Sodium Hydroxide And Sulfuric Acid Net Ionic Equation
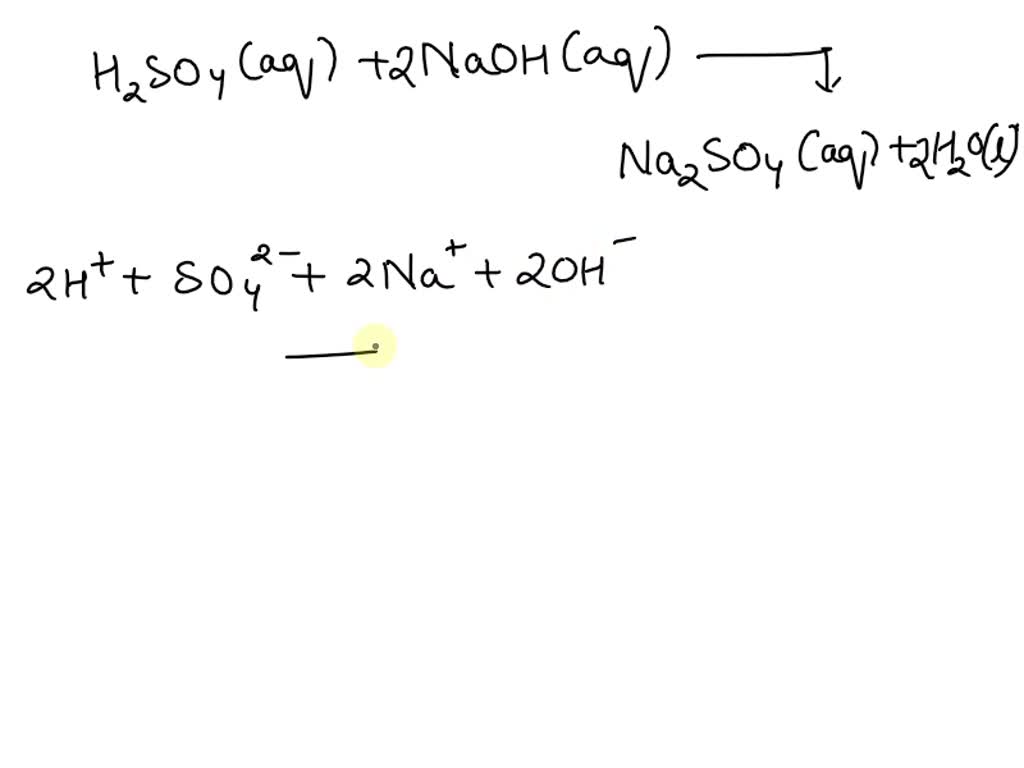
Hey there, fellow science curious folks! Grab your mug, settle in, 'cause we're about to dive into something that sounds super fancy, but is actually kinda cool, once you get the hang of it. We're talking about the dramatic showdown between sodium hydroxide and sulfuric acid. Yep, these two are like the ultimate frenemies of the chemistry world, and today, we're going to break down their "net ionic equation." Don't worry, it's way less scary than it sounds. Think of it like a movie plot, but with atoms and ions! Isn't that neat?
So, first things first, what are these guys? Sodium hydroxide, that's NaOH, right? Super common stuff. You might know it as lye, or caustic soda. It's a pretty strong base, meaning it's really good at accepting little protons. It's also, you know, a little bit dangerous if you're not careful. Don't go splashing it around, seriously. Think of it as a powerful cleaning agent, but with a bit of an attitude. It likes to break things down, in a chemical sense, of course. And it's usually a solid at room temp, but when we throw it in water, which we totally do in these reactions, it dissolves like a dream.
Then we have sulfuric acid, H₂SO₄. Oh boy, this one's a heavyweight. It's one of the most important industrial chemicals out there. Seriously, they make tons of it. It's a super strong acid. What does that mean? It's super eager to give away those little protons. And it's also, uh, really corrosive. Like, "don't breathe the fumes" corrosive. Handle with extreme caution is basically its motto. It's a powerful dehydrating agent too – it can literally suck the water out of things! Imagine that. It's like the chemistry equivalent of a super-absorbent sponge, but for water and with a burning sensation. Yikes.
Now, when these two powerhouses meet, what happens? It's a classic acid-base reaction, folks! Acids and bases love to react with each other. It's almost like they're drawn to it. Think of it like a dance, a very energetic, potentially explosive dance. The acid, with its eagerness to donate protons, and the base, with its eagerness to accept them, are going to have a little party. And what's the outcome of this party? Usually, you get a salt and water. It's like the ultimate neutral outcome, right? The two extreme personalities find common ground and produce something stable and, well, neutral. Science is so poetic sometimes, isn't it?
So, let's write out the whole shebang, the molecular equation. This is like the full cast and crew of our chemistry movie. We've got our sodium hydroxide, NaOH, and our sulfuric acid, H₂SO₄. They're going to bump into each other. And the products? We get sodium sulfate, Na₂SO₄, and water, H₂O. So the equation looks like this: NaOH + H₂SO₄ → Na₂SO₄ + H₂O. But wait! It's not quite balanced yet, is it? Chemistry demands balance, like a perfectly curated Instagram feed. So, we need two molecules of NaOH to tango with one molecule of H₂SO₄ to make one molecule of Na₂SO₄ and two molecules of H₂O. The balanced molecular equation is: 2 NaOH + H₂SO₄ → Na₂SO₄ + 2 H₂O. See? Everything's accounted for. No atoms left behind. Very responsible.
Okay, so that's the molecular equation. It tells us what the reactants and products are. But in solution, a lot of these ionic compounds, they don't really exist as whole molecules. They break apart into their individual ions. It's like when you invite a bunch of friends over, and they all split up to do their own thing. Sodium hydroxide, when it's dissolved in water, it dissociates. That means it breaks into its positively charged sodium ions (Na⁺) and its negatively charged hydroxide ions (OH⁻). So, our NaOH becomes Na⁺(aq) + OH⁻(aq). The little "(aq)" just means it's dissolved in water, aqueous solution, fancy talk. It's like saying "these guys are hanging out in the pool."
Now, sulfuric acid is a bit of a drama queen. It's a strong acid, remember? It's so strong, it can donate two protons. So, when it dissolves in water, it completely dissociates. First, it loses one proton to become a hydrogen sulfate ion (HSO₄⁻). So, H₂SO₄ → H⁺(aq) + HSO₄⁻(aq). But then, that hydrogen sulfate ion, it's still a bit acidic, so it can also lose its remaining proton, though not quite as readily as the first one. It can dissociate further to form another hydrogen ion (H⁺) and a sulfate ion (SO₄²⁻). So, HSO₄⁻ ⇌ H⁺(aq) + SO₄²⁻(aq). For the sake of simplicity in a net ionic equation, we often treat strong acids as fully dissociating into their most basic ions. So, for our purposes today, we're going to say sulfuric acid completely breaks down into 2 H⁺(aq) + SO₄²⁻(aq). It's like it's really emptying its pockets of all its protons. A generous, if slightly reckless, giver of protons.

So, our sodium sulfate, Na₂SO₄? That's also an ionic compound. When it's formed and dissolved in water, it's not going to hang around as a whole Na₂SO₄ molecule. It's going to break apart into its constituent ions: two sodium ions (Na⁺) and one sulfate ion (SO₄²⁻). So, Na₂SO₄ becomes 2 Na⁺(aq) + SO₄²⁻(aq). It's like the salt just dissolves and its ions go their separate ways, mingling with the water molecules. They're still there, you just can't see them as a solid chunk anymore.
And water, H₂O? Water is water. It's the solvent, the medium for all this action. It doesn't really break down into ions in this context. It's the party venue, not a guest who splits up. So, it just stays as H₂O. Pretty stable stuff, water. Always there for us. A true constant in the chaotic world of chemistry.
Now, let's put all of that together into what we call the complete ionic equation. This is like showing all the dissolved ions in our reaction mixture. It's like taking a snapshot of the whole party, with everyone mingling. Using our balanced molecular equation and our dissociated ions, we get:
2 Na⁺(aq) + 2 OH⁻(aq) + 2 H⁺(aq) + SO₄²⁻(aq) → 2 Na⁺(aq) + SO₄²⁻(aq) + 2 H₂O

Whoa, look at that! It's a whole lot of ions bouncing around, isn't it? It's like a crowded dance floor. You've got your sodium ions, your hydroxide ions, your hydrogen ions, and your sulfate ions, all in the same aqueous solution. And then, poof, they rearrange a bit to form the sodium sulfate and water. It's a chemical ballet.
But here's where the "net ionic equation" magic happens. We're looking for what actually changes during the reaction. What are the main players that are actively participating in the chemical transformation? We want to get rid of the spectator ions. These are the ions that are present at the beginning and at the end of the reaction, but they don't actually do anything. They're just chilling, watching the action unfold. They're the audience members, not the main actors.
In our complete ionic equation, can you spot them? Look at the left side and the right side. Do you see any ions that appear exactly the same on both sides? Yep! We have 2 Na⁺(aq) on the left and 2 Na⁺(aq) on the right. They're identical. They didn't participate in the formation of new bonds or the breaking of old ones in a significant way. They're just along for the ride. So, we're going to cross them out. Buh-bye, sodium ions! You've been great spectators.
What else do we see that's the same on both sides? How about the sulfate ion? We have SO₄²⁻(aq) on the left and SO₄²⁻(aq) on the right. Again, identical. They started out as separate ions and they ended up as separate ions. They didn't get married to anything else or get broken up. They're just watching. So, we're going to cross them out too. See ya, sulfate! Thanks for coming.
Now, what's left? What are the ions that did change? On the left, we have 2 OH⁻(aq) and 2 H⁺(aq). And on the right, we have 2 H₂O. These are the actual participants in the reaction. The hydroxide ions and the hydrogen ions are coming together to form water. This is the core of the acid-base neutralization! It's the heart of the chemical drama. These are the stars of the show. The leading actors. The ones who get all the lines.
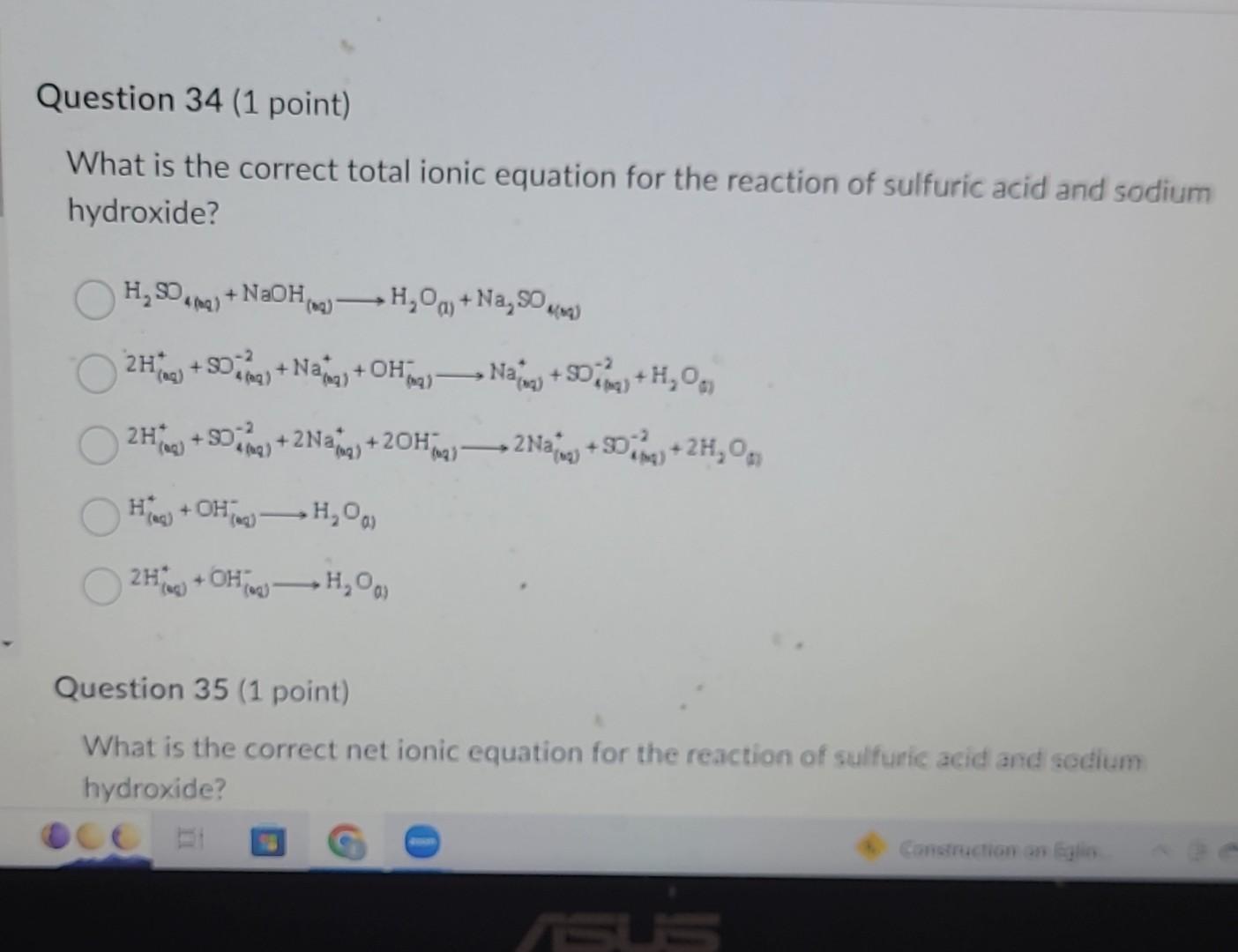
So, our net ionic equation is what remains after we've crossed out those spectator ions. It shows only the species that are directly involved in the chemical change. And in this case, it's:
2 OH⁻(aq) + 2 H⁺(aq) → 2 H₂O
And we can simplify this even further, can't we? We have a '2' in front of every single species. That means we can divide everything by 2. So, the most simplified net ionic equation is:
OH⁻(aq) + H⁺(aq) → H₂O

Ta-da! Isn't that just so much cleaner? It's like decluttering your room and only keeping the essential, most beautiful items. This equation tells us the fundamental truth of what happens when a strong base (like hydroxide ions) reacts with a strong acid (like hydrogen ions) in water. It's the essence of neutralization. It's the pure, unadulterated reaction. Just ions doing their thing, forming water. Simple, yet profound. Like a perfectly brewed cup of coffee, it's just right.
So, to recap, we started with the big picture, the molecular equation. Then we broke it down into all the individual ions in the complete ionic equation. And finally, we stripped away the bystanders, the spectator ions, to reveal the core chemical event in the net ionic equation. It's a process of peeling back the layers, like an onion, but hopefully with fewer tears involved. Unless you're actually working with these chemicals without proper safety gear, in which case, yes, tears. Lots of tears. Safety first, friends!
Why is this whole net ionic equation thing so important? Well, it highlights the actual chemical change that's happening, regardless of what else is floating around in the solution. It shows us that the reaction between sodium hydroxide and sulfuric acid is fundamentally the same as the reaction between any strong base containing hydroxide ions and any strong acid containing hydrogen ions. The sodium ions and sulfate ions are just hanging out, doing their own ionic thing. The real action, the real chemistry, is between the OH⁻ and the H⁺. It's all about the proton transfer, the essence of acid-base chemistry.
Think about it! This concept applies to so many other reactions too. If you reacted potassium hydroxide (KOH) with hydrochloric acid (HCl), what would you expect to get? You'd get potassium chloride (KCl) and water. The molecular equation would be KOH + HCl → KCl + H₂O. If you break down the ions, you'd have K⁺, OH⁻, H⁺, Cl⁻ on the reactant side, and K⁺, Cl⁻, H₂O on the product side. The spectator ions would be K⁺ and Cl⁻. And the net ionic equation? You guessed it: OH⁻(aq) + H⁺(aq) → H₂O. Exactly the same! It's like a universal solvent for understanding acid-base reactions. How cool is that? Chemistry just keeps on giving.
So, next time you hear about sodium hydroxide and sulfuric acid, you can nod sagely and think, "Ah yes, the dramatic ionic dance that ends with water." And you'll know that the net ionic equation, OH⁻(aq) + H⁺(aq) → H₂O, is the true star of the show. It's the simplified, elegant representation of this fundamental chemical transformation. It's the beauty of chemistry, really. Taking something that seems complex and finding the simple, elegant truth beneath it all. It's like finding the perfect little doodle on a napkin that perfectly sums up a whole conversation. Now, who wants another coffee?
