Sn2 Reactions Proceed Fastest With ________ Substrates.
Hey there, science curious friends! Ever feel like some things in life just… click? Like when you finally figure out how to fold a fitted sheet, or when your favorite song comes on at just the right moment? Well, in the wacky world of chemistry, there’s a similar kind of “aha!” moment, especially when we’re talking about how molecules like to play musical chairs. Today, we’re going to peek behind the curtain at something called an SN2 reaction, and it all comes down to one simple idea: Sn2 reactions proceed fastest with less crowded substrates.
Now, before you get that glazed-over look, let’s break it down. Imagine you’re at a really popular party. It’s packed! People are bumping into each other, trying to squeeze past, and getting a drink at the bar is an Olympic sport. It’s chaotic, right? Now, imagine a much smaller gathering, maybe just a few close friends in your living room. Much easier to move around, chat, and get that refill of your favorite beverage without any fuss. That, my friends, is the essence of what makes an SN2 reaction zoom!
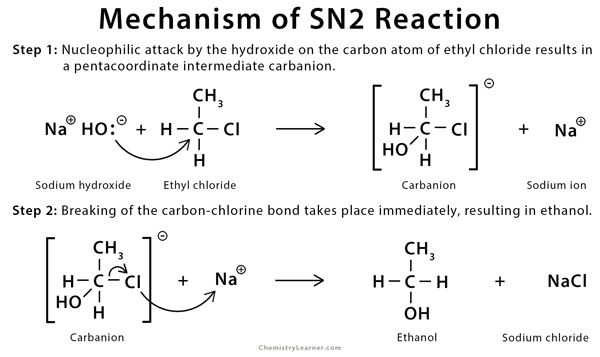
In chemistry, we have these tiny building blocks called molecules. Sometimes, these molecules need to swap out a piece, like trading a worn-out tire for a new one on a car. An SN2 reaction is one of the ways this trading can happen. Think of it as a very specific kind of hand-off.
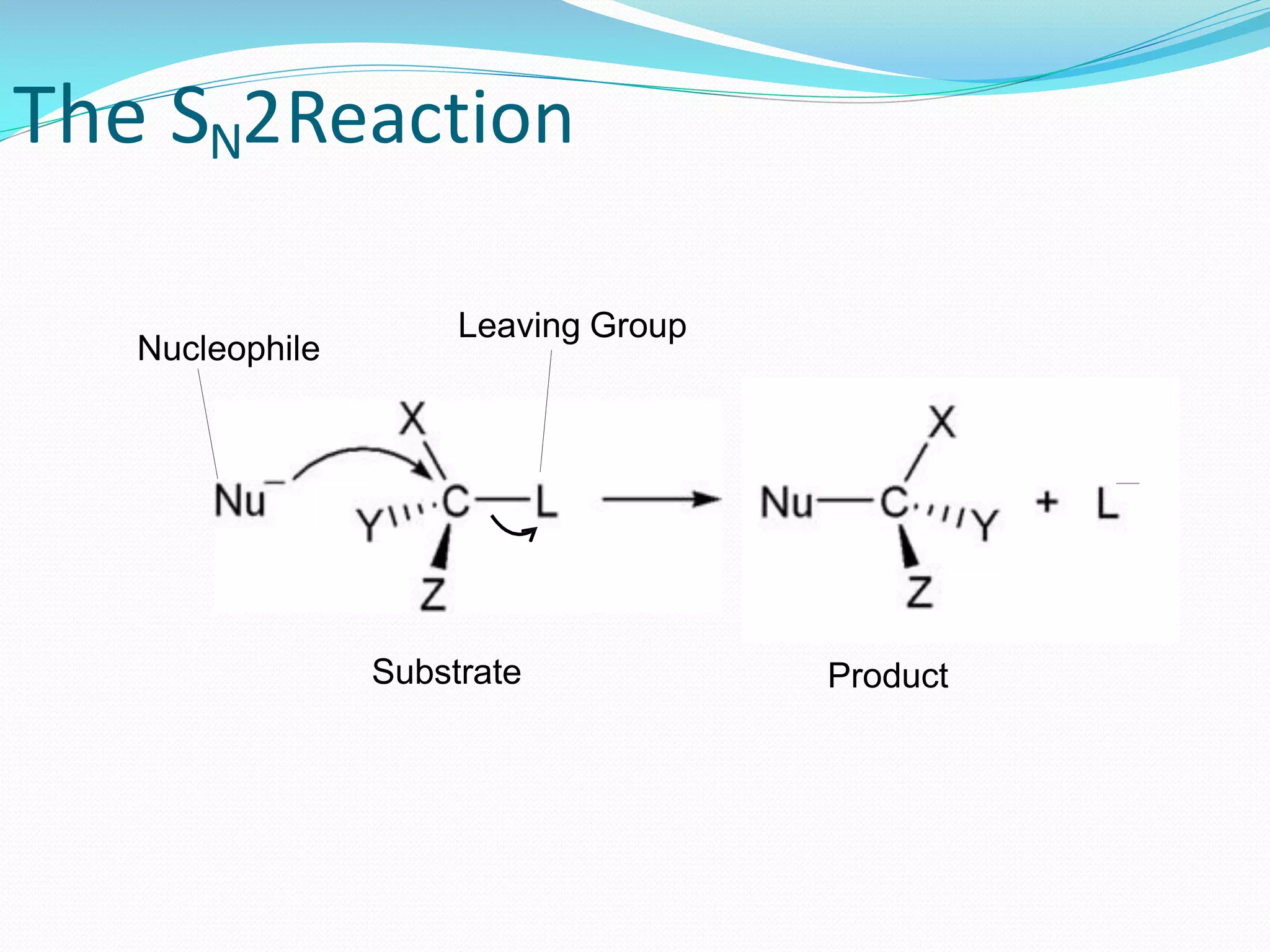
Here’s where our “less crowded” idea comes in. In an SN2 reaction, one molecule (let’s call it the “attacker”) comes in to nudge another molecule (the “host”) and simultaneously steal one of its attachments (we call these “leaving groups”). It’s like a super-efficient tag-team move. But, for this to happen smoothly, the “host” molecule needs to be easily accessible. It needs to be, well, uncluttered.
The Party Analogy, Revisited
Let’s get back to our party. The “host” molecule is like the person trying to hand something over. The “attacker” is the person trying to grab it. The “leaving group” is the item being handed over. If there are a lot of other people (other groups attached to the molecule) crowding around the “host,” it’s going to be really hard for the “attacker” to get close enough to make the hand-off happen.

Imagine trying to give your friend a high-five in the middle of a mosh pit. Not gonna happen easily, right? You need clear space. In chemistry, this “clear space” is what we call steric hindrance. It’s just a fancy way of saying “physical blocking” by bulky groups around the reaction center.
So, when we say Sn2 reactions proceed fastest with less crowded substrates, we mean molecules where the part that’s about to be attacked isn’t surrounded by a bunch of big, bulky bits. Think of it like a super-organized tidy desk versus a desk piled high with papers, coffee cups, and forgotten snacks. Which one would you rather try to find a specific paper on?
Meet the Star Players: The Substrates
The “substrate” is just the molecule that’s undergoing the reaction. And for SN2, the absolute VIPs, the ones that make the reaction happen at lightning speed, are the ones with the least amount of clutter. Let’s give them some nicknames:
Methyl: This is the king of speed! Imagine a single, tiny atom attached to our reaction center. It’s like having a vast, open ballroom. The attacker can waltz right in, no problem. This is the most reactive type of substrate for SN2. It’s like the person who is always ready to greet you at the door with open arms.
Primary (1°): Next up are primary substrates. These have our reaction center attached to just one other carbon atom (which then has a bunch of hydrogens). Think of it as a small, cozy living room. Still plenty of space for everyone to mingle comfortably. They’re super fast too, just a smidge slower than our methyl friend.

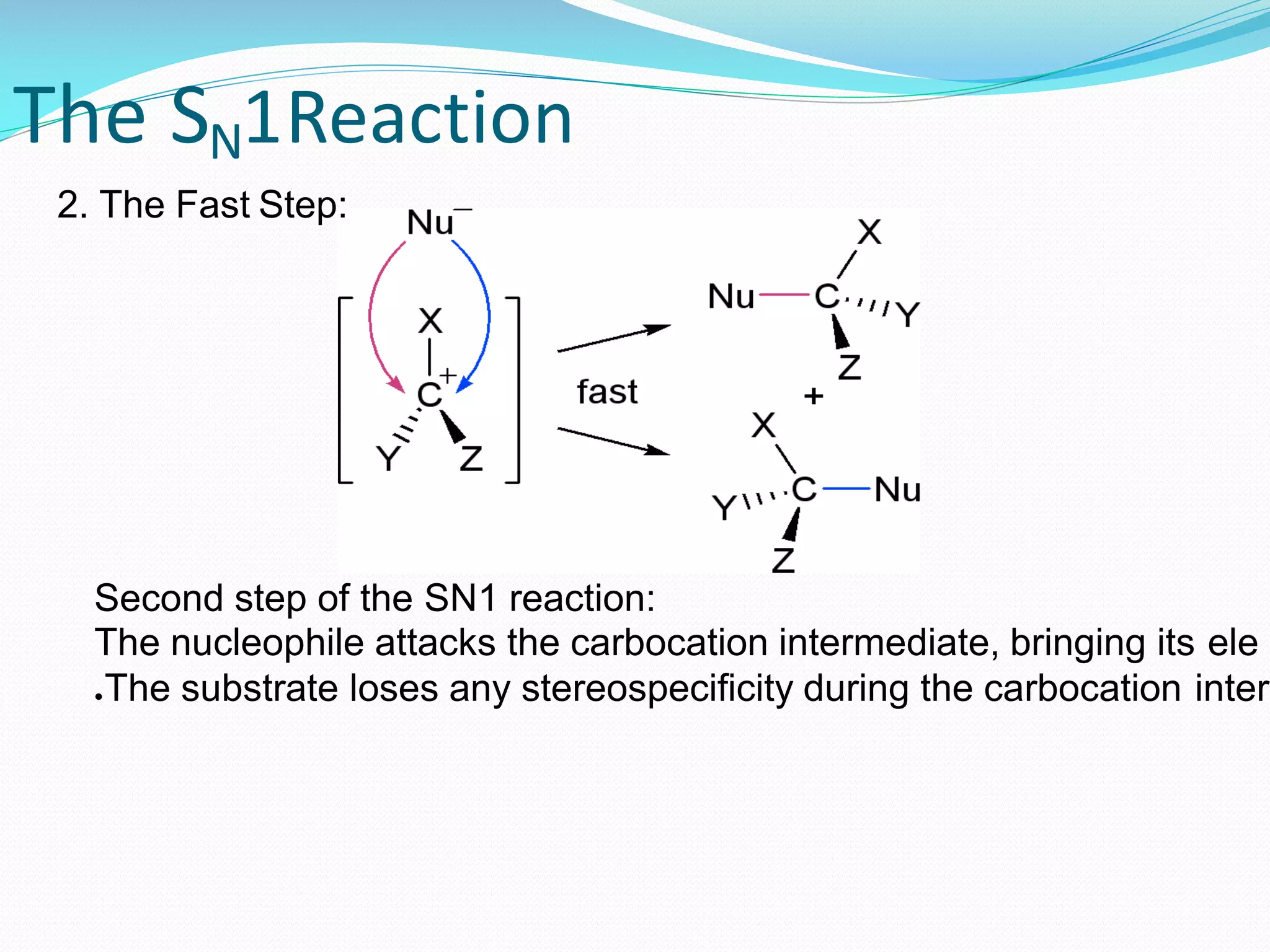
Secondary (2°): Now things start to get a little more popular. Secondary substrates have our reaction center attached to two other carbon atoms. This is like your living room starting to fill up with guests. It’s still manageable, but you might have to do a little more maneuvering. SN2 reactions still happen here, but they’re starting to slow down a bit. The attacker has to work a little harder to find its opening.
Tertiary (3°): And then we have the tertiary substrates. These have our reaction center attached to three other carbon atoms. Imagine a packed house party where everyone’s in the kitchen, trying to get to the snack table. It’s crowded! The attacker has a really tough time getting close enough to do its job. In fact, SN2 reactions barely happen, or don’t happen at all, with tertiary substrates. It’s like trying to deliver a secret message in the middle of a rock concert – just too much noise and chaos.
Why Should You Even Care?
Okay, so why is this whole “crowdedness” thing important? Well, these SN2 reactions are actually super common and incredibly useful in making all sorts of things we use every day! From the medicines that help us feel better to the materials that make up our phones and clothes, chemists use these reactions to build complex molecules.
Understanding how to make these reactions go fast and efficiently is like having a superpower for chemists. It means they can create new medicines, develop better materials, and solve all sorts of scientific puzzles faster and more effectively. It’s about making the tools they need to improve our lives.
Think about it: if you’re trying to bake a cake and the recipe calls for you to add a specific ingredient, you want it to be easy to add, right? You don’t want to have to excavate a pile of flour just to sprinkle in some baking soda. SN2 reactions are similar. We want that attacker to easily find its target and make the swap without a struggle.
So, the next time you hear about an SN2 reaction, remember the party analogy. The secret to a speedy reaction is a less crowded party, or in chemistry terms, a less hindered substrate. It’s a simple concept with a huge impact on the world around us. It’s all about making those molecular interactions smooth, efficient, and successful!
