Sodium Acetate And Silver Nitrate Net Ionic Equation

Hey there, science enthusiasts (and those who just stumbled upon this page looking for something interesting)! Ever wondered what happens when you mix two seemingly random chemicals together? Well, buckle up, buttercups, because we're diving into the fascinating world of sodium acetate and silver nitrate. It might sound a bit fancy, but trust me, it's way cooler than it sounds. Think of it like a chemistry party, and we're here to spill the tea on who's dancing with whom and who's just chilling on the sidelines.
So, first things first, let's get to know our players. We've got sodium acetate. You might have heard of it before, maybe in relation to those hand warmers that get all warm and fuzzy when you bend them. Pretty neat, right? It's basically the salt of acetic acid – the stuff that makes vinegar smell… well, like vinegar. But don't worry, we're not making pickles here today. It's a pretty common and generally harmless compound. Think of it as the friendly, approachable guest at our chemistry party.
Then we have our other star, silver nitrate. Now, this one’s a bit more of a dramatic character. It's a chemical compound that’s often used in photography (remember those old-school film cameras? Yep, this was a big part of it!). It's also got some medicinal uses, like for treating warts. But be warned, silver nitrate can stain your skin a lovely shade of brown or black if you’re not careful. So, definitely not the guy you want to spill on your favorite shirt! It's got that oomph, you know?
Now, when you mix these two buddies together, something magical happens. It’s not an explosion (phew!), but it is a reaction. And in the world of chemistry, reactions are like tiny little dances, where atoms and molecules break up, reform, and create something new. Our goal today is to figure out the net ionic equation, which is basically a fancy way of saying we want to see who the real stars of the show are in this reaction.
First, we need to look at the overall reaction. When you dissolve sodium acetate (NaC₂H₃O₂) in water, it breaks apart into its ions. Remember ions? They're like atoms or molecules that have gained or lost electrons, so they have a little electrical charge. In this case, sodium acetate splits into sodium ions (Na⁺) and acetate ions (C₂H₃O₂⁻). They're just floating around, happy as clams in their watery environment.
Similarly, when you dissolve silver nitrate (AgNO₃) in water, it also breaks apart into its ions. It gives us silver ions (Ag⁺) and nitrate ions (NO₃⁻). Again, they're all dissolved and ready to mingle. So, in our beaker, we've got a delightful cocktail of Na⁺, C₂H₃O₂⁻, Ag⁺, and NO₃⁻. A real ion party!
Now, here’s where things get interesting. The silver ions (Ag⁺) from the silver nitrate are looking for a partner. They're feeling a bit lonely. And guess who's available? The acetate ions (C₂H₃O₂⁻) are also looking for someone to hang out with. However, the silver ions are particularly attracted to the nitrate ions (NO₃⁻) and the sodium ions (Na⁺) are particularly attracted to the acetate ions. But wait, that's not quite right. Let me rephrase.

The silver ions (Ag⁺) are looking for a partner, and the acetate ions (C₂H₃O₂⁻) are also looking for a partner. But here's the key: silver ions have a stronger attraction to the acetate ions than the sodium ions do. Or rather, the silver ions have a stronger attraction to the nitrate ions than the acetate ions do. No, no, no. Let's think about what actually forms a solid. In this particular reaction, when the silver ions meet up with the acetate ions, they form a new compound. This new compound is called silver acetate (AgC₂H₃O₂). And the cool thing about silver acetate is that it’s insoluble in water. That means it doesn't dissolve. It clumps together and forms a solid, which we call a precipitate. It’s like these two ions decided they’d rather form a couple and leave the party together, creating a solid little duo.
So, what does this look like in terms of an equation? We start with our reactants, which are the things we mix together. In our case, it's sodium acetate and silver nitrate. When we write the molecular equation, it looks something like this:
NaC₂H₃O₂(aq) + AgNO₃(aq) → AgC₂H₃O₂(s) + NaNO₃(aq)
The (aq) means "aqueous," which just means dissolved in water. The (s) means "solid," which is our newly formed silver acetate precipitate. And then we have NaNO₃, which is sodium nitrate. Now, here's a little trick for you: sodium nitrate is also soluble in water. So, even though it's formed in the reaction, it just stays dissolved and keeps on swimming around.

This brings us to our next step: the complete ionic equation. This is where we show all the ions that are dissolved in the water. Remember how we said our initial solutions had Na⁺, C₂H₃O₂⁻, Ag⁺, and NO₃⁻? Well, the silver acetate precipitates out as a solid, so it stays together. But the sodium nitrate stays dissolved as ions. So, the complete ionic equation looks like this:
Na⁺(aq) + C₂H₃O₂⁻(aq) + Ag⁺(aq) + NO₃⁻(aq) → AgC₂H₃O₂(s) + Na⁺(aq) + NO₃⁻(aq)
See how everything is broken down into its individual ions, except for our solid precipitate? It's like looking at all the party guests before anyone has paired off, and then showing the final scene with the couple who decided to leave together, and everyone else still mingling.
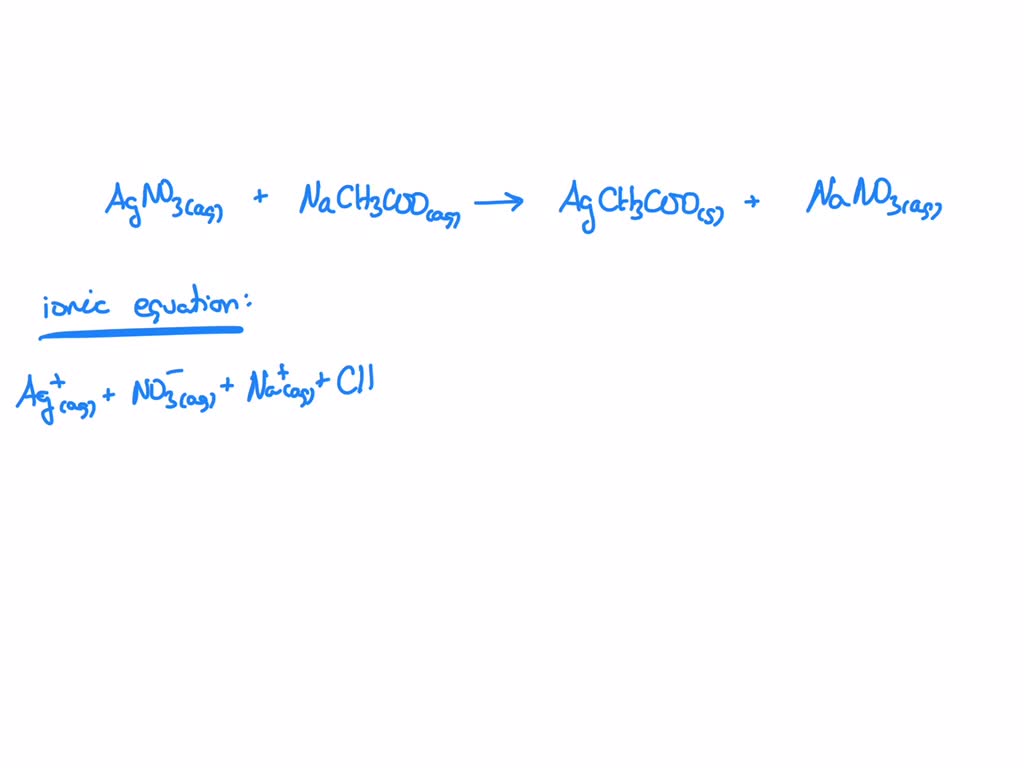
Now, for the main event: the net ionic equation. This is where we get rid of all the spectator ions. What are spectator ions, you ask? They're the ions that don't actually participate in forming the precipitate. They're just… there. Like that one person at the party who just stands in the corner, observing. In our equation, which ions are just hanging out and not really doing anything? If you look at the complete ionic equation, you'll see sodium ions (Na⁺) on both sides, and nitrate ions (NO₃⁻) on both sides. They’re the same on the reactant side as they are on the product side. They haven’t changed one bit! They’re just spectators.
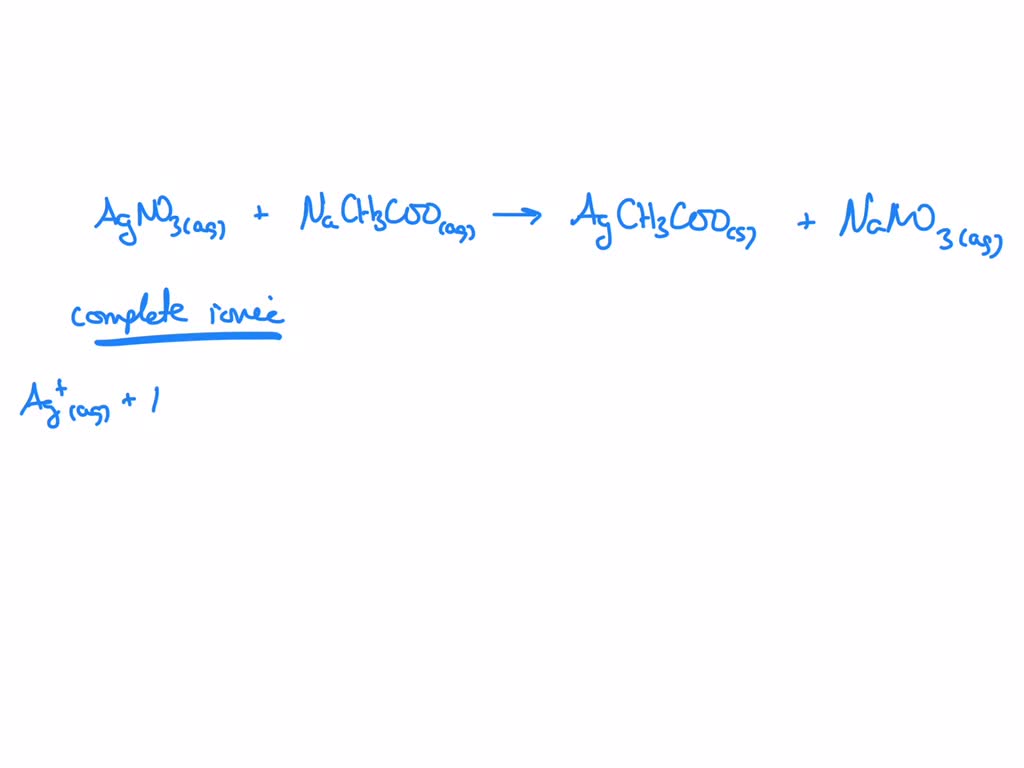
So, we’re going to cross them out. Poof! Gone! Like they never even existed. And what are we left with? The ions that actually did something, the ones that formed the solid precipitate. This is our net ionic equation, and it’s the real story of the reaction:
Ag⁺(aq) + C₂H₃O₂⁻(aq) → AgC₂H₃O₂(s)
And there you have it! The net ionic equation for the reaction between sodium acetate and silver nitrate. It’s a beautiful, simple representation of what’s truly going on at the heart of the reaction. The silver ions and the acetate ions get together and form solid silver acetate, while the sodium and nitrate ions just keep on swimming, totally unbothered.
It's like a tiny chemical soap opera. The silver ions, feeling a bit bold, see the lovely acetate ions and can't resist. They decide to elope, forming a stable, insoluble union. The sodium and nitrate ions, on the other hand, are the bystanders. They’re the friends who are happy for the couple but don't really get involved in the drama. They’re happy to just keep doing their own thing in the solution.
Understanding these net ionic equations might seem a bit niche, but it's super helpful in chemistry. It helps us focus on the essential participants in a reaction, cutting out all the unnecessary noise. It’s like getting the director’s cut of a movie, but instead of extra scenes, you’re just getting the core action. And in this case, the core action is the formation of that silver acetate precipitate.
Think of it this way: if you were at a big, noisy party, and you wanted to know who ended up dating who, you wouldn't pay attention to the guy standing by the punch bowl the whole night, would you? You'd focus on the couple who actually went for a walk outside. The net ionic equation is our way of focusing on that couple.
So, next time you hear about sodium acetate and silver nitrate, you can impress your friends with your knowledge of their ion dance. You can explain how the silver ions and acetate ions are the real MVPs of this particular chemical story, forming a solid bond while others spectate. It’s a testament to how even seemingly simple ingredients can create something entirely new and observable.
And the beauty of it all is that this is just one tiny example of the incredible reactions happening all around us, all the time. From the food we eat to the air we breathe, chemistry is constantly at play, creating and transforming. It’s a never-ending dance of atoms and molecules, and equations like our net ionic equation help us peek behind the curtain and understand the choreography.
So, don't be intimidated by the jargon. Chemistry, at its heart, is about understanding how things interact and change. And sometimes, all it takes is a little bit of mixing, a little bit of observing, and a whole lot of curiosity to uncover some pretty cool stuff. So, go forth, my friends, and may your chemical reactions always be insightful and your precipitates delightfully observable! You’ve got this!
