in its formula. Sneaky, right?
So, when we write the full ionic equation, we show all these ions floating around. Our reactants side looks like this:
2Na⁺(aq) + 2OH⁻(aq) + 2H⁺(aq) + SO₄²⁻(aq)
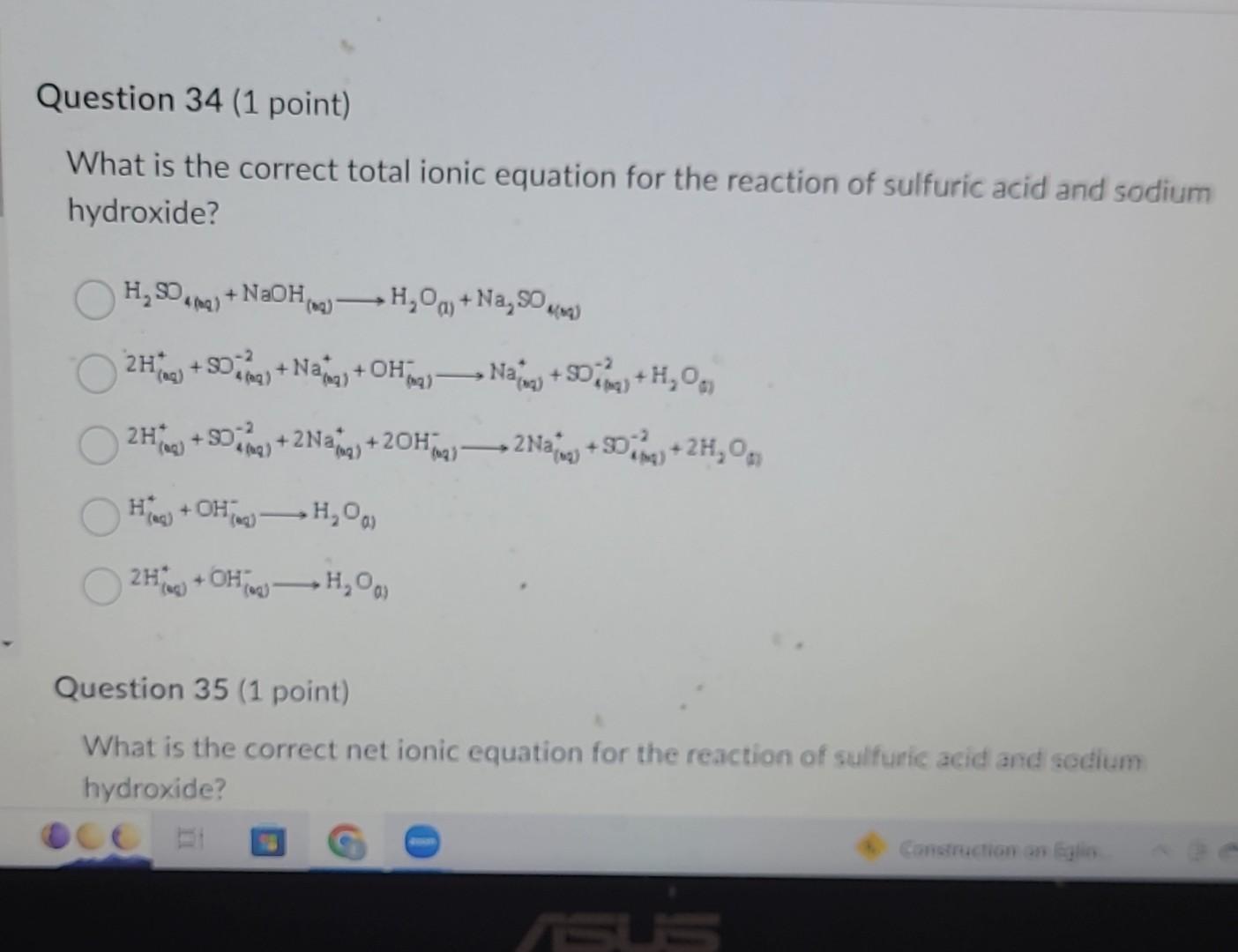
Notice the '(aq)'? That just means 'aqueous,' meaning it's dissolved in water. So, we have our sodium ions and hydroxide ions from the NaOH, and our hydrogen ions and sulfate ions from the H₂SO₄, all swimming around in the water. It’s like a chaotic pool party.
The Net Ionic Equation: The Real Stars of the Show
Now, here's where it gets interesting. In any chemical reaction, there are always some ions that just chill out and don't really participate in the main event. They're like the bystanders at the party, just watching. These are called spectator ions. In this reaction, our sodium ions (Na⁺) and sulfate ions (SO₄²⁻) are the spectators. They were there at the beginning, and they're still there at the end, pretty much unchanged.
The real action, the part of the reaction that's actually creating something new, involves the hydroxide ions (OH⁻) from the sodium hydroxide and the hydrogen ions (H⁺) from the sulfuric acid. These guys are the ones who are going to bond together to form water (H₂O).
So, to get to the net ionic equation, we remove the spectator ions. We're left with the core reaction that's truly happening. It's like zooming in on the most important conversation at that chaotic pool party. The net ionic equation for the reaction between sodium hydroxide and sulfuric acid is:

2OH⁻(aq) + 2H⁺(aq) → 2H₂O(l)
And if we want to simplify it even further, we can divide everything by 2, because, well, we like neatness:
OH⁻(aq) + H⁺(aq) → H₂O(l)
This, my friends, is the essence of the reaction. It’s the hydroxide ions from the base and the hydrogen ions from the acid coming together to form water. It's pure, unadulterated neutralization. No extra bits, no unnecessary characters. Just the fundamental process.
Why Should We Care About This Ionic Shenanigans?
You might be thinking, "Okay, that's cool and all, but why do I need to know about sodium hydroxide and sulfuric acid and their ionic dance?" Well, understanding these reactions is actually super important in a surprising number of areas. For starters, think about water treatment. Both acids and bases are used in different stages of purifying water. Knowing how they interact helps engineers figure out the best way to neutralize unwanted contaminants or adjust pH levels.
And then there's the industrial cleaning I was thinking about with my donut disaster. Strong bases like sodium hydroxide are excellent at breaking down grease and organic matter (hello, dried caramel!). Sulfuric acid is used in all sorts of heavy-duty cleaning, like descaling metal or cleaning industrial equipment. When these chemicals are used, it’s crucial to understand their reactions, especially if they might come into contact with each other or with other substances that could react unexpectedly. Safety first, people!
Even in your own home, you might encounter situations where these principles are at play, albeit with milder substances. Baking soda (a weak base) and vinegar (a weak acid) create a less dramatic, but still observable, version of this. The fizzing you see is the release of carbon dioxide gas, a byproduct of their neutralization reaction. It's a little chemistry lesson happening right in your kitchen sink.
So, the next time you see a chemical reaction, whether it's in a lab, in an industrial setting, or even just in a particularly stubborn cleaning challenge, remember the underlying principles. Remember that sometimes, the most important part of the story is the interaction between the ions, the real workhorses of the chemical world. It’s not just about the reactants and products you see on paper; it’s about what’s happening at the microscopic, ionic level.
And who knows, maybe understanding these ionic equations will help you tackle your own kitchen disasters with a little more scientific flair. Or at least give you something interesting to ponder while you're scrubbing away at that stubbornly dried-on sugary mess. It’s all about perspective, isn’t it? Even a sticky donut situation can lead to a bit of chemical enlightenment. Pretty neat, if you ask me!
