Sodium Carbonate And Sulfuric Acid Net Ionic Equation

Hey there, chemical adventurers! Ever feel like your kitchen sink is staging a full-blown science experiment without your permission? You know, that moment when you dump a bunch of baking soda down there after trying to clear a stubborn clog, and then, whoosh, it starts fizzing like it's trying to escape to a tropical island? Well, buckle up, buttercups, because we're about to dive into a chemical reaction that’s like the more dramatic, slightly more potent cousin of that baking soda saga: sodium carbonate and sulfuric acid. Think of it as the "full-on spa day for your drainpipes, but with a little more oomph."
Now, I know what you're thinking. "Sulfuric acid? Isn't that what they use in car batteries? Am I going to accidentally dissolve my toaster?" Relax! We're not talking about the super-concentrated, evil-scientist-grade stuff here. We're talking about the everyday, slightly-less-terrifying version that sometimes hangs out in places you might not expect. It’s like that one relative who’s a little eccentric but mostly harmless… until they start telling that story at Thanksgiving dinner.
Let's break down our main players. First up, we have sodium carbonate. You might know this guy better as washing soda or soda ash. It’s that trusty sidekick that helps your laundry detergent get out those grass stains that are practically tattooed onto your kid’s pants. It’s a salt, but a pretty friendly one. Think of it as the reliable friend who always brings snacks to the party. It’s got a little sparkle, it’s good at its job, and it doesn't cause too much fuss. Sodium carbonate is basically the unsung hero of sparkling clean surfaces, making your dishes gleam and your laundry smell like a fresh meadow… or at least, less like a gym sock.
On the other side of the chemical ring, we have sulfuric acid. Now, this one’s a bit more… intense. It’s a strong acid, which means it's eager to give away those little positively charged hydrogen bits (protons, for you fancy folks). It’s like that friend who’s always got a million ideas and is super enthusiastic, sometimes a little too enthusiastic. Sulfuric acid is a workhorse in industry, from making fertilizers to refining petroleum. It’s the ultimate problem-solver, but you gotta handle it with respect. Imagine it as the super-charged espresso shot of the chemical world. It gets things done, but maybe don't chug it straight.
So, what happens when these two get together? It’s like a chemical mosh pit! When you mix an acid (sulfuric acid) with a base or a carbonate (sodium carbonate), things get lively. Remember that baking soda and vinegar volcano you made in third grade? This is kind of like that, but with more… sophistication. Instead of baking soda (sodium bicarbonate), we’re using washing soda (sodium carbonate). And instead of vinegar (acetic acid), we’re stepping it up with sulfuric acid.
The whole point of a net ionic equation, my friends, is to cut through all the fluff and get to the real action. It’s like watching a movie and skipping all the boring exposition to get straight to the car chase. We want to see which particles are actually doing the chemistry, the ones that are getting their hands dirty, so to speak. We ignore the spectators, the ones who are just hanging out in the solution, not really participating in the main event.

Let’s start with the full, glorious, slightly overwhelming chemical equation. This is where we list everything that goes into the beaker, all the ingredients before the magic happens. For sodium carbonate, the formula is Na₂CO₃. For sulfuric acid, it’s H₂SO₄. When these two meet in water, they’re usually dissolved, which means they break apart into their individual ions. Think of them as being like a bunch of teenagers at a party, all mingling and bouncing around. Sodium carbonate, when dissolved, splits into two sodium ions (Na⁺) and one carbonate ion (CO₃²⁻). Sulfuric acid, being a strong acid, splits into two hydrogen ions (H⁺) and one sulfate ion (SO₄²⁻). So, the reactants side looks like this: 2Na⁺(aq) + CO₃²⁻(aq) + 2H⁺(aq) + SO₄²⁻(aq).
Now for the products. What do we get when this energetic pair dukes it out? We get sodium sulfate (Na₂SO₄), water (H₂O), and carbon dioxide gas (CO₂). The sodium sulfate is a salt, and it's usually soluble, meaning it stays dissolved in water, so it also splits into its ions: two sodium ions (2Na⁺) and one sulfate ion (SO₄²⁻). Water, of course, is just water (H₂O). And carbon dioxide, that's the stuff that makes your soda fizzy, so it bubbles off as a gas (CO₂). So, the products side of the full equation looks like this: 2Na⁺(aq) + SO₄²⁻(aq) + H₂O(l) + CO₂(g).
Putting it all together, the complete, unabridged, director's-cut chemical equation is: Na₂CO₃(aq) + H₂SO₄(aq) → Na₂SO₄(aq) + H₂O(l) + CO₂(g) Or, in all its dissociated glory: 2Na⁺(aq) + CO₃²⁻(aq) + 2H⁺(aq) + SO₄²⁻(aq) → 2Na⁺(aq) + SO₄²⁻(aq) + H₂O(l) + CO₂(g)

Phew! That’s a mouthful, right? It’s like reading the terms and conditions for a new app – you know there’s important stuff in there, but your eyes glaze over a bit. But we’re not done yet. We’re on a mission to find the net ionic equation. This is where we identify the spectator ions. These are the ions that appear on both sides of the equation, unchanged. They’re like the guy at the party who just stands in the corner, nursing a drink, not really interacting with anyone. They’re present, but they’re not contributing to the main drama.
In our equation, we have 2Na⁺(aq) on the reactant side and 2Na⁺(aq) on the product side. See? They’re identical. They’re just chilling, being sodium ions. And we also have SO₄²⁻(aq) on the reactant side and SO₄²⁻(aq) on the product side. More spectators! They’re not participating in the chemical fireworks. So, we get to cross them out. It's like saying, "Okay, you two, thanks for coming, but we don't need you for the main show."
Once we’ve kicked out the spectator ions, what are we left with? The real stars of the show! On the reactant side, we have the carbonate ion (CO₃²⁻) and the hydrogen ions (2H⁺). These are the ones that are going to tango. On the product side, we're left with water (H₂O) and carbon dioxide gas (CO₂). These are the results of their epic chemical dance.
So, the net ionic equation, the one that tells the true story of what's happening, is: CO₃²⁻(aq) + 2H⁺(aq) → H₂O(l) + CO₂(g)

This is the heart of the reaction! It’s the core message. It tells us that when carbonate ions and hydrogen ions meet, they transform into water and carbon dioxide. This is the fizz, the bubble, the dramatic escape of gas that you’d see if you were actually mixing these two. It’s the chemical equivalent of a mic drop.
Think about it in everyday terms. Imagine you're making a super-fizzy lemonade. You've got your lemon juice (the acid, with lots of H⁺ ions) and you're adding something like a special fizzy powder (the carbonate, with CO₃²⁻ ions). The fizz you see? That's the CO₂ escaping. The water that's left? Well, that's just water, still doing its thing. The other stuff, the things that were just dissolved in your lemonade mix or already in the water, they’re the spectator ions. They’re just there for the ride.
Another analogy: You’re at a lively party. The H⁺ ions are the super enthusiastic dancers, the life of the party, eager to mingle and get things going. The CO₃²⁻ ions are also ready to party, maybe a little more reserved at first, but they’re definitely up for some fun. When they meet, bam! They start a wild dance, and the energy released turns into water (a calm, steady presence) and carbon dioxide (the exuberant guest who can't stop talking and gesturing wildly, hence the bubbles).
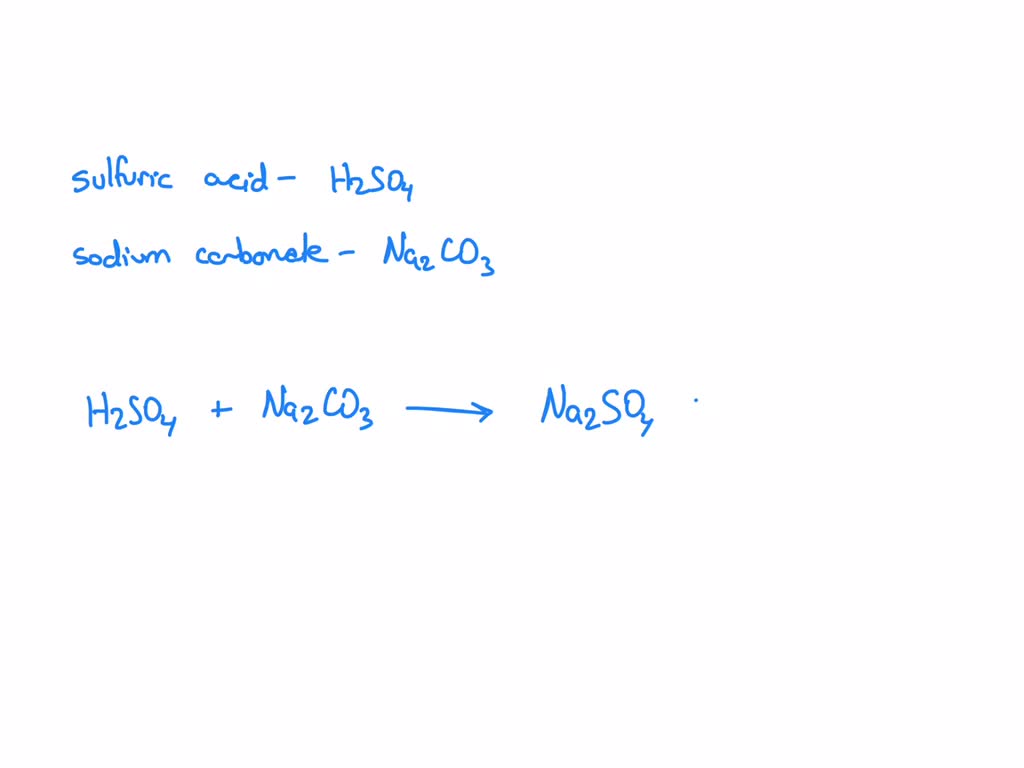
Meanwhile, the Na⁺ ions are like the couple who came to the party together, and they’re just enjoying each other’s company, maybe a little oblivious to the main dance floor drama. They’re still there, but they’re not actively joining the main dance. And the SO₄²⁻ ions? Maybe they’re like the chaperones, watching everything unfold, making sure things don’t get too out of hand, but not really part of the chaotic fun themselves. They’re present, but their role is passive.
The beauty of the net ionic equation is that it highlights the essential chemistry. It’s not about the brand of soda you used or the type of glass you're drinking from; it’s about the fundamental interaction between the acidic and the carbonate components. This reaction is crucial in many industrial processes, like in the production of glass, detergents, and even in water treatment. So, while it might seem like a complex chemical dance, it’s actually the foundation for a lot of the things we use every day.
So, the next time you see something fizzing, or you’re dealing with a particularly stubborn stain that requires a bit of chemical elbow grease, remember the net ionic equation. It’s the simplified, action-packed version of what’s really going on. It’s the core of the chemical conversation, stripping away the unnecessary chit-chat to reveal the essential dialogue. It’s a reminder that even in the most seemingly mundane situations, there’s a fascinating chemical ballet happening, complete with energetic dancers, serene observers, and the occasional dramatic exit.
And that, my friends, is the not-so-secret life of sodium carbonate and sulfuric acid, as told through the lens of a net ionic equation. It’s a reaction that’s both powerful and elegant, a testament to the fascinating world of chemistry that surrounds us, even if we sometimes only notice it when our drain starts to sing opera.
